Triacylglycerol Synthesis Pathways Decoded: Kennedy vs. Acyl-CoA Independent Mechanisms in Health and Disease
This comprehensive review delineates the two principal pathways for triacylglycerol (TAG) biosynthesis: the canonical Kennedy pathway and the alternative acyl-CoA independent route.
Triacylglycerol Synthesis Pathways Decoded: Kennedy vs. Acyl-CoA Independent Mechanisms in Health and Disease
Abstract
This comprehensive review delineates the two principal pathways for triacylglycerol (TAG) biosynthesis: the canonical Kennedy pathway and the alternative acyl-CoA independent route. Targeted at researchers, scientists, and drug development professionals, it provides foundational knowledge on enzymatic steps and cellular localization, explores methodological approaches for pathway-specific study, addresses common experimental challenges in distinguishing pathway contributions, and offers a critical comparative analysis of their validation, physiological roles, and dysregulation in metabolic diseases. The synthesis aims to inform targeted therapeutic strategies for lipid-associated disorders.
Understanding TAG Assembly: Core Enzymes, Cellular Compartments, and Historical Discovery of Dual Pathways
Triacylglycerols (TAGs) are neutral lipids consisting of a glycerol backbone esterified with three fatty acyl chains. They serve as the primary storage form of metabolic energy in eukaryotes, providing more than twice the energy yield per gram compared to carbohydrates or proteins. Within the context of metabolic disease research, particularly non-alcoholic fatty liver disease (NAFLD) and obesity, the molecular pathways governing TAG synthesis are critical therapeutic targets. This whitepaper details the core aspects of TAG biology, framed by the ongoing investigative tension between the canonical Kennedy pathway and emerging acyl-CoA-independent pathways, highlighting methodologies and reagents pivotal for contemporary research.
Core Structure and Quantitative Composition
TAG structure is defined by the chemical nature and positional distribution (sn-1, sn-2, sn-3) of its fatty acyl chains. This composition directly influences its physical properties and metabolic fate.
Table 1: Common Fatty Acyl Constituents of Mammalian Triacylglycerols
| Fatty Acid (Common Name) | Systematic Name | Chain Length:Double Bonds | Typical Sn-Position Preference | Approximate Percentage in Adipose TAG* |
|---|---|---|---|---|
| Palmitic | Hexadecanoic | 16:0 | sn-1, sn-3 | 20-30% |
| Stearic | Octadecanoic | 18:0 | sn-1 | 2-6% |
| Oleic | cis-9-Octadecenoic | 18:1 | sn-1, sn-2, sn-3 | 40-50% |
| Linoleic | cis,cis-9,12-Octadecadienoic | 18:2 | sn-2 | 10-20% |
*Values are approximate and vary by diet and tissue. Data compiled from recent lipidomic studies.
Primary Functions and Metabolic Significance
TAGs are stored in dedicated organelles: lipid droplets in non-adipose cells and adipocytes in white adipose tissue. Beyond energy storage, TAG synthesis and hydrolysis are integral to membrane homeostasis, signal transduction, and acting as a sink for excess fatty acids, protecting against lipotoxicity. Dysregulated TAG metabolism is a hallmark of metabolic syndrome.
Synthesis Pathways: Kennedy vs. Acyl-CoA-Independent
Kennedy (Glycerol-3-Phosphate) Pathway: The established, primary route for de novo TAG synthesis in most tissues. It is an acyl-CoA-dependent process occurring in the endoplasmic reticulum (ER) membrane.
Table 2: Enzymatic Steps of the Kennedy Pathway
| Step | Enzyme (Abbreviation) | EC Number | Substrate(s) | Product | Compartment | Key Inhibitors/Modulators |
|---|---|---|---|---|---|---|
| 1 | Glycerol-3-phosphate acyltransferase (GPAT) | 2.3.1.15 | G3P, acyl-CoA | Lysophosphatidic acid (LPA) | ER/ Mitochondria | FSG67 (GPAT1 inhibitor) |
| 2 | 1-Acylglycerol-3-phosphate acyltransferase (AGPAT) | 2.3.1.51 | LPA, acyl-CoA | Phosphatidic acid (PA) | ER | - |
| 3 | Phosphatidate phosphatase (Lipin) | 3.1.3.4 | PA | Diacylglycerol (DAG) | ER (translocates) | Propranolol (non-specific) |
| 4 | Diacylglycerol acyltransferase (DGAT) | 2.3.1.20 | DAG, acyl-CoA | Triacylglycerol (TAG) | ER | DGAT1i (e.g., AZD7687), DGAT2 ASOs |
Acyl-CoA-Independent Pathways: Complementary pathways that recycle pre-existing membrane phospholipids or utilize other activated donors, gaining prominence in contexts of high lipolytic flux or specific tissues.
- Leci thin:Cholesterol Acyltransferase (LCAT)-like Activity: Uses phosphatidylcholine (PC) as an acyl donor.
- Acyl-CoA:Cholesterol Acyltransferase (ACAT)-like Activity: Can utilize cholesterol esters under certain conditions.
- Monoacylglycerol Acyltransferase (MGAT) Pathway: Primarily in the intestine, utilizes 2-MAG from dietary fat digestion. MGAT3 shows DGAT2-independent TAG synthesis capability.
Key Experimental Protocols
Protocol 1: In Vitro DGAT Activity Assay (Microsomal Fraction) Objective: Measure DGAT1 vs. DGAT2 activity from tissue/cell lysates. Methodology:
- Microsome Preparation: Homogenize liver tissue or cells in ice-cold sucrose buffer (0.25 M sucrose, 10 mM HEPES, pH 7.4). Centrifuge at 12,000 x g for 15 min (4°C). Collect supernatant and ultracentrifuge at 100,000 x g for 60 min (4°C). Resuspend pellet (microsomal fraction) in storage buffer.
- Reaction Setup: Prepare two parallel reaction mixes in a final volume of 200 µL: 50 µg microsomal protein, 100 µM 1,2-dioleoyl-sn-glycerol (DAG substrate), 50 µM [¹⁴C]oleoyl-CoA (acyl donor). For specificity, pre-incubate samples with 10 µM DGAT1 inhibitor (T863) or DGAT2 inhibitor (PF-06424439).
- Incubation & Extraction: Incubate at 37°C for 10 min. Stop reaction with 1.5 mL chloroform:methanol (2:1). Add 0.3 mL 0.9% KCl, vortex, and centrifuge. Collect organic (lower) phase.
- Separation & Quantification: Spot lipid extract on a TLC plate (Silica Gel 60). Develop in hexane:diethyl ether:acetic acid (70:30:1). Visualize standards, scrape TAG bands, and quantify radioactivity by liquid scintillation counting.
Protocol 2: Tracing Acyl Flux via Stable Isotopes (LC-MS/MS) Objective: Distinguish Kennedy pathway-derived TAG from acyl-CoA-independent synthesis. Methodology:
- Cell Labeling: Treat hepatocytes (e.g., HepG2 or primary) with 100 µM deuterated palmitate (D31-C16:0) and 500 µM unlabeled glycerol (to trace glycerol backbone) or vice-versa.
- Lipid Extraction: After 4-24h, wash cells and lyse. Perform Bligh & Dyer lipid extraction.
- Mass Spectrometry Analysis: Reconstitute lipids in methanol:chloroform. Inject onto a reverse-phase C18 column coupled to a triple quadrupole mass spectrometer. Use multiple reaction monitoring (MRM) for TAG species.
- Data Interpretation: Calculate isotopic enrichment (M+31/M+0 ratio for palmitate-labeled TAG). Compare incorporation kinetics into TAG vs. phospholipid precursors (PA, PC) to infer pathway preference.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for TAG Synthesis Pathway Research
| Reagent / Material | Function / Application | Example Product / Cat. No. (Vendor) |
|---|---|---|
| T863 (DGAT1 Inhibitor) | Selective chemical inhibition of DGAT1 enzyme to delineate its contribution to total cellular TAG synthesis. | Cayman Chemical #13228 |
| PF-06424439 (DGAT2 Inhibitor) | Potent and selective inhibitor of DGAT2 for functional studies. | MedChemExpress #HY-15801 |
| [¹⁴C]Oleoyl-CoA / [³H]Glycerol | Radiolabeled substrates for in vitro enzyme assays or in vivo metabolic flux studies. | PerkinElmer #NEC-691 |
| Deuterated Fatty Acids (D31-Palmitate) | Stable isotope tracers for LC-MS/MS-based flux analysis and lipidomic profiling. | Cambridge Isotope Laboratories #DLM-215 |
| siRNA Pools (DGAT1, DGAT2, AGPATs) | RNA-mediated knockdown for genetic validation of protein function in cell models. | Dharmacon ON-TARGETplus |
| Lipin-1 (PAP) Activity Assay Kit | Colorimetric measurement of phosphatidate phosphatase activity in cell lysates. | Cell Biolabs #MET-5032 |
| Lipid Extraction Kit (Bligh & Dyer or MTBE method) | Standardized, high-recovery total lipid extraction from cells/tissues. | Avanti # 850404 |
| C18 Solid Phase Extraction (SPE) Columns | Clean-up and fractionation of neutral lipids (TAG) from complex lipid extracts prior to MS. | Waters #WAT020515 |
Visualizations
Diagram 1: The Kennedy (Acyl-CoA-Dependent) Pathway
Diagram 2: Alternative TAG Synthesis Pathways
Conclusion The structural definition of TAG belies its complex metabolic life cycle. The dominant Kennedy pathway and auxiliary acyl-CoA-independent mechanisms are not mutually exclusive but operate in a tissue- and nutrient-state-dependent network. Disentangling their relative contributions through the methodologies outlined is essential for developing pathway-specific therapeutics aimed at correcting pathogenic TAG accumulation without disrupting essential lipid homeostasis.
Within the ongoing research thesis comparing mechanisms of triacylglycerol (TAG) synthesis, the Kennedy pathway remains the canonical, acyl-CoA-dependent route. This in-depth guide details its enzymatic machinery, regulatory nodes, and experimental interrogation, providing a technical reference for researchers investigating lipid metabolism in metabolic disease and drug development.
The synthesis of triacylglycerols is fundamental to energy homeostasis and is dysregulated in diseases such as obesity, NAFLD, and diabetes. The central research dichotomy contrasts the acyl-CoA-dependent Kennedy pathway with acyl-CoA-independent mechanisms (e.g., acyl remodeling via phospholipids, MGAT/DGAT activities with non-CoA donors). This whitepaper focuses on the former, providing a definitive enzymatic walkthrough of the canonical four-step sequence from glycerol-3-phosphate (G3P) to TAG, essential for understanding its metabolic flux control and therapeutic targeting.
Enzymatic Walkthrough of the Kennedy Pathway
The pathway occurs primarily in the endoplasmic reticulum and involves four sequential acylations.
Step 1: Synthesis of Lysophosphatidic Acid (LPA)
- Enzyme: Glycerol-3-phosphate acyltransferase (GPAT), isoforms GPAT1-4.
- Reaction: Acyl-CoA + sn-glycerol-3-phosphate → Lysophosphatidic acid (LPA) + CoASH.
- Key Detail: GPAT1 (mitochondrial) and GPAT4 (ER) are major isoforms. This is the first committed and rate-influencing step.
Step 2: Synthesis of Phosphatidic Acid (PA)
- Enzyme: 1-Acylglycerol-3-phosphate acyltransferase (AGPAT), isoforms AGPAT1-11.
- Reaction: LPA + Acyl-CoA → Phosphatidic Acid (PA) + CoASH.
- Key Detail: AGPAT2 mutations cause congenital generalized lipodystrophy, underscoring pathway necessity.
Step 3: Dephosphorylation to Diacylglycerol (DAG)
- Enzyme: Lipin (PAP) enzymes, isoforms Lipin 1-3.
- Reaction: Phosphatidic Acid (PA) → Diacylglycerol (DAG) + Pi.
- Key Detail: Lipin-1 also acts as a transcriptional co-regulator, linking synthesis to gene expression.
Step 4: Acylation to Triacylglycerol (TAG)
- Enzyme: Diacylglycerol acyltransferase (DGAT), isoforms DGAT1 & DGAT2.
- Reaction: DAG + Acyl-CoA → Triacylglycerol (TAG) + CoASH.
- Key Detail: DGAT1 and DGAT2 are non-redundant, with distinct roles and therapeutic profiles. DGAT2 is often linked to TAG synthesis de novo.
Pathway Diagram
Diagram 1: The Four-Step Kennedy Pathway for TAG Synthesis.
Quantitative Data & Kinetic Parameters
Key enzymatic constants highlight regulatory points and isoform differences.
Table 1: Kinetic Parameters of Core Kennedy Pathway Enzymes
| Enzyme (Major Isoform) | Km for Acyl-CoA (μM) | Km for Glycerol Backbone | Preferred Acyl Chain Length | Inhibitors (Example) |
|---|---|---|---|---|
| GPAT1 (mt) | 50-100 (palmitoyl-CoA) | 20-40 μM (G3P) | C16:0, C18:1 | FSG67, specific antisense oligonucleotides |
| AGPAT2 (ER) | ~10 (oleoyl-CoA) | 30-50 μM (LPA) | C18:1 | DGAT2-inhibitors show cross-activity |
| Lipin-1 (PAP) | N/A (not substrate) | ~0.5 mM (PA) | - | Propranolol, Mg²⁺ depletion |
| DGAT1 | 8-12 (oleoyl-CoA) | 10-20 μM (DAG) | Broad (C12-C22) | T863, A922500 |
| DGAT2 | 3-5 (palmitoyl-CoA) | 5-10 μM (DAG) | C16:0 | PF-06424439 |
Table 2: Comparative Flux Distribution (Model HepG2 Cells)
| Pathway Step | Estimated Relative Flux (%) | Primary Regulatory Mechanism |
|---|---|---|
| G3P Supply (GK/GPD) | 100% (baseline) | Insulin, substrate availability |
| GPAT Reaction | ~15-20% (major flux control point) | Malonyl-CoA inhibition, enzyme expression |
| AGPAT Reaction | ~80-85% of LPA flux | Isoform specificity, substrate channeling |
| PA to DAG (Lipin) | ~90% of PA flux | Cytoplasmic-nuclear shuttling, phosphorylation |
| DAG to TAG (DGAT) | ~70% of DAG flux | DGAT2 activity linked to new lipid droplet formation |
Key Experimental Protocols
In Vitro TAG Synthesis Assay (Microsomal Fraction)
- Purpose: Measure combined or individual Kennedy pathway enzyme activities.
- Protocol:
- Preparation: Isolate microsomes from liver/tissue/cultured cells via differential centrifugation (100,000 x g pellet).
- Reaction Mix: 50-100 μg microsomal protein, 100 mM Tris-HCl (pH 7.4), 1-5 μM radiolabeled ([¹⁴C] or [³H]) acyl-CoA or glycerol-3-phosphate, 5 mM MgCl₂, 1 mg/mL fatty acid-free BSA, in 200 μL total volume.
- Incubation: 37°C for 5-15 minutes (linear rate period).
- Lipid Extraction: Stop reaction with 3:2 (v/v) heptane:isopropanol. Extract lipids via Folch method (CHCl₃:MeOH, 2:1).
- Separation & Quantification: Separate lipids by TLC (hexane:diethyl ether:acetic acid, 70:30:1). Visualize standards, scrape TAG (or PA, DAG) bands, quantify by liquid scintillation counting.
- Key Controls: Zero-time point, heat-denatured enzyme, no-substrate control.
Genetic Knockdown/CRISPR-Cas9 Validation of Pathway Flux
- Purpose: Establish necessity of specific isoforms for de novo TAG synthesis.
- Protocol:
- Gene Targeting: Design siRNAs (for knockdown) or sgRNAs (for CRISPR KO) targeting human GPAM, AGPAT2, LPIN1, DGAT1/2.
- Cell Transfection/Transduction: Deliver constructs into hepatocyte model (HepG2, primary hepatocytes) using lipid nanoparticles or lentivirus.
- Validation: Confirm knockdown/KO via qPCR (mRNA) and Western blot (protein) at 48-72h post-treatment.
- Functional Assay: Pulse cells with [¹⁴C]acetate or [¹⁴C]glycerol for 2-4h. Extract total lipids, separate by TLC, and quantify radiolabel incorporated into TAG versus phospholipids to determine pathway-specific flux.
Experimental Workflow Diagram
Diagram 2: Workflow for Measuring TAG Synthesis Flux.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Reagents for Kennedy Pathway Investigation
| Reagent/Solution | Function & Application | Key Consideration |
|---|---|---|
| Radiolabeled Substrates: [¹⁴C(U)]-Glycerol, [³H]-Acyl-CoAs | Tracing de novo glycerol backbone or specific acylation steps. | Specific activity and purity critical for kinetic assays. |
| Acyl-CoA Regenerating System (CoASH, ATP, Mg²⁺, Acyl-CoA Synthetase) | Maintains constant acyl-CoA concentration in long assays. | Prevents feedback inhibition from accumulating CoASH. |
| Isoform-Selective Inhibitors: T863 (DGAT1), PF-06424439 (DGAT2) | Pharmacological dissection of terminal step contributions. | Verify selectivity in your model; off-target effects common. |
| Fatty Acid-Free Bovine Serum Albumin (BSA) | Binds free fatty acids and acyl-CoAs, prevents micelle formation. | Essential for accurate kinetics; standardize concentration. |
| Silica Gel 60 TLC Plates | High-resolution separation of neutral lipids (TAG, DAG) and phospholipids (PA, LPA). | Pre-run plates in solvent system to remove impurities. |
| Lipin Activity Buffer (CTP:phosphocholine cytidylyltransferase assay) | Measures PAP activity indirectly via CDP-choline production. | More specific than general phosphatase assays. |
| GPAT/AGPAT Activity Detection Kits (colorimetric, non-radioactive) | Measures CoASH release via DTNB (Ellman's reagent). | Suitable for high-throughput screening; may be less sensitive. |
| CRISPR/Cas9 Knockout Pool (e.g., for DGAT1, GPAM) | Create stable knockout cell lines for flux redistribution studies. | Always sequence-validate clones; account for compensatory effects. |
The synthesis of triacylglycerol (TAG) is a central metabolic process for energy storage and lipid homeostasis. The canonical Kennedy (or sn-glycerol-3-phosphate) pathway represents the primary de novo biosynthetic route, utilizing activated fatty acyl-CoA esters. This pathway operates sequentially in the endoplasmic reticulum (ER) membrane and involves four key enzymes: Glycerol-3-phosphate acyltransferase (GPAT), 1-Acylglycerol-3-phosphate acyltransferase (AGPAT), Phosphatidic Acid Phosphatase (PAP, also known as Lipin), and Diacylglycerol acyltransferase 1 (DGAT1). In contrast, acyl-CoA independent pathways, such as those mediated by DGAT2 (which may use alternative acyl donors) or the phospholipid:diacylglycerol acyltransferase (PDAT) pathway in yeast and plants, provide complementary or alternative mechanisms for TAG assembly, particularly under conditions of lipid re-modeling or acyl-CoA limitation. Current research is focused on dissecting the distinct roles, regulation, and contributions of these pathways to whole-organism lipid metabolism, with significant implications for treating metabolic diseases, cancer, and for bioengineering.
The Enzymatic Cascade: Functions, Isoforms, and Regulation
Glycerol-3-Phosphate Acyltransferase (GPAT)
GPAT catalyzes the initial and committing step: the acylation of sn-glycerol-3-phosphate (G3P) at the sn-1 position, producing lysophosphatidic acid (LPA). Mammals possess four GPAT isoforms with distinct subcellular localizations and roles.
- GPAT1 & 2: Mitochondrial-associated enzymes (ER-bound). GPAT1 is the most studied, is insulin-sensitive, and its activity correlates with TAG synthesis rates.
- GPAT3 & 4: ER-resident enzymes. GPAT4 (also called AGPAT6) is implicated in adipose lipid synthesis and skin barrier function.
1-Acylglycerol-3-Phosphate Acyltransferase (AGPAT)
AGPAT (also called LPAAT) catalyzes the acylation of LPA at the sn-2 position to yield phosphatidic acid (PA). The AGPAT family comprises multiple isoforms (AGPAT1-11 in humans) with tissue-specific expression and potential roles in signal lipid generation beyond TAG synthesis.
Phosphatidic Acid Phosphatase (PAP/Lipin)
This Mg²⁺-dependent enzyme dephosphorylates PA to produce diacylglycerol (DAG), a critical branch-point intermediate for TAG and phospholipid synthesis. The Lipin family (Lipin 1, 2, 3) acts as both a metabolic enzyme and a transcriptional co-regulator, linking lipid synthesis to gene expression.
Diacylglycerol Acyltransferase 1 (DGAT1)
DGAT1 catalyzes the final and often rate-limiting step: the acyl-CoA-dependent acylation of DAG to form TAG. It is an integral ER membrane protein and a major therapeutic target for obesity, type 2 diabetes, and non-alcoholic fatty liver disease (NAFLD).
Table 1: Key Enzymes of the Kennedy Pathway
| Enzyme | EC Number | Major Isoforms (Human) | Subcellular Localization | Substrate | Product | Key Inhibitors/Modulators (Research Tools) |
|---|---|---|---|---|---|---|
| GPAT | 2.3.1.15 | GPAT1 (GPAM), GPAT2, GPAT3, GPAT4 | Mitochondria (GPAT1,2), ER (GPAT3,4) | G3P, Acyl-CoA | LPA | FSG67 (GPAT1 inhibitor), siRNA/shRNA knockdown |
| AGPAT | 2.3.1.51 | AGPAT1, AGPAT2, AGPAT3, etc. | ER | LPA, Acyl-CoA | PA | CT-32501 (AGPAT2 inhibitor), thiol-reactive reagents |
| PAP/Lipin | 3.1.3.4 | Lipin1 (LPIN1), Lipin2, Lipin3 | Cytosol (translocates to ER) | PA | DAG | Propranolol (non-specific PAP inhibitor), Li⁺ ions |
| DGAT1 | 2.3.1.20 | DGAT1 | ER | DAG, Acyl-CoA | TAG | T863 (specific inhibitor), DGAT1-IN-1, Pradigastat |
Experimental Methodologies for Pathway Analysis
In VitroEnzyme Activity Assays
Principle: Measure the incorporation of radio-labeled or fluorescent substrates into products using isolated microsomes or purified recombinant enzymes.
Protocol for DGAT1 Activity Assay (Radioactive):
- Microsome Preparation: Homogenize tissues or lyse cultured cells in ice-cold homogenization buffer (250 mM sucrose, 10 mM Tris-HCl pH 7.4, 1 mM EDTA). Perform differential centrifugation (10,000 x g for 15 min, then 100,000 x g for 60 min) to pellet the microsomal (ER) fraction.
- Reaction Setup: In a tube, combine 50-100 µg microsomal protein, 100 µM sn-1,2-dioleoylglycerol (sonicated in 5% fatty acid-free BSA), and 50 µM [¹⁴C]oleoyl-CoA (55 mCi/mmol) in assay buffer (100 mM Tris-HCl pH 7.4, 5 mM MgCl₂).
- Incubation: Incubate at 37°C for 10-20 minutes. Terminate the reaction by adding 1.5 mL chloroform:methanol (2:1 v/v).
- Lipid Extraction: Add 0.3 mL of 0.9% KCl, vortex, and centrifuge. The lower organic phase contains lipids.
- Separation & Detection: Spot the organic phase on a TLC plate. Develop in hexane:diethyl ether:acetic acid (70:30:1). Expose plate to a phosphorimager screen or scrape TAG spots for scintillation counting.
Genetic Manipulation: Knockout/Knockdown Studies
Principle: Use siRNA (transient) or CRISPR/Cas9 (stable) to deplete specific enzymes and analyze metabolic consequences. Protocol for CRISPR/Cas9 DGAT1 Knockout in HEK293 Cells:
- Guide RNA Design: Design a 20-nt sgRNA targeting an early exon of human DGAT1 (e.g., using online tools like CHOPCHOP).
- Transfection: Co-transfect HEK293 cells with a plasmid expressing Cas9 and the sgRNA, or a pre-assembled ribonucleoprotein (RNP) complex.
- Selection & Cloning: Apply puromycin selection (if plasmid-based). Isolate single cells by serial dilution into 96-well plates to generate clonal populations.
- Genotype Validation: Extract genomic DNA. PCR-amplify the target region and sequence to confirm frameshift indels.
- Phenotype Validation: Confirm loss of DGAT1 protein via western blot (using anti-DGAT1 antibody) and functional assay (reduced [¹⁴C]oleate incorporation into TAG).
Stable Isotope Tracer Analysis for Flux Determination
Principle: Use mass spectrometry to track the incorporation of ¹³C-labeled precursors (e.g., ¹³C-glucose or ¹³C-acetate) into pathway intermediates and TAG. Protocol: Incubate cells with [U-¹³C]glucose for 0-24 hours. Extract lipids, derivatize if necessary, and analyze by LC-MS/MS. Calculate isotopic enrichment in G3P, PA, DAG, and TAG pools to infer relative flux through the Kennedy pathway versus acyl-CoA independent routes.
Visualizing the Pathway and Experimental Workflow
Diagram 1: Kennedy Pathway vs. Alternative TAG Synthesis.
Diagram 2: Core Lipid Metabolism Experiment Workflow.
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Kennedy Pathway Research
| Reagent Category | Specific Example(s) | Function & Application |
|---|---|---|
| Chemical Inhibitors | T863 (DGAT1i), FSG67 (GPAT1i), Propranolol (PAPi) | Pharmacological disruption of specific enzymatic steps to assess function and metabolic consequence. |
| Radioactive Tracers | [¹⁴C]Oleoyl-CoA, [³H]Glycerol, [³H]Oleic Acid | Substrates for in vitro activity assays or in vivo metabolic labeling to measure synthetic flux. |
| Stable Isotope Tracers | [U-¹³C]Glucose, [¹³C]Acetate, ²H₂O (Deuterated water) | Tracing de novo lipogenesis and pathway flux via MS-based metabolomics/lipidomics without radiation. |
| siRNA/shRNA Libraries | ON-TARGETplus siRNA pools (Dharmacon) | Transient or stable gene knockdown to validate enzyme-specific roles in cellular models. |
| CRISPR/Cas9 Tools | sgRNA vectors, RNP complexes (e.g., from IDT) | Generation of stable knockout cell lines for phenotypic and biochemical analysis. |
| Activity Assay Kits | DGAT Activity Assay Kit (colorimetric/fluorometric) | Non-radioactive, convenient measurement of enzyme activity in lysates. |
| Lipid Standards | DAG (16:0/18:1), TAG (tri-17:0), PA (16:0/18:1) (Avanti Polar Lipids) | Internal standards for mass spectrometry quantification and calibration for TLC. |
| Antibodies | Anti-DGAT1 (Cell Signaling, ab clone EPR6147), Anti-Lipin1 (Proteintech) | Validation of protein expression and localization via western blot or immunofluorescence. |
| Lipid Extraction Solvents | Chloroform, Methanol (HPLC grade), Methyl-tert-butyl ether (MTBE) | Critical for high-efficiency, reproducible lipid extraction from biological samples. |
For decades, the Kennedy pathway has been considered the canonical and principal route for the de novo biosynthesis of triacylglycerols (TAGs) in eukaryotes. This pathway relies on the sequential acylation of a glycerol-3-phosphate backbone using acyl-CoA donors. However, recent research has unveiled a complementary and quantitatively significant acyl-CoA independent pathway, fundamentally challenging our understanding of lipid homeostasis. This whitepaper details the discovery, core enzymatic principles, and experimental delineation of this pathway, framing it within the broader thesis of its metabolic competition and cooperation with the Kennedy pathway. Understanding this dichotomy is crucial for researchers and drug development professionals targeting metabolic diseases, cancer, and lipid storage disorders.
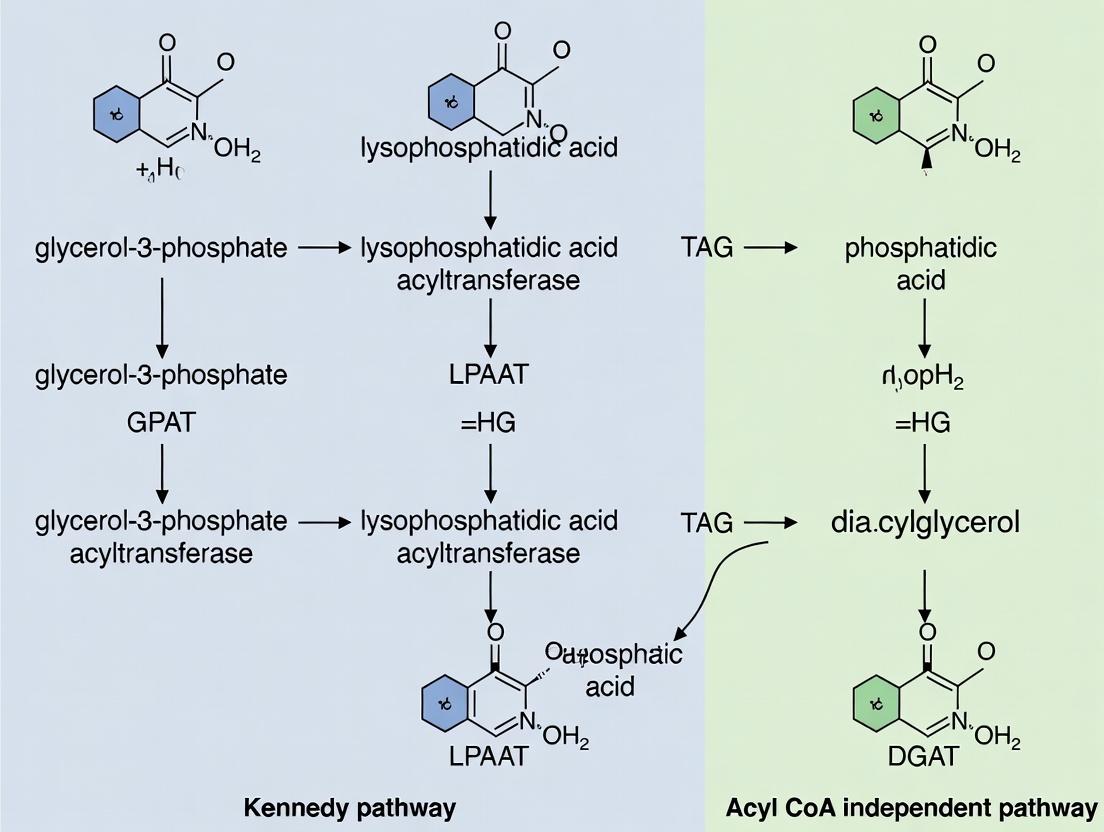
The Kennedy (or glycerophosphate) pathway is a well-characterized, three-step enzymatic process located primarily in the endoplasmic reticulum. Its core reaction sequence is:
- Acylation: Glycerol-3-phosphate (G3P) is acylated by GPAT (Glycerol-3-phosphate acyltransferase) to form lysophosphatidic acid (LPA).
- Second Acylation: LPA is acylated by AGPAT (1-acylglycerol-3-phosphate O-acyltransferase) to form phosphatidic acid (PA).
- Dephosphorylation & Final Acylation: PA is dephosphorylated by Lipin (PA phosphatase) to form diacylglycerol (DAG), which is then acylated by DGAT (Diacylglycerol acyltransferase) to form TAG.
The defining feature of this pathway is its absolute dependence on activated fatty acyl-CoA thioesters as donors.
The discovery of an acyl-CoA independent pathway emerged from observations of residual TAG synthesis in cells or models where Kennedy pathway enzymes (particularly DGAT1/2) were genetically or pharmacologically inhibited. This pointed to the existence of alternative mechanisms. The core principle of this pathway is the utilization of pre-existing membrane phospholipids, notably phosphatidylcholine (PC), as acyl donors, transferring a fatty acid directly to DAG to form TAG without the intermediate formation of acyl-CoA.
Core Enzymatic Machinery: The LCAT Family and Beyond
The primary enzymatic activity responsible for acyl-CoA independent TAG synthesis is attributed to enzymes within the Lecithin:Cholesterol Acyltransferase (LCAT) family.
- DGAT1 & DGAT2: Kennedy pathway enzymes (CoA-dependent).
- DGAT3: A soluble, plant-specific acyl-CoA dependent enzyme.
- DGAT1/2-Independent Activity: The residual activity led to the investigation of other protein families.
The key player is LPCAT3 (Lysophosphatidylcholine Acyltransferase 3), which, in addition to its primary re-acylation role in Lands' cycle, can operate in reverse under certain metabolic conditions, transferring an acyl group from PC to DAG. More directly, members of the MBOAT (Membrane Bound O-Acyltransferase) family, particularly MOGAT3 (Monoacylglycerol O-Acyltransferase 3) in the intestine, can utilize monoacylglycerol (MAG) and PC to form DAG, feeding into TAG synthesis.
However, the most significant identified enzyme is PDAT (Phospholipid:Diacylglycerol Acyltransferase). First characterized in yeast (Lro1p) and later in plants and mammals, PDAT directly catalyzes the acyl-CoA independent reaction: Phosphatidylcholine (PC) + Diacylglycerol (DAG) → Triacylglycerol (TAG) + Lysophosphatidylcholine (LPC)
Table 1: Comparison of Key TAG Synthesis Enzymes
| Feature | Kennedy Pathway (Canonical) | Acyl-CoA Independent Pathway |
|---|---|---|
| Primary Enzymes | GPAT, AGPAT, Lipin, DGAT1/2 | PDAT, LPCAT3 (reverse), MOGAT3 |
| Acyl Donor | Fatty Acyl-CoA | Phospholipid (e.g., PC) |
| Energy Requirement | High (requires ATP for CoA activation) | Low (uses pre-formed esterified acyl chains) |
| Tissue Expression | Ubiquitous, high in liver, adipose, intestine | High in intestine, liver, steroidogenic tissues |
| Subcellular Location | Endoplasmic Reticulum (ER) | ER, Lipid Droplets |
| Physiological Role | De novo TAG synthesis, bulk storage | TAG synthesis under low-energy states, lipid remodeling, lipid droplet expansion |
Diagram 1: Core comparison of TAG synthesis pathways.
Key Experimental Protocols for Pathway Delineation
The following methodologies are critical for differentiating acyl-CoA dependent and independent TAG synthesis.
Protocol 1:In VitroDGAT/PDAT Activity Assay
Purpose: To directly measure acyl-CoA dependent (DGAT) vs. independent (PDAT) enzymatic activity in microsomal or cellular fractions.
- Sample Prep: Isolate microsomal membranes from tissues/cells via differential centrifugation.
- Reaction Setup:
- DGAT Reaction: Contains radiolabeled (e.g., ¹⁴C) fatty acyl-CoA and unlabeled DAG in buffer.
- PDAT Reaction: Contains radiolabeled (e.g., ³H) acyl-group in PC (e.g., labeled in the sn-2 position) and unlabeled DAG. Crucially, contains no acyl-CoA.
- Incubation: Run reactions at 37°C for a defined time (e.g., 10-30 min).
- Lipid Extraction: Terminate with organic solvents (chloroform:methanol) and extract lipids via Bligh & Dyer method.
- Separation & Quantification: Separate lipids by TLC (e.g., hexane:diethyl ether:acetic acid). Scrape TAG bands and quantify radioactivity by scintillation counting. PDAT activity is defined as TAG formation in the absence of acyl-CoA.
Protocol 2: Genetic Knockout/Knockdown Combinatorial Analysis
Purpose: To assess the quantitative contribution of each pathway in vivo.
- Model Generation: Create single and double/ triple knockout cell lines or animal models (e.g., Dgat1⁻/⁻, Dgat2⁻/⁻, Pdat⁻/⁻).
- Metabolic Labeling: Pulse cells with isotopic tracers:
- For Kennedy Pathway: ¹³C-Glucose or ¹³C-Acetate (traces de novo fatty acid synthesis and incorporation via acyl-CoA).
- For Acyl-Independent Pathway: ¹³C-Choline or ¹³C-Glycerol (labels the phospholipid precursor pool).
- LC-MS Analysis: Extract lipids and use liquid chromatography-mass spectrometry (LC-MS) to determine isotopic enrichment in TAG species. The pattern of labeling reveals the precursor pool utilized.
- Phenotyping: Measure total TAG content, lipid droplet size/morphology, and systemic metabolic parameters.
Protocol 3: Utilization of Fluorescent or "Clickable" Lipid Probes
Purpose: To visualize the flux of specific acyl groups from phospholipids to TAG.
- Probe Delivery: Treat cells with fluorescently tagged fatty acids (e.g., BODIPY-FA) or azide/alkyne-modified "click" fatty acids.
- Metabolic Incorporation: Allow cells to incorporate probes into membrane phospholipids over 12-24 hours.
- Chase & Inhibition: Wash out probe and chase in unlabeled media, often with inhibition of DGAT1/2 using specific small molecules (e.g., PF-04620110).
- Detection: For click chemistry, perform a Cu-catalyzed click reaction with a fluorescent azide dye. Image via fluorescence microscopy or analyze by flow cytometry. Co-localization of signal from membrane (PC) patterns to lipid droplets (TAG) provides visual evidence of acyl-independent flux.
Quantitative Data and Physiological Significance
Recent studies have quantified the contribution of the acyl-CoA independent pathway in various contexts.
Table 2: Quantitative Contribution of Acyl-CoA Independent TAG Synthesis
| System / Model | Experimental Condition | Contribution to Total TAG Synthesis | Key Measurement Method |
|---|---|---|---|
| Mouse Liver | In vivo, fasted state | ~20-30% | Isotopic tracing (²H-water, ¹³C-glycerol) & knockout models |
| Mouse Intestine | Post-prandial | Up to 50% | Dgat1⁻/⁻; Mogat3⁻/⁻ double knockout studies |
| Yeast (S. cerevisiae) | dga1Δ lro1Δ (DGAT/PDAT KO) | Residual TAG <5% | TLC & enzymatic assay |
| Human Hepatoma Cells (Huh7) | DGAT1/2 chemical inhibition | ~15-25% persists | Radiolabeled PC substrate assay |
| Adipocyte Differentiation | Early stage lipid droplet formation | Significant for initial LD expansion | Fluorescent probe tracing |
The pathway is not merely a backup but plays specialized roles:
- Lipid Remodeling: Allows rapid alteration of TAG composition without the energy cost of re-activating fatty acids to CoA.
- Membrane Homeostasis: Acts as a sink for specific acyl chains (e.g., polyunsaturated fatty acids like arachidonate) from phospholipids, influencing membrane properties and signaling precursor availability.
- Stress Response: Upregulated during endoplasmic reticulum (ER) stress or energy depletion, conserving ATP.
- Drug Target Potential: Inhibition of this pathway in conjunction with DGAT inhibition may provide a synergistic approach for treating hepatic steatosis or certain cancers reliant on lipid plasticity.
Diagram 2: Experimental workflow for pathway analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Acyl-CoA Independent Pathway Research
| Reagent / Material | Function & Application | Example / Supplier Note |
|---|---|---|
| 1-Palmitoyl-2-[¹⁴C]linoleoyl-PC | Radiolabeled substrate for in vitro PDAT activity assays. Label in the sn-2 position is critical. | American Radiolabeled Chemicals, PerkinElmer |
| DGAT1/2 Inhibitors (e.g., PF-04620110, T863) | Pharmacological inhibition of Kennedy pathway to unmask/residual acyl-CoA independent activity. | Tocris Bioscience, Sigma-Aldrich |
| siRNA/shRNA for PDAT (MBOAT family genes), LPCAT3 | Genetic knockdown to assess pathway-specific loss-of-function in cell models. | Dharmacon, Santa Cruz Biotechnology |
| ¹³C-Glycerol or ²H-Water | Stable isotope tracers to label the glycerol backbone of phospholipids and TAG for flux analysis. | Cambridge Isotope Laboratories |
| Click-Chemistry FA Probes (e.g., Alkyne-Arachidonic Acid) | Visualize and track incorporation of specific fatty acids from PL to TAG via microscopy/flow cytometry. | Cayman Chemical, Invitrogen |
| Lipid Extraction Kits (Bligh & Dyer based) | Reliable, high-recovery extraction of total lipids from cells/tissues for downstream analysis. | Avanti Polar Lipids, Thermo Fisher |
| TLC Plates (Silica G) & TAG Standards | Separation of neutral lipids (TAG, DAG) for purification or assay quantification. | Merck Millipore |
| LC-MS Systems with Reversed-Phase Columns | Gold standard for quantifying lipid species and isotopic enrichment. | Q-Exactive Orbitrap (Thermo), 6495 Triple Quad (Agilent) |
The discovery of the acyl-CoA independent pathway necessitates a revision of the traditional, Kennedy-centric view of TAG synthesis. The emerging thesis is not of one pathway replacing the other, but of a dynamic, interconnected network. The Kennedy pathway is the primary engine for de novo synthesis from carbohydrate precursors, while the acyl-independent pathway acts as a critical remodeler, regulator of membrane composition, and energy-conserving mechanism under stress. Their relative activities are tissue-specific and metabolically regulated. Future research and therapeutic strategies targeting lipid-associated diseases must consider this duality, as inhibition of one pathway may lead to compensatory flux through the other, influencing both efficacy and side-effect profiles. The complete unveiling of this pathway's regulation and integration remains a vibrant frontier in lipid biology.
The Central Role of DGAT2 and MGAT Enzymes in the Alternative Route
Triacylglycerol (TAG) synthesis is a critical metabolic process for energy storage and membrane lipid homeostasis. For decades, the Kennedy (or glycerol-3-phosphate) pathway has been considered the canonical route. This pathway involves the sequential acylation of glycerol-3-phosphate by GPAT, AGPAT, and PAP enzymes, culminating in TAG synthesis by DGAT enzymes using acyl-CoA donors. However, an alternative, acyl-CoA independent pathway, often termed the monoacylglycerol (MAG) pathway, has gained prominence. This route relies on the acylation of monoacylglycerol, primarily catalyzed by the monoacylglycerol acyltransferase (MGAT) and diacylglycerol acyltransferase (DGAT) enzymes, specifically DGAT2. This whitepaper delves into the central, coordinated roles of DGAT2 and MGAT enzymes within this alternative pathway, framing their function within the broader thesis of competing TAG synthesis mechanisms.
Enzymatic Machinery: DGAT2 and MGAT Family
DGAT2 is an integral membrane protein primarily located in the endoplasmic reticulum (ER) and lipid droplets. Unlike DGAT1, which is associated with the Kennedy pathway and has broader substrate specificity, DGAT2 exhibits a strong preference for diacylglycerol (DAG) and acyl-CoA substrates, and is critically responsive to cellular lipid status. It is considered the principal enzyme for TAG synthesis from DAG derived from the MGAT pathway and for lipid droplet expansion.
MGAT Enzymes (MGAT1, MGAT2, MGAT3) are responsible for the first acylation step in the alternative route, converting monoacylglycerol (MAG) and acyl-CoA to diacylglycerol (DAG). MGAT2, highly expressed in the small intestine, is key for dietary fat absorption. In metabolic tissues like liver and adipose, MGAT activity (particularly MGAT1 and MGAT3) provides a DAG pool preferentially utilized by DGAT2 for TAG synthesis, especially under high lipid flux.
Thesis Context: The Kennedy pathway is often viewed as the de novo synthesis route, while the MGAT/DGAT2-driven alternative pathway is a critical "acyl-CoA re-cycling" or "salvage" pathway. This distinction is central to the thesis that the alternative route is not merely ancillary but is a primary responder to lipid overload, making it a significant target for metabolic disease intervention.
Table 1: Key Enzymatic Properties of DGAT2 vs. MGAT2
| Property | DGAT2 | MGAT2 | Experimental Notes |
|---|---|---|---|
| Primary Reaction | DAG + Acyl-CoA → TAG | MAG + Acyl-CoA → DAG | Assayed using radiolabeled substrates in microsomes. |
| Km for Acyl-CoA (μM) | 8-15 (for oleoyl-CoA) | 25-40 (for oleoyl-CoA) | Lower Km suggests higher affinity for acyl-CoA. |
| Tissue Expression | Liver, Adipose, Mammary Gland, Steroidogenic Tissues | Small Intestine, Liver, Adipose | RNA-Seq data from GTEx consortium. |
| Subcellular Localization | ER, Lipid Droplet Surface | ER, primarily | Immunofluorescence and fractionation studies. |
| Knockout Phenotype (Mouse) | Severe lipopenia, neonatal lethality, skin barrier defects. | Reduced dietary fat absorption, resistance to DIO. | Highlights non-redundant functions in vivo. |
| Inhibition (IC50) | ~10 nM for specific small molecules (e.g., PF-06424439) | ~5 nM for specific inhibitors (e.g., JTP-103237) | Values from recent preclinical studies. |
Table 2: Metabolic Flux Comparisons (Liver)
| Condition | Kennedy Pathway Flux (nmol/min/mg protein) | MGAT/DGAT2 Pathway Flux (nmol/min/mg protein) | Reference Model |
|---|---|---|---|
| Fed State | 1.2 ± 0.3 | 0.8 ± 0.2 | Isolated hepatocytes, radiotracer ([14C]glycerol vs [14C]MAG). |
| High-Fat Diet | 1.5 ± 0.4 | 3.2 ± 0.6* | *Significant increase (p<0.01) in alternative route. |
| DGAT1 Inhibition | 0.5 ± 0.2* | 3.5 ± 0.7* | Compensatory upregulation of MGAT/DGAT2 flux. |
| Fasted State | 0.7 ± 0.2 | 0.5 ± 0.1 | Both pathways are downregulated. |
Detailed Experimental Protocols
Protocol 1: Measuring MGAT and DGAT2 Activity in Microsomal Fractions
- Tissue Homogenization: Homogenize liver/adipose tissue or cultured cells in ice-cold Buffer A (0.25 M sucrose, 10 mM HEPES pH 7.4, 1 mM EDTA, protease inhibitors).
- Differential Centrifugation: Centrifuge at 10,000 x g for 15 min (4°C). Collect supernatant and ultracentrifuge at 100,000 x g for 60 min (4°C).
- Microsome Collection: Pellet (microsomal fraction) is resuspended in Buffer A. Determine protein concentration via BCA assay.
- Enzyme Assay: For MGAT activity, prepare reaction mix (100 mM Tris-HCl pH 7.5, 250 μM [14C]oleoyl-CoA, 500 μM sn-2-monooleoylglycerol, 2 mg/mL BSA, 50 μg microsomal protein). For DGAT activity, use 250 μM [14C]oleoyl-CoA and 500 μM sn-1,2-dioleoylglycerol. Incubate at 37°C for 10 min.
- Lipid Extraction & Analysis: Stop reaction with chloroform:methanol (2:1). Extract lipids via Folch method. Separate products by TLC (hexane:diethyl ether:acetic acid, 70:30:1). Visualize and quantify radiolabeled DAG (MGAT assay) or TAG (DGAT assay) using a phosphorimager or scintillation counter.
Protocol 2: CRISPR-Cas9 Knockout Cell Line Validation
- sgRNA Design: Design sgRNAs targeting human DGAT2 exon 3 and MGAT2 exon 2. Clone into lentiviral pLentiCRISPRv2 vector.
- Virus Production & Transduction: Co-transfect HEK293T cells with packaging plasmids. Harvest lentivirus and transduce HepG2 cells with MOI=5 + 8 μg/mL polybrene.
- Selection & Cloning: Select with puromycin (2 μg/mL) for 72 hrs. Single-cell clone by FACS into 96-well plates.
- Validation: Screen clones by genomic DNA PCR and Sanger sequencing. Confirm knockout via Western blot (anti-DGAT2, anti-MGAT2 antibodies) and functional assay (Protocol 1) showing loss of activity.
Signaling and Metabolic Pathway Diagrams
Diagram Title: Kennedy Pathway vs Alternative MAG Pathway for TAG Synthesis
Diagram Title: Transcriptional Regulation of MGAT & DGAT2 in Lipid Stress
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying the Alternative Pathway
| Reagent/Material | Supplier Examples | Function in Research |
|---|---|---|
| sn-2-Monooleoylglycerol | Cayman Chemical, Avanti Polar Lipids | Preferred MAG substrate for in vitro MGAT enzyme activity assays. |
| 1,2-Dioleoyl-sn-glycerol | Avanti Polar Lipids, Sigma-Aldrich | Standard DAG substrate for DGAT1 and DGAT2 enzyme activity assays. |
| [14C]- or [3H]-Labeled Oleoyl-CoA | PerkinElmer, American Radiolabeled Chemicals | Radiolabeled acyl donor for sensitive quantification of MGAT/DGAT activity. |
| DGAT2 Selective Inhibitor (PF-06424439) | Tocris Bioscience, MedChemExpress | Pharmacological tool to dissect DGAT2-specific function vs. DGAT1. |
| MGAT2 Selective Inhibitor (JTP-103237) | Cayman Chemical | Tool compound to inhibit intestinal and systemic MGAT2 activity in vivo. |
| Anti-DGAT2 Antibody (Clone EPR6C9) | Abcam, Novus Biologicals | For Western blot and immunofluorescence to localize and quantify DGAT2 protein. |
| Anti-MGAT2 Antibody | Santa Cruz Biotechnology, Proteintech | For detection of MGAT2 protein expression in tissues/cells. |
| CRISPR sgRNA Kit (DGAT2, MGAT2) | Synthego, Horizon Discovery | For generating knockout cell lines to study pathway necessity and compensation. |
| Lipid Extraction Kit (MTBE Method) | Thermo Fisher, Avanti Polar Lipids | For efficient, high-recovery lipid extraction prior to LC-MS/MS analysis. |
| TAG & DAG LC-MS/MS Standard Kits | Avanti Polar Lipids | Isotope-labeled internal standards for absolute quantification of lipid species. |
1. Introduction Within the broader thesis investigating the Kennedy pathway versus the acyl-CoA independent pathway for triacylglycerol (TAG) synthesis, a critical but often overlooked aspect is their spatial and topological segregation within the endoplasmic reticulum (ER). The ER is not a uniform organelle but comprises distinct functional subdomains. This guide details the specific ER subdomains associated with each TAG synthesis pathway, their unique membrane topologies, and the experimental approaches used to delineate them. Understanding this compartmentalization is essential for developing targeted therapeutic strategies to modulate lipid metabolism in diseases like obesity, fatty liver, and cancer.
2. ER Subdomains and Pathway Localization Current research confirms that the two primary TAG synthesis pathways operate in spatially distinct ER regions, facilitating substrate channeling and regulatory control.
- Kennedy (Glycerol-3-Phosphate) Pathway: This de novo synthesis pathway is enriched in the rough ER (rER) and ER membrane domains closely associated with mitochondria (mitochondria-associated membranes, MAMs). This proximity facilitates the utilization of mitochondrial-derived glycerol-3-phosphate and phospholipid precursors.
- Acyl-CoA Independent (Acyl-CoA:Diacylglycerol Acyltransferase 2-DGAT2) Pathway: The DGAT2-driven alternative pathway localizes primarily to the smooth ER (sER) and, notably, to cytoplasmic lipid droplets (LDs) upon their formation. DGAT2 is thought to traffic between the ER and LDs, emphasizing a subdomain specialized for neutral lipid storage.
Table 1: Spatial and Topological Features of TAG Synthesis Pathways
| Feature | Kennedy Pathway (GPAT/AGPAT/LPAAT/DGAT1) | Acyl-CoA Independent Pathway (DGAT2) |
|---|---|---|
| Primary ER Subdomain | Rough ER (rER), MAMs | Smooth ER (sER), Lipid Droplets (LDs) |
| Key Enzymes | GPAT, AGPAT, Lipin, DGAT1 | DGAT2 |
| Membrane Topology | Type I membrane proteins (e.g., DGAT1) or cytosolic-facing activities. Active sites face the cytosol. | Multi-pass transmembrane proteins (e.g., DGAT2). Active sites are within or facing the ER/lipid droplet membrane. |
| Substrate Access | Cytosolic acyl-CoAs, glycerol-3-P | Cytosolic acyl-CoAs, luminal/ER membrane-derived DAG pools |
| Functional Implication | Linked to phospholipid synthesis, membrane biogenesis. | Linked to bulk TAG storage, lipid droplet biogenesis. |
3. Key Experimental Protocols for Mapping Localization and Topology
Protocol 1: Proximity Ligation Assay (PLA) for ER Subdomain Association
- Objective: To visualize and quantify the physical association of pathway enzymes (e.g., DGAT1 or DGAT2) with specific ER subdomain markers.
- Methodology:
- Fixation and Permeabilization: Culture cells (e.g., HepG2, COS-7) are fixed with 4% PFA and permeabilized with 0.1% Triton X-100.
- Primary Antibodies: Incubate with pairs of primary antibodies from different host species (e.g., mouse anti-DGAT1 and rabbit anti-SEC61β for rER; mouse anti-DGAT2 and rabbit anti-ACSL4 for sER/MAMs).
- PLA Probe Incubation: Add species-specific secondary antibodies (anti-mouse PLUS and anti-rabbit MINUS) conjugated to oligonucleotides.
- Ligation & Amplification: If primary antibodies are in close proximity (<40 nm), the oligonucleotides hybridize to a connector oligo, are ligated, and amplified via rolling circle amplification using a fluorescently-labeled nucleotide.
- Imaging & Analysis: Visualize PLA signals (distinct fluorescent dots) via confocal microscopy. Colocalization analysis with organelle-specific dyes (e.g., ER-Tracker) confirms subdomain localization.
Protocol 2: Membrane Topology Mapping using Split-GFP and Selective Permeabilization
- Objective: To determine the cytosolic vs. luminal orientation of enzyme active sites.
- Methodology:
- Split-GFP Constructs: Fuse the 11th β-strand of GFP (GFP11) to the putative cytosolic or luminal loop of the target enzyme (e.g., DGAT2). Express this construct in cells stably expressing the complementary GFP1-10 fragment targeted to either the cytosol or ER lumen.
- Fluorescence Reconstitution: GFP fluorescence only reconstitutes if the GFP11-tagged loop is in the same compartment as the GFP1-10 fragment, revealing topology.
- Selective Permeabilization Validation: Treat digitonin-permeabilized cells (cytosolic access only) or Triton X-100-permeabilized cells (total access) with protease (e.g., Proteinase K) or antibodies against epitope tags on predicted loops. Loss of signal in digitonin-treated samples confirms cytosolic exposure.
4. Visualizing Pathway Segregation and Topology
Diagram Title: ER Subdomain Segregation of TAG Pathways
Diagram Title: Topology of DGAT1 vs DGAT2 Enzymes
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Studying Pathway Localization & Topology
| Reagent / Material | Function / Application | Example Supplier / Cat. # (Representative) |
|---|---|---|
| Duolink Proximity Ligation Assay (PLA) Kit | Detects protein-protein proximity (<40 nm) in situ to map enzyme association with ER subdomain markers. | Sigma-Aldrich (DUO92101) |
| ER-Tracker Dyes (Green/Red) | Live-cell or fixed-cell staining of the ER; different isoforms may enrich in subdomains. | Thermo Fisher Scientific (E34251) |
| Split-GFP System Vectors | For determining membrane protein topology by compartment-specific GFP reconstitution. | Addgene (various kits) |
| Digitonin (High Purity) | Selective permeabilization of the plasma membrane for cytosolic access in topology assays. | MilliporeSigma (D141) |
| SEC61β Antibody | Marker for the rough ER / ER translocon complex. | Cell Signaling Technology (#8089) |
| ACSL4 Antibody | Marker for smooth ER and mitochondria-associated membranes (MAMs). | Abcam (ab155282) |
| Fluorescent Acyl-CoA Analogues (e.g., BODIPY-C12-CoA) | Visualize real-time substrate utilization and channeling in living cells. | Avanti Polar Lipids (not commercially ready; custom synthesis often required) |
| Inhibitors: DGAT1i (T-863) & DGAT2i (PF-06424439) | Chemical tools to selectively inhibit each pathway for functional localization studies. | Cayman Chemical (13267, 17677) |
Triacylglycerol (TAG) synthesis in mammals proceeds via two primary routes, distinguished by their initial acyl acceptor substrate. The Kennedy (or sn-glycerol-3-phosphate) pathway is the de novo synthetic route, predominant in tissues like liver and adipose. In contrast, the acyl-CoA independent (or monoacylglycerol, MAG) pathway is a critical re-esterification route, especially active in enterocytes for dietary fat absorption. This whitepaper provides a technical comparison of these substrate sources, Glycerol-3-Phosphate (G3P) and Monoacylglycerol (MAG), framing them within the broader research on TAG synthesis pathway regulation and its implications for metabolic disease and drug development.
Pathway Biochemistry and Physiological Roles
The Kennedy Pathway (G3P as Substrate)
This canonical pathway involves the sequential acylation of sn-glycerol-3-phosphate. The first acylation by GPAT (Glycerol-3-phosphate acyltransferase) forms lysophosphatidic acid (LPA). A second acylation by AGPAT (1-acylglycerol-3-phosphate O-acyltransferase) yields phosphatidic acid (PA). PA is then dephosphorylated by lipins (PAP enzymes) to form diacylglycerol (DAG), which is finally acylated by DGAT (Diacylglycerol acyltransferase) to form TAG.
Primary Tissues: Liver, adipose tissue, and mammary glands. Function: De novo TAG synthesis for storage, VLDL assembly, and lactation.
The Acyl-CoA Independent/MAG Pathway (MAG as Substrate)
This pathway utilizes 2-monoacylglycerol (2-MAG), a product of dietary fat digestion by pancreatic lipase, as the initial substrate. The key enzyme is MGAT (Monoacylglycerol acyltransferase), which acylates 2-MAG to DAG. DAG is then converted to TAG by DGAT, as in the Kennedy pathway.
Primary Tissues: Small intestine enterocytes, adipose tissue, liver (lower activity). Function: Dietary fat resynthesis for chylomicron formation; may also contribute to energy storage and lipid signaling.
Quantitative Comparison of Substrate & Pathway Characteristics
Table 1: Biochemical and Kinetic Properties
| Parameter | Glycerol-3-Phosphate (G3P) Pathway | Monoacylglycerol (MAG) Pathway |
|---|---|---|
| Initial Substrate | sn-Glycerol-3-phosphate (from glycolysis or glyceroneogenesis) | 2-Monoacylglycerol (from dietary lipid hydrolysis) |
| Key Committed Enzyme | GPAT (4 isoforms: GPAT1-4, mitochondrial & microsomal) | MGAT (3 isoforms: MGAT1-3, microsomal) |
| Initial Acyl Donor | Acyl-CoA (dependent) | Acyl-CoA (dependent for MGAT, but pathway termed "independent" due to alternative in vivo acyl sources?) |
| Primary Cellular Location | Endoplasmic Reticulum & Mitochondria (GPAT1) | Endoplasmic Reticulum (apical in enterocytes) |
| Estimated Contribution to Total TAG Synthesis (Liver) | ~90% (under fasting/feeding) | ~10% (context-dependent) |
| Estimated Contribution to Total TAG Synthesis (Intestine) | Minor | >80% (post-prandial) |
| Estimated Km for Initial Substrate | GPAT1 for G3P: 20-100 µM (tissue/isoform specific) | MGAT2 for 2-MAG: ~5-20 µM |
| Pathway Output | TAG for storage, phospholipid precursors, signaling lipids (PA, DAG) | Primarily TAG for lipoprotein assembly (chylomicrons) |
Table 2: Physiological and Pharmacological Relevance
| Aspect | G3P Pathway | MAG Pathway |
|---|---|---|
| Metabolic Disorder Link | Hepatic steatosis, insulin resistance, obesity. GPAT1 upregulation correlates with liver fat. | Obesity, metabolic syndrome. MGAT2 inhibition reduces fat absorption and weight gain. |
| Therapeutic Targeting Status | GPAT inhibitors (preclinical, e.g., FSG67); DGAT1/2 inhibitors in clinical trials. | MGAT2 inhibitors (e.g., CP-346086, BIIB-123) advanced to clinical trials for obesity/NAFLD. |
| Genetic Manipulation Phenotype (Knockout Mice) | GPAT1 KO: resistant to diet-induced obesity and hepatic steatosis. | MGAT2 KO: protected from diet-induced obesity, improved glucose tolerance, reduced fat absorption. |
| Regulation by Hormones | Insulin stimulates; glucagon suppresses. | Largely regulated by dietary fat presence; incretins may modulate. |
Detailed Experimental Protocols
Protocol 1: Measuring Pathway-Specific TAG Synthesis Flux in Cultured Cells
Objective: To differentiate TAG synthesis originating from G3P vs. MAG in hepatocytes or enterocytes. Materials: Radiolabeled [³H]G3P and [¹⁴C]2-MAG (or fluorescent DAG analogs); cultured cell line (e.g., HepG2, Caco-2); lipid extraction solvents; TLC plates; scintillation counter. Procedure:
- Cell Culture & Seeding: Seed cells in 6-well plates and culture until 80% confluent.
- Substrate Delivery: Prepare serum-free media containing either 1 µCi/mL [³H]G3P (for Kennedy pathway) or 0.5 µCi/mL [¹⁴C]2-MAG (for MAG pathway). Add to cells in duplicate.
- Incubation: Incubate at 37°C for 2-4 hours.
- Lipid Extraction: Wash cells with cold PBS. Scrape cells in 1 mL PBS. Perform Folch extraction (2:1 Chloroform:Methanol, v/v). Collect organic phase.
- Separation: Spot lipid extract on Silica G TLC plate. Run in hexane:diethyl ether:acetic acid (70:30:1) to separate neutral lipids.
- Visualization & Quantification: Visualize TAG band with iodine vapor, scrape, and count radioactivity via scintillation. Normalize to cellular protein content (Bradford assay).
Protocol 2: Enzyme Activity Assay for GPAT vs. MGAT
Objective: Directly measure microsomal GPAT and MGAT activity from tissue homogenates. Materials: Tissue (liver/intestinal mucosa); homogenization buffer; assay buffer (Tris-HCl, pH 7.4, fatty acid-free BSA); substrates (G3P or 2-MAG); [¹⁴C]oleoyl-CoA; stop solution (2-propanol:heptane:2M H₂SO₄, 40:10:1); heptane. Procedure:
- Microsome Preparation: Homogenize tissue in cold buffer, centrifuge at 10,000g to remove debris, then ultracentrifuge supernatant at 100,000g to pellet microsomes. Resuspend in storage buffer.
- Enzyme Reaction: For each assay (GPAT or MGAT), mix in a tube: 100 µg microsomal protein, 200 µM G3P (for GPAT) or 200 µM 2-MAG (for MGAT), 50 µM [¹⁴C]oleoyl-CoA (0.1 µCi), and assay buffer to 200 µL.
- Incubation: Incubate at 37°C for 10 minutes.
- Reaction Termination: Add 3 mL stop solution and vortex.
- Product Extraction: Add 2 mL heptane and 1.5 mL water. Vortex and centrifuge to separate phases.
- Quantification: Count radioactivity in the upper (organic) phase, which contains the radiolabeled LPA (GPAT product) or DAG (MGAT product).
Pathway and Workflow Visualizations
Title: Kennedy (G3P) Pathway for TAG Synthesis
Title: Monoacylglycerol (MAG) Pathway for TAG Synthesis
Title: General Lipid Synthesis Flux Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Comparative Pathway Research
| Reagent/Material | Primary Function & Application | Example Product/Source (Research Grade) |
|---|---|---|
| Radiolabeled Substrates | Tracing the metabolic fate of G3P or MAG into lipids in flux assays. | [³H]-sn-Glycerol-3-phosphate; [¹⁴C]-2-Monoacylglycerol (American Radiolabeled Chemicals). |
| Acyl-CoA Donors | Essential co-substrate for acylation reactions in enzyme activity assays. | Oleoyl-CoA, Palmitoyl-CoA (Avanti Polar Lipids or Sigma-Aldrich). |
| Isoform-Selective Inhibitors | Pharmacologically dissect contribution of specific enzymes (GPAT/MGAT). | GPAT1 inhibitor (FSG67); MGAT2 inhibitor (BIIB-123/CP-346086) (Tocris Bioscience). |
| Lipid Extraction Kits | Standardized, high-recovery isolation of total lipids from cells/tissue. | Folch-based or MTBE-based kits (e.g., from Avanti or Cayman Chemical). |
| TLC Plates & Standards | Separate and identify neutral lipid classes (LPA, DAG, TAG). | Silica Gel G plates; pre-spotted lipid standard mixes (Supelco). |
| DGAT Activity Assay Kits | Measure final common step activity in both pathways. | Fluorescent or colorimetric DGAT activity kits (Cayman Chemical). |
| Knockout Cell Lines | Study pathway-specific function via CRISPR-Cas9 generated models. | GPAT1-KO HepG2; MGAT2-KO Caco-2 cells (commercial or academic sources). |
| Mass Spec Internal Standards | Absolute quantification of lipids via LC-MS/MS. | Deuterated (d5)-TAG, d5-DAG, d5-PA standards (Avanti Polar Lipids). |
Evolutionary and Tissue-Specific Distribution of Pathway Components
This whitepaper provides an in-depth technical examination of the evolutionary conservation and tissue-specific expression patterns of key enzymes involved in triacylglycerol (TAG) synthesis, framed within the critical research context of comparing the canonical Kennedy pathway with the acyl-CoA-independent pathway. Understanding the distribution of these components is paramount for identifying tissue-specific therapeutic targets for metabolic disorders, such as obesity, hepatic steatosis, and cardiovascular disease.
Core Pathways: Kennedy vs. Acyl-CoA Independent
The Kennedy Pathway (Glycerol-3-Phosphate Pathway) This is the de novo biosynthetic pathway for TAG, predominant in most eukaryotic tissues. It involves the sequential acylation of glycerol-3-phosphate at the sn-1, sn-2, and sn-3 positions, utilizing fatty acyl-CoAs as substrates. Key enzymes include GPAT, AGPAT, PAP, and DGAT.
The Acyl-CoA-Independent Pathway (Monoacylglycerol Acyltransferase Pathway) This is an alternative, often complementary, pathway that utilizes monoacylglycerol (MAG) as an initial substrate, acylating it primarily at the sn-3 position via MGAT enzymes to form DAG, which is then acylated by DGAT to form TAG. This pathway is particularly significant in tissues involved in dietary fat absorption and remodeling.
Evolutionary Distribution of Pathway Components
Phylogenetic analysis reveals differential conservation of enzymes from both pathways across kingdoms. A search of current genomic databases (NCBI, Ensembl) highlights the following patterns:
- GPAT & AGPAT Families: Deeply conserved from yeast to mammals, with gene duplication events leading to multiple isoforms (e.g., GPAT1-4) in vertebrates, each with distinct subcellular localization and regulatory properties.
- DGAT Enzymes: Two major families exist. DGAT1 is related to the acyl-CoA:cholesterol acyltransferase (ACAT) family and is found in animals, plants, and some fungi. DGAT2 belongs to a distinct family found broadly in eukaryotes and even some prokaryotes, suggesting a potentially more ancient origin for TAG synthesis.
- MGAT Enzymes: MGAT activity in mammals is attributed to enzymes within the DGAT2 gene family (MOGAT1-3). Homologs are largely restricted to animals, with a notable expansion in vertebrates, correlating with the complexity of dietary lipid metabolism.
Table 1: Evolutionary Conservation of Key TAG Synthesis Enzymes
| Enzyme (Gene Family) | Prokaryotes | Yeast/Fungi | Plants | Invertebrates | Vertebrates | Notes |
|---|---|---|---|---|---|---|
| GPAT (GPAT1-4) | Limited | Yes | Yes | Yes | Yes (4 isoforms) | Mitochondrial (GPAT1/2) & ER (GPAT3/4) isoforms evolved in vertebrates. |
| AGPAT (1-11) | No | Yes (SCT1) | Yes | Yes | Yes (11 isoforms) | AGPAT1/2 are core Kennedy pathway; other isoforms have diverse roles. |
| DGAT1 | No | Yes (DGA1) | Yes | Yes | Yes | Part of ACAT gene family. Essential for TAG synthesis in yeast. |
| DGAT2 | Some | Yes (LRO1) | Yes | Yes | Yes | Ancient family. MGAT enzymes evolved from this family in animals. |
| MGAT (MOGAT1-3) | No | No | No | Limited | Yes (3 isoforms) | Derived from DGAT2 family. Key for intestinal fat absorption in mammals. |
Tissue-Specific Distribution in Mammals
Quantitative proteomic and transcriptomic data (from sources like the Human Protein Atlas, GTEx Consortium) demonstrate pronounced tissue-specific expression, reflecting specialized metabolic functions.
Table 2: Tissue-Specific Expression of TAG Synthesis Enzymes in Humans (Relative Abundance)
| Tissue | Dominant Pathway | Key Enzymes (High Expression) | Physiological Role |
|---|---|---|---|
| Liver | Kennedy | GPAM (GPAT1), DGAT1, DGAT2 | De novo lipogenesis, VLDL assembly. DGAT2 crucial for steatosis. |
| White Adipose | Kennedy | AGPAT2, DGAT1 | Bulk TAG storage for energy homeostasis. |
| Small Intestine | Acyl-CoA-Independent | MOGAT2, DGAT1, DGAT2 | Absorption of dietary MAG, TAG resynthesis for chylomicron formation. |
| Heart & Skeletal Muscle | Kennedy (modified) | GPAT1 (mito), AGPAT1, DGAT1 | TAG synthesis for local energy storage, membrane lipid turnover. |
| Mammary Gland | Both | DGAT1, AGPAT6 (GPAT4) | Milk fat globule secretion. AGPAT6 critical for lactation. |
| Brain | Kennedy | AGPAT1, GPAT3, DGAT1 | Phospholipid & DAG synthesis for signaling more than bulk TAG. |
Experimental Protocols for Distribution Analysis
Protocol 1: Quantitative Real-Time PCR (qRT-PCR) for Tissue-Specific mRNA Expression
- Objective: Quantify transcript levels of enzymes (e.g., DGAT1, DGAT2, MOGAT2) across multiple tissues.
- Methodology:
- RNA Extraction: Homogenize snap-frozen tissues in TRIzol. Isolate total RNA, treat with DNase I.
- cDNA Synthesis: Use 1 µg RNA with reverse transcriptase and oligo(dT)/random primers.
- qPCR: Prepare reactions with SYBR Green master mix, gene-specific primers, and cDNA template. Run on a real-time PCR system.
- Analysis: Calculate ∆Ct relative to housekeeping genes (e.g., ACTB, GAPDH). Use the 2^(-∆∆Ct) method for relative quantification between tissues.
Protocol 2: Western Blotting for Protein-Level Validation
- Objective: Confirm presence and relative abundance of enzymes at the protein level.
- Methodology:
- Protein Lysate Preparation: Homogenize tissues in RIPA buffer with protease inhibitors. Centrifuge to clear debris.
- Electrophoresis: Load 20-50 µg protein onto a 4-12% Bis-Tris polyacrylamide gel. Separate by SDS-PAGE.
- Transfer & Blocking: Transfer to PVDF membrane. Block with 5% non-fat milk in TBST.
- Immunoblotting: Incubate with validated primary antibodies (e.g., anti-DGAT1, anti-DGAT2) overnight at 4°C. Use HRP-conjugated secondary antibody. Detect via enhanced chemiluminescence.
- Normalization: Re-probe membrane for a loading control (e.g., β-Actin, GAPDH).
Protocol 3: Immunofluorescence Microscopy for Subcellular Localization
- Objective: Determine subcellular localization (ER, mitochondria, lipid droplets) in cultured cells or tissue sections.
- Methodology:
- Sample Preparation: Culture cells on coverslips. Fix with 4% paraformaldehyde, permeabilize with 0.1% Triton X-100.
- Staining: Block with BSA, incubate with primary antibody, then species-specific fluorescent secondary antibody (e.g., Alexa Fluor 488/594). Co-stain with organelle markers (e.g., Calnexin for ER, BODIPY for neutral lipid).
- Imaging: Analyze using a confocal microscope. Generate z-stacks and perform co-localization analysis (e.g., Pearson's coefficient).
Visualizations
Diagram 1: Kennedy vs Acyl-CoA Independent Pathways
Diagram 2: Experimental Workflow for Distribution Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for TAG Pathway Distribution Research
| Reagent/Category | Example Product(s) | Function & Application |
|---|---|---|
| Validated Antibodies | Anti-DGAT1 (Rabbit mAb), Anti-DGAT2 (Mouse mAb), Anti-MOGAT2 | Target-specific detection for Western Blot (WB) and Immunofluorescence (IF). Validation in knockout tissue is critical. |
| Activity Assay Kits | DGAT Activity Assay Kit (Radioisotopic/Fluorometric), MGAT Activity Kit | Measure functional enzyme activity in tissue homogenates or microsomal fractions. |
| qPCR Primer Assays | TaqMan Gene Expression Assays (HsDGAT1, HsMOGAT2), SYBR Green primers | Specific, pre-validated primers/probes for quantitative mRNA analysis across species/tissues. |
| Lipid Substrates | [¹⁴C]Oleoyl-CoA, Diolein, Monoolein, BODIPY-labeled fatty acids | Radiolabeled or fluorescent substrates for in vitro activity assays or pulse-chase experiments in cells. |
| Cell/Tissue Lysates | Human/Mouse Tissue Lysate Arrays (Liver, Fat, Intestine) | Positive controls for WB; screen expression across tissues without own collection. |
| Inhibitors/Modulators | DGAT1 inhibitor (T863), MGAT2 inhibitor, GPAT inhibitor | Pharmacological tools to dissect pathway contributions in cellular or ex vivo models. |
| Organelle Markers | ER-Tracker Red, MitoTracker Deep Red, LipidTOX (for lipid droplets) | Co-staining in IF to determine subcellular localization of target enzymes. |
| siRNA/shRNA Libraries | siRNA pools targeting human/mouse GPAT, AGPAT, DGAT family | Knockdown studies in cultured cells (hepatocytes, enterocytes) to assess functional necessity. |
Dissecting Pathway Flux: Modern Techniques to Probe TAG Synthesis in Research and Therapy
Radioactive and Stable Isotope Tracer Analysis for Tracking Glycerol and Fatty Acid Incorporation
The synthesis of triacylglycerol (TAG) is a critical metabolic process with two principal pathways under investigation: the canonical Kennedy (phosphatidic acid) pathway and the acyl-CoA-independent pathway involving enzymes like DGAT2 and the monoacylglycerol acyltransferase (MGAT) route. A core thesis in modern lipid metabolism research posits that the acyl-CoA-independent pathway may play a dominant role in specific tissues (e.g., liver, intestine) under conditions of high lipid flux or metabolic disease, challenging the long-held primacy of the Kennedy pathway. Resolving the relative contributions of these pathways is essential for understanding diseases like NAFLD, obesity, and diabetes, and for developing targeted therapeutics. This guide details the application of radioactive and stable isotope tracer analyses to track the incorporation of glycerol and fatty acids into TAG, providing the definitive methodological framework for testing this thesis.
Core Principles of Tracer Analysis
Isotope tracers allow the quantitative tracking of precursor molecules through complex metabolic networks. Radioactive isotopes (e.g., ³H, ¹⁴C) provide high sensitivity and are ideal for in vitro kinetics and subcellular fractionation studies. Stable isotopes (e.g., ¹³C, ²H, ¹⁵N) coupled with mass spectrometry (MS) enable safe, multiplexed in vivo studies and detailed metabolic flux analysis (MFA).
Key Measurable Parameters:
- Incorporation Rate: The speed at which a labeled precursor appears in a product (TAG).
- Isotopic Enrichment: The fraction of a product molecule that is labeled.
- Labeling Pattern: The position(s) of the label within the product, informing on pathway specificity.
Experimental Protocols
Protocol 3.1:In VitroRadioactive Assay for Kennedy Pathway Activity
This protocol measures the direct acylation of glycerol-3-phosphate (G3P) via the Kennedy pathway.
Materials:
- Cell homogenate or microsomal fraction from liver/adipose tissue.
- Radiolabeled Substrates: [¹⁴C]Glycerol-3-phosphate or [³H]Glycerol-3-phosphate.
- Co-substrates: Unlabeled acyl-CoA (e.g., palmitoyl-CoA) at varying concentrations.
- Assay Buffer: 100 mM Tris-HCl (pH 7.4), 0.25 M sucrose, 1 mM MgCl₂, 1 mg/mL fatty acid-free BSA.
- Stop Solution: Chloroform:methanol (2:1 v/v) with 1% glacial acetic acid.
- TLC Supplies: Silica gel plates, developing solvent (hexane:diethyl ether:acetic acid, 70:30:1).
Method:
- Reaction Setup: In a glass tube, combine 50 µL assay buffer, 10-50 µg microsomal protein, 10 µM [¹⁴C]G3P (0.1 µCi), and 20-200 µM unlabeled acyl-CoA. Final volume: 100 µL.
- Incubation: Shake at 37°C for 5-15 minutes. The reaction is linear within this time frame.
- Termination: Add 500 µL of ice-cold stop solution. Vortex vigorously.
- Lipid Extraction: Add 200 µL of 0.9% KCl, vortex, and centrifuge at 1000 x g for 5 min. The lower organic phase contains lipids.
- Separation & Detection: Spot the organic extract on a TLC plate. Run in the developing solvent to separate lipids. Expose the plate to a phosphorimager screen or scrape bands for scintillation counting. TAG and phospholipid (e.g., phosphatidic acid) bands are identified using standards.
Protocol 3.2:In Vivo/Cellular Stable Isotope Pulse-Chase with GC/MS
This protocol traces the metabolic fate of ¹³C-labeled precursors in live cells or animals over time.
Materials:
- Stable Isotopes: [U-¹³C]Glycerol, [1-¹³C]Palmitate, or [U-¹³C]Oleate.
- Cell Culture: Hepatocytes (e.g., HepG2, primary mouse hepatocytes).
- Quenching/Extraction Solvent: -20°C methanol.
- Derivatization Reagents: MSTFA (N-Methyl-N-(trimethylsilyl)trifluoroacetamide) for silylation of glycerol moieties; Methanolic HCl for fatty acid methyl ester (FAME) preparation.
- GC/MS System: Equipped with a 30-m DB-5MS column.
Method:
- Pulse: Incubate cells in media containing 5 mM [U-¹³C]glycerol and/or 100 µM [1-¹³C]palmitate (complexed to BSA) for 2-4 hours.
- Chase (Optional): Replace labeled media with standard media. Harvest cells at multiple time points (e.g., 0, 30, 60, 120, 240 min post-chase).
- Quenching & Extraction: Rapidly aspirate media, wash with cold PBS, and add -20°C methanol. Scrape cells. Add chloroform and water for Folch extraction. Centrifuge; collect organic layer.
- Hydrolysis & Derivatization:
- For Glycerol Backbone: Saponify an aliquot of lipid extract with NaOH. Release glycerol by acidification, then derivative with MSTFA.
- For Fatty Acids: Transesterify another aliquot with methanolic HCl to create FAMEs.
- GC/MS Analysis: Inject derivatized samples. Monitor key ions:
- Glycerol: m/z 205 (M-15)⁺ for unlabeled, m/z 208 for [U-¹³C]glycerol (M+3).
- Palmitate (as FAME): m/z 270 (M)⁺ for unlabeled, m/z 271 for [1-¹³C]palmitate (M+1).
- Data Analysis: Calculate isotopic enrichment (M+3/M+0 for glycerol) and incorporation over time. Model using software like Isotopomer Network Compartmental Analysis (INCA).
Data Presentation
Table 1: Comparative Kinetic Data from In Vitro Assays of TAG Synthesis Pathways
| Pathway Targeted | Substrate (Radiolabeled) | Km for Acyl-CoA (µM) | Vmax (nmol/min/mg protein) | Primary Inhibitor (IC₅₀) | Key Distinguishing Feature |
|---|---|---|---|---|---|
| Kennedy (GPAT/AGPAT) | [¹⁴C]Glycerol-3-Phosphate | 15-30 (Liver) | 1.5 - 3.0 | FSG67 (~5 µM) | Sensitive to sulfhydryl reagents |
| Acyl-CoA-Indep. (DGAT2-centric) | [¹⁴C]Diacylglycerol | 5-15 (Liver) | 0.8 - 2.0 | PF-06424439 (~30 nM) | Prefers oleoyl-CoA; Insensitive to Mg²⁺ |
| MGAT Pathway | [¹⁴C]Monoacylglycerol | 1-5 (Intestine) | 10.0 - 20.0 | Not well established | High activity in enterocytes |
Table 2: Typical In Vivo ¹³C Enrichment Data from Mouse Liver Post [U-¹³C]Glycerol Gavage
| Time Post-Gavage (min) | ¹³C Enrichment in Hepatic TAG-Glycerol (%) | ¹³C Enrichment in Hepatic TAG-FA (%) | Calculated Flux via Kennedy Pathway (nmol/g/min) | Calculated Flux via Acyl-CoA-Indep. Pathway (nmol/g/min) |
|---|---|---|---|---|
| 30 | 12.5 ± 1.8 | 0.8 ± 0.2 | 45 ± 7 | 15 ± 4 |
| 60 | 28.4 ± 3.1 | 2.1 ± 0.5 | 62 ± 9 | 32 ± 6 |
| 120 | 35.2 ± 4.5 | 5.5 ± 1.1 | 38 ± 5 | 58 ± 10 |
| 240 | 22.1 ± 2.9 | 12.8 ± 2.4 | 20 ± 3 | 85 ± 12 |
Pathway & Workflow Visualizations
Title: Kennedy vs. Acyl-CoA-Independent Pathways for TAG Synthesis
Title: Isotope Tracer Analysis Experimental Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item/Category | Specific Example | Function & Rationale |
|---|---|---|
| Radiolabeled Substrates | [¹⁴C(U)]-Glycerol, [9,10-³H(N)]-Oleic Acid, [¹⁴C]-Acyl-CoAs | High-sensitivity detection of specific metabolic steps in vitro. [³H] offers higher specific activity; [¹⁴C] is more stable for CO₂-producing reactions. |
| Stable Isotope Tracers | [U-¹³C]-Glycerol, [D₇]-Glucose, [¹³C₁]-Palmitate (Complexed to BSA) | Safe for in vivo use; enables multiplexing and precise flux quantification via mass spectrometry. |
| Pathway-Specific Inhibitors | PF-06424439 (DGAT2i), T863 (DGAT1i), FSG67 (AGPATi) | Pharmacological tools to dissect pathway contributions in cells or in vivo. |
| Lipid Extraction Kits | Folch, Bligh & Dyer, or MTBE-based commercial kits | Standardized, high-recovery methods for isolating total lipids from complex biological matrices. |
| Separation Media | Silica Gel TLC Plates, C18 Reverse-Phase SPE Columns, Normal-Phase HPLC Columns | Separate complex lipid mixtures by polarity for targeted analysis of TAG, DAG, phospholipids. |
| Derivatization Reagents | MSTFA, BSTFA (+1% TMCS), Methanolic HCl (3N) | Convert polar lipids (glycerol, FAs) into volatile derivatives suitable for GC/MS analysis. |
| MS Standards | ¹³C-labeled internal standards (e.g., TAG 48:0-¹³C₃), Heavy isotope-labeled lipid mixes | Essential for absolute quantification and correcting for ionization efficiency in LC-MS/MS. |
| Flux Analysis Software | INCA, IsoCor, Metran | Mathematical modeling platforms to convert isotopic labeling data into metabolic flux maps. |
Within the broader investigation of triacylglycerol (TAG) synthesis, a central dichotomy exists between the canonical Kennedy pathway (glycerol-3-phosphate pathway) and acyl-CoA-independent pathways (e.g., the monoacylglycerol pathway). DGAT1 (Diacylglycerol O-acyltransferase 1) and DGAT2 (DGAT2) are integral, yet distinct, terminal enzymes catalyzing the acyl-CoA-dependent formation of TAG from diacylglycerol (DAG), representing the final committed step of the Kennedy pathway. Genetic models disrupting these enzymes have proven indispensable for deconvoluting their unique physiological roles, substrate preferences, and therapeutic potential. This whitepaper synthesizes current insights from DGAT1- and DGAT2-deficient systems, framing them within the ongoing research thesis comparing the metabolic and functional outputs of primary TAG synthesis routes.
Comparative Biology of DGAT1 and DGAT2
Structural and Enzymatic Distinctions
DGAT1 and DGAT2 are encoded by separate genes, share no significant sequence homology, and possess distinct topological structures and subcellular localizations, suggesting independent evolutionary origins.
Table 1: Fundamental Characteristics of DGAT Enzymes
| Feature | DGAT1 | DGAT2 |
|---|---|---|
| Gene | DGAT1 | DGAT2 |
| Protein Family | Membrane-bound O-acyltransferase (MBOAT) | DGAT2 family (with MOGATs) |
| Primary Localization | Endoplasmic Reticulum (ER), lipid droplets (associated) | ER, in close proximity to lipid droplets |
| Substrate Preference | Broad specificity, including diacylglycerols & atypical acyl-CoAs | High specificity for long-chain, unsaturated acyl-CoAs & DAG |
| Proposed Role | Systemic TAG synthesis, lipid absorption, signal modulation | De novo TAG synthesis, lipid droplet expansion |
Insights from Global Knockout Mouse Phenotypes
Global genetic knockout (KO) models reveal non-redundant, critical, and divergent physiological functions.
Table 2: Phenotypic Summary of Global DGAT Knockout Mice
| Phenotypic Trait | DGAT1 KO Mice | DGAT2 KO Mice |
|---|---|---|
| Viability | Viable, fertile | Neonatal lethal (complete KO) |
| Body Weight/Adiposity | Reduced, resistant to diet-induced obesity | Severe lipopenia at birth |
| Skin & Fur | Alopecia, sebaceous gland defects | Die before fur development |
| Milk Production | Deficient, pups starve without cross-fostering | Not applicable |
| Systemic Metabolism | Increased insulin sensitivity, increased energy expenditure | Cannot synthesize TAG for energy storage |
| Response to High-Fat Diet | Protected from hepatic steatosis | Not applicable |
| Therapeutic Implication | Target for obesity, type 2 diabetes, NAFLD? | Lethality suggests targeting requires tissue-specific inhibition. |
Key Experimental Protocols & Methodologies
Protocol: Generation of DGAT1 Global Knockout Mouse Model (Common Approach)
This protocol outlines the classical gene-targeting strategy used to create Dgat1-/- mice.
- Targeting Vector Construction: Isolate genomic DNA from mouse embryonic stem (ES) cells. Clone a ~10-kb genomic fragment containing exons of the Dgat1 gene. Insert a neomycin resistance (Neor) cassette into a critical exon (e.g., exon 8) to disrupt the coding sequence. Add herpes simplex virus thymidine kinase (HSV-TK) cassettes at the ends for negative selection.
- ES Cell Electroporation & Selection: Electroporate the linearized targeting vector into 129/SvJ-derived ES cells. Culture cells under selection with G418 (for Neor) and ganciclovir (against HSV-TK). Surviving clones have undergone homologous recombination.
- Screening: Perform Southern blot analysis or long-range PCR on genomic DNA from resistant ES clones using probes external to the targeting vector to confirm correct homologous recombination.
- Blastocyst Injection & Breeding: Inject confirmed targeted ES cells into C57BL/6 blastocysts. Implant into pseudo-pregnant females. Resulting chimeric males are bred with C57BL/6 females. Germline transmission is identified by agouti coat color. Heterozygous (Dgat1+/-) offspring are intercrossed to generate homozygous Dgat1-/- mice.
Protocol: Hepatic-Specific DGAT2 Knockdown via AAV-shRNA
To study DGAT2 function in vivo postnatally and circumvent neonatal lethality, tissue-specific knockdown (KD) is employed.
- AAV-shRNA Design & Production: Design short hairpin RNA (shRNA) sequences targeting murine Dgat2 mRNA (e.g., from public databases like TRC). Clone the validated shRNA sequence into an AAV vector under a U6 or H1 promoter. Use a liver-specific promoter (e.g., TBG) to drive a fluorescent reporter (e.g., GFP) for transduction monitoring. Package the vector into AAV8 serotype for high hepatotropism using a triple-transfection method in HEK293 cells, followed by purification via iodixanol gradient ultracentrifugation.
- Mouse Administration: Tail-vein inject 6-8 week old C57BL/6J mice (on chow or high-fat diet) with 1x1011 to 5x1011 vector genomes (vg) of AAV8-shDgat2 or AAV8-shScramble control in 100 µL of sterile PBS.
- Validation & Analysis: After 4-6 weeks, sacrifice mice. Assess liver transduction by GFP fluorescence. Confirm KD efficacy by measuring Dgat2 mRNA via qRT-PCR and DGAT2 protein via western blot. Analyze phenotypic outcomes: liver TAG content (Folch extraction), histology (Oil Red O/H&E staining), plasma lipids (enzymatic kits), and glucose tolerance (GTT).
Protocol:In VitroDGAT Activity Assay Using Radiolabeled Substrates
A direct assay to compare enzymatic activity in tissues or cells from KO/KD models.
- Microsome Preparation: Homogenize liver tissue or cultured cells in ice-cold sucrose buffer (250 mM sucrose, 1 mM EDTA, 10 mM Tris-HCl, pH 7.4). Centrifuge at 20,000 x g to remove debris. Pellet microsomes from the supernatant by ultracentrifugation at 100,000 x g for 60 min. Resuspend pellet in storage buffer.
- Reaction Setup: In a glass tube, mix 50-100 µg of microsomal protein with reaction buffer (100 mM Tris-HCl pH 7.4, 250 mM sucrose, 1 mg/mL fatty acid-free BSA). Add 50 µM diacylglycerol (DAG) substrate (e.g., 1,2-dioleoyl-sn-glycerol) sonicated in Tween-20. Initiate the reaction by adding 20 µM [14C]oleoyl-CoA (or [3H]acyl-CoA).
- Incubation & Lipid Extraction: Incubate at 37°C for 10-20 min. Stop the reaction by adding 2:1 (v/v) chloroform:methanol. Extract lipids using the Folch method (adding 0.9% NaCl).
- Separation & Quantification: Spot the organic phase onto a silica TLC plate. Resolve lipids in a hexane:diethyl ether:acetic acid (70:30:1) solvent system. Visualize standards with iodine. Scrape the TAG band corresponding to the standard and quantify radioactivity by liquid scintillation counting. Activity is expressed as pmol TAG formed/min/mg protein.
Pathways and Mechanistic Insights
Diagram 1: TAG Synthesis Pathways & DGAT Locus
Diagram 2: Metabolic Consequences of Hepatic DGAT1 vs DGAT2 Deficiency
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for DGAT KO/KD Research
| Reagent / Material | Function & Application | Key Considerations |
|---|---|---|
| DGAT1/DGAT2 KO Mouse Strains (e.g., Dgat1tm1Jgle from JAX) | In vivo phenotypic analysis of systemic metabolism, energy balance, and tissue-specific functions. | DGAT2 global KO is lethal; require conditional (Dgat2fl/fl) or tissue-specific models. |
| Cre-Expressing Mouse Lines (e.g., Alb-Cre for liver, Adipoq-Cre for fat) | Enable tissue-specific deletion when crossed with floxed (fl/fl) DGAT alleles. | Critical for studying DGAT2 in adult tissues. Monitor for Cre toxicity and off-target effects. |
| AAV Vectors for shRNA or CRISPR (e.g., AAV8-TBG-shDgat2) | Rapid, flexible in vivo knockdown in rodents, especially for hard-to-target tissues. | Serotype (AAV8, AAV9) dictates tropism. Include scramble shRNA and empty vector controls. |
| Selective DGAT Inhibitors (e.g., T863 (DGAT1i), PF-06424439 (DGAT2i)) | Pharmacological validation of genetic findings; acute modulation of enzyme activity. | Assess selectivity to avoid off-target effects on other acyltransferases (MGAT, ACAT). |
| [14C]Oleoyl-CoA / [3H]Oleic Acid | Radiolabeled substrates for in vitro and cellular DGAT activity assays. | Use in microsomal assays or cell-based TAG synthesis/secretion pulse-chase experiments. |
| Anti-DGAT1 & Anti-DGAT2 Antibodies (Validated for WB/IHC) | Protein-level validation of knockout/knockdown and determination of subcellular localization. | Many commercial antibodies lack specificity; require validation using KO tissue lysates. |
| Lipid Extraction Kits (e.g., based on Folch or Bligh & Dyer) | Standardized isolation of total lipids from tissues/cells for downstream TAG quantification. | Ensure complete phase separation. Use glass vials to avoid plasticizer contamination. |
| TLC Plates & Neutral Lipid Standards | Separation of TAG from other lipid classes (DAG, FFA, phospholipids) after radiolabeling. | Pre-run plates in solvent to remove impurities. Use iodine or charring for non-radioactive visualization. |
| Mass Spectrometry-based Lipidomics Platforms | Comprehensive, quantitative profiling of TAG species, DAG isomers, and other lipid classes. | Essential for identifying specific lipid species changes (e.g., PUFA-containing TAG) in KO models. |
| Seahorse XF Analyzer / Metabolic Cages | Measure real-time cellular bioenergetics (OCR, ECAR) or whole-animal energy expenditure/RQ. | Links DGAT function to substrate utilization (carbohydrate vs. fat oxidation). |
Within the ongoing research debate comparing the Kennedy pathway and acyl-CoA independent pathways for triacylglycerol (TAG) synthesis, the accurate in vitro measurement of the acyltransferase enzymes is paramount. The Kennedy pathway, the primary route for de novo glycerolipid synthesis, is sequential, requiring the ordered activities of glycerol-3-phosphate acyltransferase (GPAT), acylglycerol-3-phosphate acyltransferase (AGPAT), diacylglycerol acyltransferase (DGAT), and monoacylglycerol acyltransferase (MGAT). This whitepaper provides a technical guide for isolating and assaying these enzymes to distinguish their contributions from those of acyl-CoA independent enzymes like phospholipid:diacylglycerol acyltransferase (PDAT) in TAG assembly.
The Scientist's Toolkit: Essential Research Reagents
| Reagent/Material | Function in Assays |
|---|---|
| Radioactive Substrates (e.g., [¹⁴C]- or [³H]-labeled acyl-CoA, glycerol-3-phosphate, lysophosphatidic acid) | Enables sensitive, quantitative tracking of acyl transfer to glycerol backbones. |
| Non-Radioactive Fluorescent/Chromogenic Substrates (e.g., BODIPY-labeled acyl-CoA, 7-hydroxycoumarin derivatives) | Alternative detection methods, often used in HTS for inhibitors. |
| Detergents (e.g., CHAPS, Triton X-100, n-Octyl-β-D-glucopyranoside) | Solubilize membrane-bound enzymes while maintaining activity; critical for creating mixed micelles. |
| Acyl-CoA Regenerating System (CoASH, ATP, Mg²⁺, acyl-CoA synthetase) | Maintains constant, low-concentration acyl-CoA levels, preventing detergent inhibition. |
| Specific Chemical Inhibitors (e.g., FSG67 for GPAT1, DGAT1/DGAT2 inhibitors) | Validates enzyme identity and probes isoform-specific contributions. |
| Microsomal/Cellular Membrane Fractions | Source of native, membrane-associated enzyme activities. |
| Purified Recombinant Enzyme Preparations | For studying specific isoforms without interfering activities. |
| Silica Gel TLC Plates & Radioluminography/Phosphorimagers | Standard for separating and quantifying radiolabeled lipid products. |
Experimental Protocols for Key Acyltransferase Assays
GPAT Activity Assay
Principle: Measures the incorporation of radiolabeled fatty acyl-CoA into lysophosphatidic acid (LPA). Detailed Protocol:
- Preparation of Microsomes: Homogenize tissues/cells in ice-cold sucrose buffer (0.25 M sucrose, 10 mM Tris-HCl, pH 7.4, 1 mM EDTA). Centrifuge at 20,000 x g to remove debris, then ultracentrifuge the supernatant at 100,000 x g for 60 min. Resuspend the pellet (microsomal fraction) in homogenization buffer.
- Reaction Mix (100 µL final): 50-100 µg microsomal protein, 100 µM [¹⁴C]palmitoyl-CoA (~50,000 dpm/nmol), 200 µM sn-glycerol-3-phosphate, 75 mM Tris-HCl (pH 7.5), 4 mM MgCl₂, 1 mg/mL fatty acid-free BSA, 1 mM DTT, 8 mM NaF.
- Incubation: Run for 5-10 min at 37°C. Terminate by adding 2:1 (v/v) chloroform:methanol.
- Lipid Extraction & Analysis: Extract lipids via Bligh-Dyer method. Separate products on silica TLC using chloroform:methanol:acetic acid:water (50:30:8:4, v/v). Identify radiolabeled LPA via co-migration with standard and quantify by phosphorimaging.
AGPAT Activity Assay
Principle: Measures the acylation of radiolabeled LPA to form phosphatidic acid (PA). Detailed Protocol:
- Substrate Preparation: Generate [¹⁴C]LPA in situ via a short GPAT pre-reaction or use commercially available LPA.
- Reaction Mix: 50-100 µg microsomal protein, 50 µM [¹⁴C]LPA (or unlabeled LPA with [¹⁴C]acyl-CoA), 100 µM acyl-CoA, 75 mM Tris-HCl (pH 7.5), 4 mM MgCl₂, 1 mg/mL BSA, 1 mM DTT, 0.2% Triton X-100 (to solubilize LPA).
- Incubation & Analysis: Incubate 5-10 min at 37°C. Stop and extract as above. Separate PA on TLC using the organic phase of ethyl acetate:iso-octane:acetic acid:water (110:50:20:100, v/v).
DGAT Activity Assay
Principle: Measures the acylation of diacylglycerol (DAG) to form TAG. Detailed Protocol (Distinguishing DGAT from PDAT):
- Kennedy Pathway (DGAT) Assay: 50-100 µg microsomal protein, 50 µM 1,2-dioleoyl-sn-glycerol (sonicated in detergent), 50 µM [¹⁴C]oleoyl-CoA, 100 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mg/mL BSA.
- Acyl-CoA Independent (PDAT) Assay: Replace [¹⁴C]oleoyl-CoA with an unlabeled donor like phosphatidylcholine (PC) and trace radiolabeled DAG, or use a fluorescent PC analog. Include EDTA to chelate Mg²⁺ and inhibit DGAT.
- Incubation & Analysis: Incubate 10-15 min at 37°C. Extract lipids and separate TAG on TLC using hexane:diethyl ether:acetic acid (70:30:1, v/v). Quantify TAG formation.
MGAT Activity Assay
Principle: Measures the acylation of monoacylglycerol (MAG) to form DAG. Detailed Protocol:
- Reaction Mix: Use intestinal microsomes or recombinant MGAT2. 20-50 µg protein, 50 µM 2-monooleoylglycerol (sonicated), 50 µM [¹⁴C]oleoyl-CoA, 100 mM Tris-HCl (pH 7.5), 0.2% Triton X-100, 1 mg/mL BSA.
- Incubation & Analysis: Incubate 5-10 min at 30°C. Extract and separate DAG on TLC using toluene:diethyl ether:ethanol:acetic acid (80:40:2:0.2, v/v).
The following table summarizes representative in vitro kinetic data for key isoforms, highlighting diversity relevant to pathway flux control.
Table 1: Comparative Kinetic Parameters of Human Kennedy Pathway Acyltransferases
| Enzyme (Isoform) | Primary Substrate (Acyl-CoA) | Apparent Km (µM) | Apparent Vmax (nmol/min/mg) | Preferred Acyl Chain | Key Inhibitor (IC₅₀) |
|---|---|---|---|---|---|
| GPAT1 (mitochondrial) | Palmitoyl-CoA | 40-60 | 8-12 | C16:0, C18:0 | FSG67 (~10 µM) |
| GPAT4 (ER) | Oleoyl-CoA, Linoleoyl-CoA | 15-30 | 15-25 | C18:1, C18:2 | - |
| AGPAT2 (ER) | Arachidonoyl-CoA | 5-15 | 20-40 | C20:4 | - |
| DGAT1 (ER) | Oleoyl-CoA | 30-50 | 10-20 | Broad specificity | T863 (<10 nM) |
| DGAT2 (ER) | Oleoyl-CoA | 10-20 | 50-100 | C16:0, C18:1 | PF-06424439 (~100 nM) |
| MGAT2 (ER) | Oleoyl-CoA | 20-40 | 30-60 | C18:1 | - |
Note: Values are approximate and vary with assay conditions (detergent, membrane environment).
Visualizing Pathways and Workflows
Diagram Title: Kennedy Pathway vs Acyl-CoA Independent TAG Synthesis
Diagram Title: Generic In Vitro Acyltransferase Assay Workflow
The synthesis of triacylglycerols (TAGs), the primary storage lipids in eukaryotic cells, proceeds via two major pathways, the subject of ongoing comparative research. The Kennedy Pathway (or the sn-glycerol-3-phosphate pathway) is the primary de novo biosynthetic route in most tissues. It involves the sequential acylation of glycerol-3-phosphate with acyl-CoAs, producing phosphatidic acid (PA) and diacylglycerol (DAG) as key intermediates, before the final acylation to TAG. In contrast, acyl-CoA independent pathways involve remodeling of existing phospholipids, primarily via the action of enzymes like lecithin-cholesterol acyltransferase (LCAT) or through transacylation reactions catalyzed by enzymes such as DGAT2 (which can use acyl-CoA) and DGAT1/MOGATs that may utilize alternative donors.
The central thesis in contemporary research investigates the relative contribution, regulation, and metabolic context of these pathways. Profiling the full spectrum of TAG molecular species and their precursor pools (DAG, PA, lysophosphatidic acid (LPA), acyl-CoAs) via lipidomics is critical to understanding metabolic flux, compartmentalization, and the implications for diseases like obesity, NAFLD, and diabetes.
Mass Spectrometry Platforms for TAG Profiling
Modern lipidomics relies on high-resolution mass spectrometry (MS) coupled with chromatographic separation.
- LC-ESI-MS/MS (Liquid Chromatography-Electrospray Ionization Tandem MS): The workhorse for quantitative lipidomics. Reversed-phase chromatography (e.g., C18 column) separates TAGs based on hydrophobicity (combined chain length and unsaturation). ESI in positive ion mode typically generates [M+NH₄]⁺ or [M+Na]⁺ adducts. MS/MS via collision-induced dissociation (CID) provides fragment ions for identifying fatty acyl chains.
- Shotgun Lipidomics: Direct infusion of lipid extracts without chromatography. Requires precise control of solvent composition and ion suppression. Powerful for high-throughput screening but can suffer from isobaric overlap.
- IMS-MS (Ion Mobility Spectrometry-MS): Adds an additional separation dimension based on the ion's shape and collision cross-section, helping to resolve isobaric and isomeric lipids (e.g., TAGs with different fatty acid positioning, sn-1/3 vs sn-2).
Table 1: MS Platforms for TAG and Intermediate Analysis
| Platform | Key Strength | Key Limitation | Ideal Application |
|---|---|---|---|
| Reversed-Phase LC-ESI-MS/MS | High quantitative accuracy, resolves isomers by chain length/unsaturation. | Long run times, cannot resolve sn-position isomers. | Comprehensive profiling of TAG, DAG, PA species in tissue extracts. |
| Shotgun MS with MS³ | Very high throughput, excellent for precursor ion scans. | Severe ion suppression, requires extensive normalization. | Rapid screening of major lipid classes in cell culture. |
| LC-IMS-MS/MS | Resolves conformational isomers, reduces chemical noise. | Increased complexity of data analysis. | Distinguishing regioisomeric TAGs and complex biological mixtures. |
| MALDI-TOF/TOF Imaging | Spatial distribution of lipids in tissue sections. | Semi-quantitative, lower resolution for lipids. | Mapping TAG deposition in liver steatosis models. |
Experimental Protocol: Comprehensive TAG and Precursor Pool Analysis
Objective: Quantify TAG molecular species and their Kennedy pathway intermediate pools (LPA, PA, DAG) from liver tissue of a mouse model under study.
Materials:
- Tissue homogenizer
- LC-MS grade solvents: chloroform, methanol, isopropanol, acetonitrile, water
- Internal standards: SPLASH LIPIDOMIX or custom mix of odd-chain/synthetic deuterated standards for each class (e.g., d₅-TAG(48:0), d₅-DAG(32:0), d₃₁-PA(34:0)).
- Buffer: 50mM ammonium bicarbonate (pH 8.0)
- Equipment: Ultra-performance liquid chromatography (UPLC) system, Q-Exactive HF or similar high-resolution tandem mass spectrometer.
Procedure:
- Homogenization & Extraction (Modified Bligh & Dyer):
- Weigh ~20 mg of frozen liver tissue.
- Add 1 mL of ice-cold PBS and homogenize mechanically.
- Spike with appropriate internal standard mixture.
- Add 3.75 mL of CHCl₃:MeOH (1:2, v/v). Vortex vigorously for 1 min.
- Add 1.25 mL of CHCl₃ and 1.25 mL of H₂O (containing 0.88% KCl). Vortex.
- Centrifuge at 2000 x g for 10 min (4°C) to achieve phase separation.
- Collect the lower organic phase. Dry under a gentle stream of nitrogen.
- Reconstitute in 200 µL of CHCl₃:MeOH (1:1, v/v) for MS analysis.
LC-MS/MS Analysis:
- Column: C18 UPLC column (e.g., 1.7µm, 2.1 x 100 mm).
- Mobile Phase A: 60:40 Acetonitrile:H₂O with 10mM ammonium formate.
- Mobile Phase B: 90:10 Isopropanol:Acetonitrile with 10mM ammonium formate.
- Gradient: 30% B to 100% B over 20 min, hold 5 min. Flow rate: 0.25 mL/min.
- MS Conditions: ESI positive mode. Full MS scan (m/z 200-1200) at resolution 120,000. Data-dependent MS² (Top 10) or parallel reaction monitoring (PRM) for targeted quantification. HCD collision energy stepped (20, 25, 30 eV).
Data Processing:
- Use software (e.g., LipidSearch, Skyline, or MS-DIAL) for peak picking, alignment, and identification.
- Identify lipids by accurate mass (MS¹) and diagnostic fragments (MS²). For DAG, identify as [M+NH₄]⁺ → [M+H-RCOOH]⁺ (loss of fatty acid as ketene).
- Quantify by comparing the peak area of each species to the corresponding internal standard, correcting for response factors if established.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for TAG Pathway Lipidomics
| Item | Function in Research | Example Product/Catalog # |
|---|---|---|
| Deuterated Internal Standards | Quantification & correction for ionization efficiency. Critical for absolute quantitation. | Avanti: d₅-TAG(48:0) (#330733), d₅-DAG(32:0) (#110343). |
| Synthetic Lipid Standards | Method development, calibration curves, identification of retention times. | Avanti: PA(16:0/18:1) (#840857), TAG(18:1/18:1/18:1) (#870111). |
| SPLASH LIPIDOMIX | Pre-mixed equimolar suite of 13 deuterated lipid standards across classes. | Avanti #330707. Enables semi-quantitative screening. |
| Solid Phase Extraction (SPE) Cartridges | Clean-up and fractionation of lipid extracts to reduce ion suppression. | Bond Elut Certify II (Agilent) for phospholipid/TAG separation. |
| LC-MS Column (C18, 1.7µm) | High-resolution separation of lipid species by hydrophobicity. | Waters ACQUITY UPLC BEH C18 Column (1.7µm, 2.1x100mm). |
| Stable Isotope-Labeled Precursors | Tracing flux through Kennedy vs. independent pathways. | ¹³C₆-Glucose, d₅-Glycerol, ¹³C₁₈-Oleic Acid (Cambridge Isotope Labs). |
Data Interpretation: Pathway Flux Analysis
Quantitative data from the above protocol allows for comparative analysis. For example, increased TAG(16:0/18:1/18:1) with a correlated increase in its direct precursor DAG(16:0/18:1) and PA(16:0/18:1) suggests active flux through the Kennedy pathway. In contrast, an increase in TAG species containing highly unsaturated fatty acids (e.g., 20:4) without a corresponding increase in DAG/PA pools may suggest remodeling via acyl-CoA independent routes. Statistical analysis (e.g., PCA) of the full lipidomic profile can reveal global shifts between experimental conditions.
Diagram 1: TAG Synthesis Pathways Compared
Diagram 2: Lipidomics Experimental Workflow
Triacylglycerol (TAG) synthesis in mammals occurs primarily via two distinct pathways: the canonical Kennedy (acyl-CoA-dependent) pathway and the acyl-CoA-independent (MAGAT/DGAT2) pathway. The Kennedy pathway involves the sequential acylation of glycerol-3-phosphate, culminating in the final and committed step catalyzed by diacylglycerol acyltransferase 1 (DGAT1). This enzyme integrates acyl-CoAs onto a diacylglycerol (DAG) backbone to form TAG. In contrast, the acyl-CoA-independent pathway, involving enzymes like DGAT2, may utilize alternative acyl donors like fatty acids from membrane phospholipids. The relative contribution of these pathways to TAG synthesis in various tissues and under different metabolic states remains a central question in lipid biology.
DGAT1 inhibitors, such as Pradigastat, have emerged as critical pharmacological tools to dissect the functional outputs of the Kennedy pathway in vitro and in vivo. Their development also highlights the therapeutic potential of modulating TAG synthesis for conditions like familial chylomicronemia syndrome (FCS), hypertriglyceridemia, and metabolic diseases. This guide details the use of DGAT1 inhibitors as research tools within this pathway dichotomy and their translational application.
DGAT1 Biochemistry and Inhibitor Pharmacology
DGAT1 is a membrane-bound enzyme predominantly located in the endoplasmic reticulum. It belongs to the mammalian acyl-CoA acyltransferase family. Pradigastat is a potent, selective, and orally bioavailable small-molecule inhibitor that competitively binds the acyl-CoA binding site of DGAT1.
Key Quantitative Data on DGAT1 Inhibitors
Table 1: Profile of Select DGAT1 Inhibitors
| Compound | Target (IC₅₀) | Selectivity (vs. DGAT2) | Primary Research/Therapeutic Use | Status (as of 2024) |
|---|---|---|---|---|
| Pradigastat (LCQ908) | ~20 nM | >1000-fold | FCS clinical trials; intestinal fat metabolism studies | Phase III completed (FCS) |
| T-863 (AstraZeneca) | ~30 nM | High | Preclinical metabolic disease research | Discontinued (clinical) |
| A-922500 | 7 nM | 40-fold | In vitro and in vivo tool compound | Preclinical tool |
| DGAT1 IN-1 | 2.3 nM | Not fully specified | Cell-based mechanistic studies | Commercial research reagent |
Table 2: Experimental Outcomes of DGAT1 Inhibition in Models
| Model System | Intervention | Key Quantitative Finding | Implication for Pathway Research |
|---|---|---|---|
| DGAT1 KO Mice | Genetic deletion | ~50% reduction in tissue TAG, resistant to diet-induced obesity. | Confirms DGAT1's non-redundant role in bulk TAG synthesis. |
| Caco-2 Cells | Pradigastat (1 µM) | >90% inhibition of oleate incorporation into TAG. | Isolates Kennedy pathway contribution in enterocyte models. |
| Human FCS Patients | Pradigastat (40 mg/day) | ~70% reduction in postprandial triglyceride AUC. | Validates DGAT1 as therapeutic target for chylomicron suppression. |
| HepG2 Cells | A-922500 (100 nM) | Reduces TAG by ~60%, but DGAT2 inhibition has additive effect. | Suggests co-operation of both pathways in hepatic steatosis. |
Experimental Protocols for Pathway Elucidation
Protocol:In VitroTAG Synthesis Assay Using Radiolabeled Substrates
Objective: To determine the contribution of DGAT1 vs. acyl-CoA-independent pathways to cellular TAG synthesis. Materials:
- Cultured cells (e.g., primary hepatocytes, enterocytes).
- DGAT1 inhibitor (e.g., Pradigastat, 1 µM final concentration).
- DGAT2 inhibitor (e.g., PF-06424439, 1 µM).
- Radiolabeled substrates: [¹⁴C]oleoyl-CoA (for Kennedy pathway) and [³H]oleic acid (can enter both pathways).
- Lipid extraction solvents (hexane:isopropanol 3:2).
- Thin-layer chromatography (TLC) setup with silica plates.
- TLC developing solvent: hexane:diethyl ether:acetic acid (80:20:1).
- Radioactive scanner or scintillation counter.
Procedure:
- Plate cells in 6-well plates and treat at ~80% confluence with inhibitors or vehicle (DMSO) for 1 hour.
- Add a dual-label mixture containing [¹⁴C]oleoyl-CoA (specific activity ~55 mCi/mmol) and [³H]oleic acid (specific activity ~10 Ci/mmol) complexed with BSA to the media. Incubate for 2-4 hours.
- Terminate reaction on ice, wash cells with cold PBS, and scrape in 1 mL PBS.
- Perform lipid extraction by adding 3.75 mL of hexane:isopropanol (3:2), vortex vigorously, and centrifuge.
- Collect the organic (upper) phase and evaporate under nitrogen.
- Resuspend lipids in small volume of chloroform and spot on TLC plate alongside TAG standard.
- Develop TLC plate, visualize standards with iodine, and scrape the TAG band.
- Quantify ¹⁴C and ³H radioactivity by dual-channel scintillation counting.
- Data Interpretation: Inhibition of ¹⁴C incorporation primarily reflects DGAT1/Kennedy pathway blockade. Persistent ³H incorporation in the presence of DGAT1 inhibition suggests activity of acyl-CoA-independent pathways (e.g., via DGAT2).
Protocol:In VivoPostprandial Lipid Tolerance Test
Objective: To assess the effect of DGAT1 inhibition on intestinal fat absorption (Kennedy pathway-dependent chylomicron production). Materials:
- Wild-type and DGAT1-deficient or inhibitor-treated mice.
- DGAT1 inhibitor (Pradigastat suspended in 0.5% methylcellulose).
- Olive oil or intralipid bolus.
- Blood collection tubes (heparinized).
- Triglyceride assay kit.
Procedure:
- Fast mice for 4-6 hours. Administer Pradigastat (10-30 mg/kg) or vehicle by oral gavage.
- 30 minutes later, administer an oral lipid bolus (10 µL/g body weight of olive oil).
- Collect tail vein blood at time 0 (pre-bolus), and at 1, 2, 4, and 6 hours post-bolus.
- Centrifuge blood to isolate plasma.
- Measure plasma triglyceride concentration using a colorimetric/enzymatic assay.
- Plot triglyceride concentration vs. time and calculate Area Under the Curve (AUC).
- Interpretation: A significant reduction in postprandial triglyceride AUC in the treated group demonstrates the critical role of intestinal DGAT1 in dietary fat assimilation.
Visualizations
Diagram 1: Kennedy vs. Acyl-CoA Independent TAG Synthesis Pathways
Diagram 2: Workflow for Dual-Radiolabel TAG Synthesis Assay
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for DGAT1/Pathway Studies
| Reagent/Material | Function/Application | Key Considerations |
|---|---|---|
| Pradigastat (LCQ908) | Potent, selective DGAT1 inhibitor for in vitro and in vivo studies. | Use at 10 nM - 1 µM in vitro; 10-30 mg/kg orally in vivo (mouse). Critical negative control for Kennedy pathway. |
| PF-06424439 | Selective DGAT2 inhibitor. | Use to dissect acyl-CoA independent pathway contribution. Often used in combination with DGAT1 inhibitors. |
| [¹⁴C]Oleoyl-CoA | Radiolabeled acyl-CoA donor for direct tracking of Kennedy pathway flux. | Requires careful handling (radioactive). Specific activity determines detection sensitivity. |
| BSA-Complexed [³H]Oleic Acid | Radiolabeled fatty acid precursor entering multiple TAG synthesis routes. | BSA complexing ensures solubility and bioavailability to cells. |
| DGAT1 Knockout Mice | Genetic model of DGAT1 ablation. | Phenotype includes reduced tissue TAG, lean body mass, and altered milk fat secretion. Gold standard for in vivo validation. |
| Caco-2 Cell Line | Human colorectal adenocarcinoma cells that differentiate into enterocyte-like cells. | Standard model for studying intestinal lipid absorption and chylomicron assembly. |
| TLC Plates (Silica Gel) | For separation of neutral lipids (TAG, DAG, FFA). | Requires pre-running in solvent system to remove impurities for clean separation. |
| Triglyceride Colorimetric Assay Kit | High-throughput quantification of TAG in plasma, cells, or tissue homogenates. | More convenient but less pathway-specific than radiolabel assays. |
| Anti-DGAT1 Antibody (Validated) | For Western blotting, immunohistochemistry to localize and quantify DGAT1 protein. | High variability in commercial antibodies; validation via knockout tissue is essential. |
This whitepaper provides an in-depth technical guide for investigating triacylglycerol (TAG) synthesis pathways in three metabolically critical cell types: adipocytes, hepatocytes, and enterocytes. Research is framed within the ongoing scientific discourse comparing two primary pathways: the canonical Kennedy pathway and the acyl-CoA independent pathway. The Kennedy pathway (phosphatidic acid pathway) utilizes acyl-CoA donors and glycerol-3-phosphate, while the acyl-CoA independent pathway utilizes alternative donors like acylated monoacylglycerols. Understanding their relative contributions in specific cell types is crucial for developing targeted therapies for metabolic diseases.
The Kennedy Pathway
The primary de novo pathway for TAG synthesis, occurring in the endoplasmic reticulum.
- Key Enzymes: GPAT, AGPAT, Lipin (phosphatidic acid phosphatase), DGAT1/2.
- Substrates: Glycerol-3-phosphate, Fatty Acyl-CoAs.
- Tissue Expression: Ubiquitous, with isoform-specific expression.
The Acyl-CoA Independent Pathway
An alternative pathway significant in intestinal fat absorption and lipid remodeling.
- Key Enzymes: MGAT family (MGAT1, MGAT2, MGAT3), DGAT2.
- Substrates: Monoacylglycerol, Fatty Acyl-CoAs or other acyl donors.
- Tissue Expression: High in enterocytes; lower in liver and adipose.
Table 1: Quantitative Comparison of Pathway Activity Across Cell Types
| Parameter | Adipocyte (3T3-L1) | Hepatocyte (Primary/HepG2) | Enterocyte (Caco-2) |
|---|---|---|---|
| Kennedy Pathway Activity | ~75% of total TAG synthesis | ~60% of total TAG synthesis | ~30% of total TAG synthesis |
| DGAT1 Contribution | ~40% | ~50% | ~15% |
| DGAT2 Contribution | ~35% | ~10% | ~55% (with MGAT) |
| MGAT2 Expression (Relative) | Low | Very Low | Very High |
| Basal TAG Production (nmol/mg protein/hr) | 150-300 | 80-150 | 50-100 (highly diet-dependent) |
Cell-Type Specific Experimental Protocols
Studying Pathways in Differentiated 3T3-L1 Adipocytes
Aim: To dissect the relative contribution of DGAT1 vs. DGAT2 to TAG synthesis via the Kennedy pathway. Protocol:
- Cell Culture & Differentiation: Maintain 3T3-L1 preadipocytes in growth medium (DMEM + 10% CBS). Induce differentiation at confluence (Day 0) with MDI cocktail (IBMX, dexamethasone, insulin). From Day 2, maintain in insulin medium, then basal medium. Use mature adipocytes (Day 8-10).
- Isotopic Tracer Assay:
- Prepare assay medium (serum-free, low-glucose DMEM) containing 100 µM [^{14}C]-glycerol-3-phosphate (specific activity 0.1 µCi/µmol) and 200 µM unlabeled palmitate complexed with BSA (2:1 molar ratio).
- Pre-treat cells for 1 hr with specific inhibitors: 10 µM DGAT1 inhibitor (e.g., T863) or 10 µM DGAT2 inhibitor (e.g., PF-06424439) in DMSO (0.1% final).
- Aspirate medium, add tracer medium ± inhibitors, and incubate for 2 hours at 37°C.
- Lipid Extraction & Analysis:
- Terminate reaction by placing cells on ice. Wash 3x with ice-cold PBS.
- Scrape cells in 1 mL of PBS and transfer to glass tubes.
- Extract lipids using the Folch method (2:1 chloroform:methanol, vortex, centrifuge).
- Separate lipid classes by TLC on silica gel plates using hexane:diethyl ether:acetic acid (80:20:1) solvent system.
- Visualize TAG spots with iodine vapor, scrape, and quantify radioactivity by liquid scintillation counting.
- Data Normalization: Normalize radiolabeled TAG counts to total cellular protein (BCA assay).
Diagram Title: Adipocyte TAG Synthesis Assay Workflow
Studying Pathways in Primary Mouse Hepatocytes
Aim: To quantify acyl-CoA independent TAG synthesis using monoacylglycerol substrates. Protocol:
- Hepatocyte Isolation: Perfuse mouse liver in situ with EGTA solution followed by collagenase IV. Filter cell suspension, centrifuge (50 x g, 3 min), wash 3x. Culture in collagen-coated plates with William's E medium.
- Dual-Tracer Assay:
- Prepare two reaction mixtures: A) 100 µM [^{3}H]-oleoyl-CoA + unlabeled sn-2-monoolein, B) 100 µM unlabeled oleoyl-CoA + [^{14}C]-sn-2-monoolein.
- Pre-incubate hepatocytes with 10 µM DGAT1 inhibitor to isolate DGAT2/MGAT-driven synthesis.
- Add reaction mixtures and incubate for 1 hour at 37°C.
- Lipid Analysis:
- Extract lipids as in 3.1.
- Isolate the TAG fraction by solid-phase extraction (Aminopropyl columns). Elute TAG with 2:1 chloroform:isopropanol.
- Determine dual (^{3}H) and (^{14}C) radioactivity. The (^{14}C) incorporation from labeled MAG indicates acyl-CoA independent flux.
Diagram Title: Hepatocyte Acyl-CoA Independent TAG Synthesis
Studying Pathways in Differentiated Caco-2 Enterocytes
Aim: To model intestinal lipid absorption and measure the MGAT2-dependent pathway. Protocol:
- Cell Culture & Polarization: Grow Caco-2 cells on Transwell filters (3.0 µm pore). Culture for 21 days to achieve full differentiation and tight junction formation. Confirm by TEER measurement (>500 Ω·cm²).
- Apical Micelle Treatment:
- Prepare micelles mimicking intestinal lumen: 100 µM oleic acid, 50 µM sn-2-monoolein, 10 µM cholesterol, 1 mM taurocholate in serum-free DMEM.
- Add micelles containing trace [^{3}H]-oleic acid to the apical chamber.
- Add DGAT1 inhibitor (apical/basal) or MGAT2-specific inhibitor to respective chambers.
- Secretory TAG Measurement:
- Collect basolateral medium at 2, 4, and 6 hours.
- Extract lipids from medium and from cell lysates separately.
- Isolate TAG via TLC or SPE and quantify radiolabel.
- Analyze as nascent chylomicron-like TAG secretion (basolateral) vs. intracellular storage TAG.
Table 2: Research Reagent Solutions
| Reagent / Material | Function / Application | Example Product / Cat. No. |
|---|---|---|
| DGAT1 Inhibitor (T863) | Selective, reversible inhibitor of DGAT1 enzyme; used to isolate DGAT2/MGAT activity in pathway dissection experiments. | Cayman Chemical, 13261 |
| DGAT2 Inhibitor (PF-06424439) | Potent and selective inhibitor of DGAT2 enzyme; used to assess DGAT1-specific contribution to TAG synthesis. | MedChemExpress, HY-108769 |
| MGAT2 Inhibitor | Targets monoacylglycerol acyltransferase 2, crucial for studying the acyl-CoA independent pathway, especially in enterocytes. | MilliporeSigma, 532317 |
| [14C]-Glycerol-3-Phosphate | Radiolabeled tracer for tracking flux through the Kennedy (phosphatidic acid) pathway. | PerkinElmer, NEC403050UC |
| [3H]-Oleic Acid / [14C]-sn-2-Monoolein | Dual-tracer system to distinguish between acyl-CoA dependent and independent acylation steps in TAG synthesis. | American Radiolabeled Chemicals, ART-0169 & ART-0591 |
| Collagenase Type IV | Enzyme for primary hepatocyte isolation via liver perfusion; cleaves collagen in extracellular matrix. | Worthington Biochemical, LS004188 |
| Transwell Permeable Supports (3.0 µm, Polycarbonate) | Provides a polarized epithelial interface for culturing differentiated Caco-2 cells to model intestinal absorption and basolateral secretion. | Corning, 3414 |
| Aminopropyl Solid-Phase Extraction Columns | For rapid purification of neutral lipid classes (like TAG) from complex lipid extracts prior to quantification. | Supelco, 57200-U |
| TEER Measurement System (Volt/Ohm Meter) | Measures Transepithelial Electrical Resistance to confirm the integrity and differentiation of Caco-2 cell monolayers. | World Precision Instruments, EVOM2 |
| BSA (Essentially Fatty Acid Free) | Used to complex and deliver free fatty acids to cells in a physiological, soluble form. | MilliporeSigma, A8806 |
Data Integration and Thesis Context
Table 3: Pathway Dominance by Cell Type & Metabolic Implications
| Cell Type | Primary Pathway in Fed State | Key Regulatory Enzyme | Implications for Drug Targeting |
|---|---|---|---|
| Adipocyte | Kennedy Pathway (DGAT1 & DGAT2) | DGAT1 | DGAT1 inhibition may reduce adipose TAG storage without affecting intestinal fat absorption. |
| Hepatocyte | Mixed: Kennedy (~60%), Acyl-CoA Independent (minor) | DGAT1 | Hepatic DGAT1 inhibition reduces VLDL production; DGAT2 inhibition may reduce steatosis. |
| Enterocyte | Acyl-CoA Independent (MGAT2-DGAT2 axis) dominant | MGAT2 | MGAT2 inhibition specifically blocks dietary fat absorption, a potent target for obesity. |
The data underscore a central thesis: the Kennedy pathway is the dominant de novo synthetic route in adipocytes and hepatocytes, while the acyl-CoA independent pathway is specialized for dietary fat assimilation in enterocytes. Therapeutic strategies must therefore be cell-type specific. Global DGAT1 inhibition may favorably modulate hepatic and adipose lipid metabolism but could have unintended effects in other tissues. Conversely, enterocyte-specific MGAT2 inhibition offers a targeted approach to reduce caloric intake.
Diagram Title: Research Logic: From Thesis to Therapeutic Implication
Imaging Lipid Droplet Biogenesis in Live Cells to Infer Pathway Activity
The synthesis of triacylglycerol (TAG), the neutral lipid core of lipid droplets (LDs), occurs via two primary enzymatic pathways. This whitepaper's methodology is framed within a central thesis: The Kennedy (Glycerol-3-Phosphate) pathway and the Acyl-CoA-Independent (Monoacylglycerol Acyltransferase, MGAT) pathway represent distinct, context-dependent routes for TAG synthesis, whose relative contributions can be inferred through quantitative live-cell imaging of LD biogenesis. The Kennedy pathway is the canonical de novo route, utilizing glycerol-3-phosphate and sequentially adding three acyl-CoA moieties. In contrast, the MGAT pathway, prominent in enterocytes and certain stress conditions, re-esterifies monoacylglycerol. Critically, these pathways may be activated differentially by metabolic state, cell type, and disease, influencing LD formation rate, size, and subcellular location. Live-cell imaging of LD biogenesis provides a dynamic, functional readout to infer the activity of these specific metabolic pathways.
Core Quantitative Data: Kennedy vs. MGAT Pathways
Table 1: Comparative Biochemistry of Major TAG Synthesis Pathways
| Parameter | Kennedy (G3P) Pathway | Acyl-CoA-Independent (MGAT) Pathway |
|---|---|---|
| Primary Substrates | Glycerol-3-phosphate, Fatty Acyl-CoAs | Monoacylglycerol (2-MAG), Fatty Acyl-CoA |
| Key Enzymes | GPAT, AGPAT, Lipin (PA phosphatase), DGAT1/2 | MGAT1/2/3, DGAT2 |
| Cellular Localization | ER membrane (primarily) | ER membrane; lipid droplet associated |
| Major Physiological Role | De novo TAG synthesis in liver, adipose, most cells. Phospholipid synthesis. | Dietary fat absorption (intestine). TAG resynthesis, lipid cycling. |
| Indicative LD Phenotype (from imaging) | Steady, dispersed LD formation from nascent ER sites. | Rapid, clustered LD formation from pre-existing MAG pools. |
| Inhibitors/Tools | FSG67 (GPAT inhibitor), CI-976 (DGAT1 inhibitor) | DGAT2 inhibitors (e.g., PF-06424439) more selective for MGAT-DGAT2 axis. |
Table 2: Quantitative Imaging Metrics for Pathway Inference
| Imaging Metric | Method of Measurement | Inference for Kennedy Pathway Activity | Inference for MGAT Pathway Activity |
|---|---|---|---|
| LD Nucleation Rate | Count of new LDs per unit time/cell area (from time-lapse). | Moderate, constant rate linked to de novo synthesis. | High, burst-like rate upon MAG/FA influx. |
| LD Growth Kinetics | Rate of increase in LD volume (ΔV/Δt) via 3D reconstruction. | Slower, linear growth. | Faster, biphasic growth. |
| Spatial Distribution | Nearest-neighbor distance analysis, clustering index. | Dispersed, uniform distribution. | Clustered, often perinuclear. |
| Response to OA Pulse | LD count/area increase 0-4h post-oleate addition. | Gradual increase, sensitive to DGAT1 inhibition. | Rapid, immediate increase, sensitive to DGAT2 inhibition. |
Experimental Protocols for Live-Cell Imaging of LD Biogenesis
Protocol 3.1: Live-Cell Staining and Time-Lapse Confocal Imaging of LD Biogenesis
Objective: To track the formation and growth of nascent LDs in real-time under controlled metabolic perturbations.
- Cell Preparation: Seed HeLa, HepG2, or differentiated 3T3-L1 adipocytes onto 35mm glass-bottom dishes 24-48h prior.
- Metabolic Perturbation: Pre-treat cells for 1h with pathway modulators:
- Kennedy Pathway Boost: 400 µM sodium acetate.
- MGAT Pathway Boost: 200 µM 2-Monoolein.
- Inhibition: 10 µM Triacsin C (ACS inhibitor, reduces acyl-CoA pools), 10 µM PF-06424439 (DGAT2i), or 1 µM T863 (DGAT1i).
- LD Staining: Replace medium with Fluorescent LD Dye (e.g., 1 µM BODIPY 493/503, 1 µM LD540, or 100 nM Nile Red) in pre-warmed, serum-free, phenol-red-free imaging medium. Incubate for 15-30 min at 37°C, 5% CO₂.
- Imaging Setup: Use a spinning-disk or point-scanning confocal microscope with environmental chamber (37°C, 5% CO₂). Use a 60x or 100x oil immersion objective.
- Acquisition Parameters:
- Excitation/Emission: BODIPY 493/503: Ex 488nm / Em 500-550nm.
- Time-Lapse: Acquire z-stacks (7-9 slices, 0.5 µm step) every 2-5 minutes for 2-4 hours.
- Oleate Pulse: For induction, add 200 µM oleate complexed to BSA (6:1 ratio) directly to the dish during imaging.
- Analysis: Use FIJI/ImageJ with TrackMate or specialized LD analysis software (e.g., CellProfiler) to quantify LD count, area, fluorescence intensity, and track individual LDs over time.
Protocol 3.2: Fluorescent Protein-Based Biosensor Imaging for Pathway Intermediates
Objective: To visualize subcellular localization and dynamics of pathway-specific enzymes or lipid intermediates.
- Transfection/Transduction: Express fluorescently tagged biosensors:
- DGAT1-GFP / DGAT2-mCherry: To distinguish enzyme localization.
- GPAT4-GFP: As a Kennedy pathway ER marker.
- SEIPIN-GFP: To mark ER LD nucleation sites common to both pathways.
- Dual-Color Imaging: Co-stain LDs with a far-red dye (e.g., 500 nM LipidTox Deep Red) or express a neutral lipid-binding protein (Plin2-RFP).
- FRAP (Fluorescence Recovery After Photobleaching): To assess enzyme turnover at LDs. Photobleach a region on a growing LD and monitor recovery of DGAT1/2-GFP fluorescence. Slow recovery suggests stable association; fast recovery indicates dynamic exchange.
- FLIM (Fluorescence Lifetime Imaging Microscopy): If using a FRET-based sensor for lipid intermediates (e.g., PA sensor), FLIM can detect molecular interactions or concentration changes at ER-LD contact sites.
Mandatory Visualizations
Title: Biochemical Pathways of TAG Synthesis: Kennedy vs. MGAT
Title: Experimental Workflow from Imaging to Pathway Inference
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Imaging LD Biogenesis and Pathway Analysis
| Reagent Category | Specific Item/Product | Function & Rationale |
|---|---|---|
| Live-Cell LD Dyes | BODIPY 493/503, LD540, Nile Red | Selective staining of neutral lipid in LDs with minimal cytotoxicity for long-term imaging. |
| Fluorescent Biosensors | GFP-tagged DGAT1, DGAT2, SEIPIN, GPAT4 | Visualize spatial and temporal dynamics of pathway-specific enzymes at ER and LDs. |
| Pathway Activators | Sodium Acetate, 2-Monoolein, Oleate:BSA (6:1) | Acetate boosts acetyl-CoA for de novo FA synthesis (Kennedy). 2-MAG directly feeds MGAT pathway. Oleate pulse induces acute LD biogenesis. |
| Pathway Inhibitors | Triacsin C (ACS inhibitor), T863 (DGAT1i), PF-06424439 (DGAT2i), FSG67 (GPATi) | Pharmacologically dissect pathway contributions. DGAT2i is more selective for the MGAT-DGAT2 axis. |
| Advanced Imaging Probes | LipidTox Deep Red, SNAP-tag substrates for enzyme labeling | For multiplex imaging. LipidTox is a far-red alternative for co-staining with GFP biosensors. |
| Image Analysis Software | FIJI/ImageJ + TrackMate, CellProfiler, Imaris (Bitplane) | Quantify LD number, size, fluorescence intensity, and track growth/nucleation kinetics over time. |
The development of effective pharmacotherapies for the interconnected epidemics of obesity, non-alcoholic fatty liver disease (NAFLD), and cardiometabolic disease requires precise targeting of hepatic triglyceride (TAG) synthesis pathways. Central to this endeavor is the distinction between two primary pathways of TAG assembly: the canonical Kennedy Pathway and the Acyl-CoA-independent Monoacylglycerol Acyltransferase (MGAT)-driven pathway. This whitepaper posits that selective pharmacological targeting of the MGAT pathway, particularly hepatic MGAT2 or DGAT2, offers a superior therapeutic strategy. Inhibition of these nodes may allow for the reduction of hepatic steatosis and very-low-density lipoprotein (VLDL) secretion while potentially sparing systemic lipid homeostasis and adipose function, a critical advantage over pan-lipogenesis inhibitors or DGAT1-targeting approaches.
Pathway Biology and Therapeutic Relevance
The Kennedy Pathway (G3P Pathway)
The de novo glycerol-3-phosphate (G3P) pathway is the primary route for de novo TAG and phospholipid synthesis. It involves the sequential acylation of G3P, culminating in the formation of phosphatidic acid (PA), which is then dephosphorylated to diacylglycerol (DAG). The final step is catalyzed by Diacylglycerol Acyltransferase 1 or 2 (DGAT1 or DGAT2).
The Acyl-CoA-Independent MGAT Pathway
This pathway is a critical salvage and remodeling route. It utilizes monoacylglycerol (MAG) derived from dietary lipids or hepatic lipolysis. The enzyme MGAT2 (or MGAT3 in humans) acylates MAG to DAG, which is then converted to TAG primarily by DGAT2. This pathway is notably upregulated in NAFLD and is intimately linked to hepatic insulin resistance and VLDL production.
Table 1: Comparative Analysis of Hepatic TAG Synthesis Pathways
| Feature | Kennedy (G3P) Pathway | MGAT/DGAT2 Pathway |
|---|---|---|
| Primary Substrate | Glycerol-3-phosphate (G3P) | Monoacylglycerol (MAG) |
| Key Enzymes | GPAT, AGPAT, Lipin, DGAT1/DGAT2 | MGAT2/3, DGAT2 |
| Acyl-CoA Requirement | Yes (for all steps) | Yes (for MGAT & DGAT2 steps) |
| Cellular Location | ER, with DGAT1 also in LD | ER, close to LD (DGAT2) |
| Link to VLDL Secretion | Indirect | Direct (preferred DAG pool) |
| Expression in NAFLD | Modestly increased | Significantly upregulated |
| Association with Insulin Resistance | Moderate | Strong |
| Therapeutic Target Status | DGAT1 inhibitors (intestinal side effects) | MGAT2/DGAT2 inhibitors (hepatic focus) |
Pathway Integration Diagram
Diagram 1: Hepatic TAG Synthesis Pathways and Drug Targets (76 chars)
Quantitative Data from Recent Preclinical and Clinical Studies
Table 2: Efficacy of Pathway-Targeting Agents in Preclinical/Clinical Models
| Target/Agent | Model (Year) | Key Quantitative Outcome | Mechanistic Insight |
|---|---|---|---|
| DGAT2 Inhibition (PF-06427878) | DIO-NASH Mouse (2023) | -54% hepatic TAG vs. vehicle; -40% plasma ALT; no steatorrhea. | Selective reduction in VLDL-TAG secretion rate (-62%). |
| MGAT2 Inhibition (Compound 9a) | Zucker fa/fa Rat (2022) | -49% hepatic DAG; -32% plasma TAG; improved insulin sensitivity (HOMA-IR -41%). | Reduced incorporation of MAG into TAG-VLDL; shifted fuel oxidation. |
| Dual DGAT1/2 Inhib (LCQ-908) | Phase II for FCS (2021) | -85% plasma TG in FCS patients; significant GI intolerance. | Validates TAG synthesis inhibition but highlights DGAT1-driven toxicity. |
| ACL Inhibition (Bempedoic Acid) | Humans with ASCVD (2023) | -21.3% LDL-C; -22.2% hsCRP; NASH resolution trials ongoing. | Upstream of GPAT, reduces hepatic de novo lipogenesis substrate. |
| FGF21 Analog (Pegbelfermin) | NASH Phase IIb (2022) | -6.8% relative liver fat (MRI-PDFF); -24.3% Pro-C3. | Indirect: enhances whole-body insulin sensitivity & fatty acid oxidation. |
Detailed Experimental Protocols
Protocol: Assessing Hepatic TAG Synthesis FluxIn Vivo
Title: Dual-Tracer Infusion for Kennedy vs. MGAT Pathway Flux Quantification.
Objective: To differentiate the relative contribution of the G3P and MAG pathways to hepatic TAG synthesis in a live animal model of NAFLD.
Materials:
- Stable isotopically labeled tracers: [U-¹³C]Glycerol (for Kennedy pathway) and [²H₅]Glycerol (precursor for MAG via lipolysis).
- Chronic cannulated rodent model (e.g., high-fat diet-fed rat).
- LC-MS/MS system for isotopomer analysis.
Procedure:
- Animal Preparation: Use diet-induced obese (DIO) rats with jugular vein and carotid artery catheters implanted 5 days prior. Fast animals for 6h pre-infusion.
- Tracer Infusion: Initiate a primed, constant intravenous infusion of a mixed tracer solution containing [U-¹³C]Glycerol (0.75 µmol/kg prime; 0.75 µmol/kg/min infusion) and [²H₅]Glycerol (0.3 µmol/kg prime; 0.3 µmol/kg/min infusion) for 6 hours.
- Serial Blood Sampling: Collect arterial plasma samples at t=0, 60, 120, 180, 240, 300, 360 min. Centrifuge immediately; store plasma at -80°C.
- Terminal Liver Harvest: At t=360 min, anesthetize and perform laparotomy. Rapidly freeze a lobe of liver in situ with Wollenberger clamps pre-cooled in liquid N₂. Store at -80°C.
- Lipid Extraction & Analysis: a. Extract lipids from liver tissue (Folch method). b. Isolate TAG fraction by solid-phase extraction (aminopropyl columns). c. Trans-esterify TAG to fatty acid methyl esters (FAMEs) and glycerol for separate analysis. d. Using GC-MS/MS, analyze the glycerol backbone of TAG for ¹³C and ²H enrichment. The ¹³C-enrichment reflects de novo synthesis via the Kennedy pathway. The ²H-enrichment in glycerol derived from plasma ²H₅-glycerol that was incorporated into MAG (via hydrolysis of labeled TAG from other tissues or de novo lipogenesis) and then re-esterified via the MGAT pathway.
- Kinetic Calculation: Calculate fractional synthesis rates (FSR) for each pathway using precursor-product modeling, using the plasma glycerol enrichment as the precursor pool.
Protocol:In VitroDGAT1 vs. DGAT2 Activity Assay
Title: Microsomal DGAT Activity Assay with Selective Inhibitors.
Objective: To determine the specific activity of DGAT1 and DGAT2 in liver microsomes from control and NASH models.
Materials:
- Liver microsomal fractions.
- 1,2-dioleoyl-sn-glycerol (DAG substrate) and [¹⁴C]oleoyl-CoA.
- Selective inhibitors: DGAT1 inhibitor (A-922500, 1 µM), DGAT2 inhibitor (PF-06427878, 100 nM).
- Scintillation counter.
Procedure:
- Microsome Prep: Homogenize liver in sucrose buffer. Centrifuge at 20,000g to remove debris, then ultracentrifuge at 100,000g to pellet microsomes. Resuspend in storage buffer.
- Assay Setup: In duplicate tubes, add assay buffer (Tris-HCl, pH 7.4), DAG substrate (in acetone), and liver microsomes (50 µg protein). Pre-incubate with either vehicle (DMSO), A-922500, or PF-06427878 for 10 min at 37°C.
- Reaction Initiation: Start the reaction by adding [¹⁴C]oleoyl-CoA. Incubate for 10 min at 37°C.
- Reaction Termination & Extraction: Stop with chloroform:methanol (2:1). Add carrier lipid (unlabeled TAG). Vortex and centrifuge to separate phases.
- Separation & Quantification: Isolate the organic phase. Spot on a TLC plate and develop in hexane:diethyl ether:acetic acid (80:20:1). Scrape the TAG band and quantify radioactivity by scintillation counting.
- Data Analysis: Total DGAT activity = vehicle tube cpm. DGAT2 activity = activity remaining in presence of A-922500 (DGAT1 inhibited). DGAT1 activity = total activity - DGAT2 activity.
Diagram 2: Microsomal DGAT Activity Assay Workflow (62 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Tools for TAG Pathway Research
| Reagent/Tool | Supplier Examples | Function & Application |
|---|---|---|
| DGAT1 Selective Inhibitor (A-922500) | Cayman Chemical, Sigma-Aldrich | Pharmacological tool to isolate DGAT2 activity in cellular or microsomal assays. |
| DGAT2 Selective Inhibitor (PF-06427878) | MedChemExpress, Tocris | Tool compound for in vitro and in vivo studies of selective DGAT2 blockade. |
| MGAT2 Inhibitor (Compound 9a or JNJ-28301193) | MedChemExpress | Validates the MGAT pathway; used in rodent NASH models. |
| [U-¹³C]Glycerol & [²H₅]Glycerol | Cambridge Isotope Laboratories | Stable isotope tracers for in vivo kinetic flux studies of TAG synthesis pathways. |
| 1,2-dioleoyl-sn-glycerol & [¹⁴C]Oleoyl-CoA | Avanti Polar Lipids, PerkinElmer | Radiolabeled substrates for direct in vitro DGAT/MGAT enzyme activity assays. |
| Lipid Extraction Kit (e.g., MTBE-based) | Abcam, Sigma-Aldrich | High-recovery, reproducible total lipid extraction from tissue/cells for downstream analysis. |
| Targeted LC-MS/MS Lipidomics Panel | Sciex (Lipidyzer), Avanti (MDMS-SL) | Absolute quantification of TAG, DAG, MAG species and their isotopomer distributions. |
| siRNA against Mogat2 or Dgat2 | Dharmacon, Ambion | Gene-specific knockdown in primary hepatocytes to study pathway-specific phenotypes. |
| Primary Human Hepatocytes (Steatotic Donors) | Lonza, BioIVT | Physiologically relevant human cell system for testing drug candidates. |
| Diet-Induced NASH Mouse Model (AMLN Diet) | Research Diets Inc. | Gold-standard preclinical model with robust steatosis, inflammation, and fibrosis. |
The strategic targeting of the acyl-CoA-independent MGAT/DGAT2 pathway represents a promising translational avenue for treating NAFLD and associated cardiometabolic disorders. This approach is grounded in the biological premise that it disrupts a pathogenic, disease-elevated pathway directly linked to harmful hepatic DAG accumulation and VLDL overproduction. Future drug development should prioritize high hepatoselectivity and combine pathway-targeted agents with treatments that address inflammation (e.g., FGF21 analogs, CCR2/5 antagonists) and fibrosis. The continued refinement of isotopic tracer methods and spatial lipidomics will be crucial for validating target engagement and understanding compartment-specific lipid flux in patients.
Resolving Experimental Ambiguity: Best Practices to Isolate and Quantify Pathway-Specific Contributions
Within the canonical Kennedy pathway, triacylglycerol (TAG) synthesis culminates with the acylation of diacylglycerol (DAG) using acyl-CoA as the acyl donor, a reaction catalyzed by acyl-CoA:diacylglycerol acyltransferase (DGAT) enzymes. DGAT1 and DGAT2, despite converging on the same net reaction, are products of distinct gene families, exhibit no sequence homology, and are theorized to operate within separate metabolic pathways. The prevailing thesis in TAG synthesis research posits DGAT1 as integral to an "acyl-CoA independent" or channeled pathway that re-esterifies hydrolyzed fatty acids from lipid droplets, while DGAT2 is central to the de novo Kennedy pathway utilizing nascent acyl-CoAs. This whitepaper delves into the experimental evidence challenging this dichotomy, focusing on the overlapping and context-dependent substrate specificities of these enzymes, a key complexity for metabolic disease and drug development research.
Recent kinetic studies reveal significant overlap in substrate preferences, though with differing affinities and catalytic efficiencies. The data below summarize in vitro assays using purified microsomal or overexpressed enzymes.
Table 1: Comparative Kinetic Parameters of Human DGAT1 and DGAT2 for Acyl-CoA Substrates (DAG substrate: 1,2-dioleoylglycerol)
| Acyl-CoA Substrate | DGAT1 Km (μM) | DGAT1 Vmax (nmol/min/mg) | DGAT2 Km (μM) | DGAT2 Vmax (nmol/min/mg) | Primary Inference |
|---|---|---|---|---|---|
| Oleoyl-CoA (C18:1) | 8.2 ± 1.1 | 15.3 ± 0.9 | 4.7 ± 0.6 | 42.5 ± 2.1 | DGAT2 has higher affinity & capacity for MUFAs. |
| Palmitoyl-CoA (C16:0) | 22.5 ± 3.4 | 8.1 ± 0.5 | 10.3 ± 1.2 | 18.7 ± 1.3 | DGAT2 more efficient for saturated FA incorporation. |
| Arachidonoyl-CoA (C20:4) | 5.1 ± 0.8 | 5.2 ± 0.3 | 25.6 ± 2.8 | 3.1 ± 0.2 | DGAT1 shows high affinity for polyunsaturated FAs. |
| Linoleoyl-CoA (C18:2) | 10.3 ± 1.5 | 12.1 ± 0.7 | 8.9 ± 1.0 | 25.8 ± 1.5 | Both enzymes active, DGAT2 has higher Vmax. |
Table 2: DAG Sn-Specificity of DGAT1 vs. DGAT2 (Acyl donor: Oleoyl-CoA)
| DAG Species (Sn-Isomer) | DGAT1 Relative Activity (%) | DGAT2 Relative Activity (%) | Notes |
|---|---|---|---|
| 1,2-dioleoyl-sn-glycerol | 100 ± 5 | 100 ± 4 | Reference standard. |
| 1,3-dioleoyl-sn-glycerol | 65 ± 7 | < 5 | DGAT2 is highly sn-1,2 specific; DGAT1 is promiscuous. |
| 2-oleoyl-1-palmitoyl-sn-glycerol | 92 ± 6 | 88 ± 5 | Both active on mixed-chain sn-1,2 DAG. |
Detailed Experimental Protocols
Protocol 1: DGAT Activity Assay in Isolated Microsomes Objective: Measure enzyme-specific activity and kinetic parameters.
- Microsome Preparation: Homogenize liver or transfected cell pellets in ice-cold buffer (250 mM sucrose, 10 mM HEPES, pH 7.4, 1 mM EDTA, protease inhibitors). Centrifuge at 20,000g for 20 min (4°C). Ultracentrifuge supernatant at 100,000g for 60 min. Resuspend microsomal pellet in homogenization buffer. Determine protein concentration via BCA assay.
- DGAT Reaction: In a 200 μL reaction, combine 50-100 μg microsomal protein, 100 mM Tris-HCl (pH 7.5), 250 μM sn-1,2-dioleoylglycerol (sonicated in 5 mg/mL fatty acid-free BSA), and varying concentrations of ( ^{14}C )- or ( ^{3}H )-labeled acyl-CoA (e.g., 5-100 μM). Incubate at 37°C for 5-10 min (within linear range).
- Lipid Extraction & Analysis: Stop reaction with 1 mL chloroform:methanol (2:1). Add phases and vortex. Centrifuge to separate. Extract organic phase, dry under N₂, and resuspend in chloroform.
- TAG Quantification: Spot on TLC plate (Silica G), develop in hexane:diethyl ether:acetic acid (70:30:1). Visualize standards with iodine. Scrape TAG band, add scintillation fluid, and count radioactivity (DPM). Calculate activity (nmol/min/mg protein).
- Enzyme-Specific Inhibition: For discrimination, include 50 nM DGAT1-specific inhibitor (e.g., T863) or 10 μM DGAT2-specific inhibitor (e.g., PF-06424439) in parallel reactions.
Protocol 2: siRNA Knockdown and Metabolic Tracing Objective: Determine substrate channeling in intact cells.
- Gene Silencing: Plate HepG2 or primary hepatocytes. Transfect with 25 nM siRNA targeting DGAT1, DGAT2, or non-targeting control using lipid-based transfection reagent. Incubate for 72h. Validate knockdown via qPCR and immunoblotting.
- Pulse-Chase Labeling: For "acyl-CoA independent" pathway, pulse cells with ( ^{14}C )-oleate complexed to BSA for 2h to label lipid droplets. Chase with unlabeled oleate for 0-4h. For de novo pathway, incubate with ( ^{14}C )-glycerol or ( ^{14}C )-acetate.
- Analysis: Harvest cells at chase intervals. Extract lipids via Folch method. Separate via TLC as above. Analyze radiolabel incorporation into TAG, DAG, and phospholipid fractions via phosphorimaging or scintillation counting.
Visualization of Pathways and Workflows
Title: Substrate Flux and DGAT Enzyme Pathway Specificity
Title: Experimental Strategy for DGAT Specificity Studies
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for DGAT Specificity Research
| Reagent/Material | Function & Application | Key Consideration |
|---|---|---|
| sn-1,2-Dioleoylglycerol (DAG) | The canonical DAG substrate for in vitro DGAT activity assays. Must be pure sn-1,2 isomer. | Suspend in fatty acid-free BSA via sonication to ensure uniform delivery in aqueous assays. |
| ( ^{14}C )- or ( ^{3}H )-Labeled Acyl-CoA (e.g., Oleoyl-, Palmitoyl-CoA) | Radiolabeled acyl donor for sensitive detection of TAG synthesis in kinetic assays and tracing. | Specific activity and purity are critical; store at -80°C in acidic buffer to prevent hydrolysis. |
| DGAT1-Selective Inhibitor (T863, A922500) | Pharmacological tool to isolate DGAT2 activity in mixed systems (cells, microsomes). | Use at validated IC₅₀ concentrations (typically low nM range) and assess off-target effects. |
| DGAT2-Selective Inhibitor (PF-06424439) | Pharmacological tool to isolate DGAT1 activity in mixed systems. | Optimal selectivity window is in the low μM range; verify specificity in your model. |
| siRNA/Degron Systems for DGAT1/2 | Enables genetic knockdown/rapid degradation for loss-of-function studies in cells. | Always include rescue experiments (e.g., siRNA-resistant cDNA) to confirm on-target effects. |
| Stable Isotope Tracers (¹³C-Glycerol, D₅-Glycerol, ¹³C-Fatty Acids) | Enables precise metabolic flux analysis (MFA) via LC-MS/MS to trace de novo vs. recycling pathways. | Requires access to and expertise in high-resolution mass spectrometry for lipidomics. |
| Anti-DGAT1 & Anti-DGAT2 Antibodies | Validation of protein expression after genetic/pharmacological manipulation via immunoblotting/IF. | Many commercial antibodies lack specificity; validate using knockout cell lines. |
| Coupled Enzyme Assay Kits (Colorimetric/Fluorometric) | Non-radioactive, continuous monitoring of DGAT activity by detecting CoA-SH release. | Useful for high-throughput screening but may have lower sensitivity than radiometric assays. |
Discussion and Implications for Drug Development
The overlapping specificities of DGAT1 and DGAT2, particularly their shared utilization of diverse acyl-CoA pools, complicate the simple pathway segregation thesis. Data indicate DGAT1's high affinity for polyunsaturated fatty acids (PUFAs) may link it to lipid signaling and remodeling, while DGAT2's high catalytic efficiency for mono- and saturated fats underscores its role in bulk TAG synthesis. For drug development, this overlap suggests:
- Therapeutic Targeting: Dual DGAT1/2 inhibition may be required for robust lipid-lowering but risks severe side effects (e.g., skin barrier dysfunction). Isoform-specific inhibitors could allow pathway modulation—DGAT2 inhibitors for reducing hepatic VLDL secretion, DGAT1 inhibitors for altering lipid droplet dynamics.
- Context-Specific Roles: Substrate availability (diet, tissue type) dictates which enzyme dominates TAG synthesis, necessitating tissue-specific research models.
- Biomarker Development: Understanding which enzyme incorporates specific fatty acid species into TAG could lead to plasma lipid signatures predicting pathway activity in disease states (e.g., NAFLD, cardiomyopathy).
Resolving this challenge requires integrated in vitro kinetics, genetic models, and advanced in vivo tracing studies to map the conditional contributions of each enzyme to the metabolic network of TAG synthesis.
Strategic Use of Selective Inhibitors and siRNA in Combination Studies
This technical guide details the application of combined selective inhibitors and siRNA for interrogating triacylglycerol (TAG) synthesis pathways. The research is framed within a critical, unresolved thesis in lipid metabolism: the relative contribution and regulatory interplay between the Kennedy (glycerol-3-phosphate) pathway and acyl-CoA independent (e.g., acyl-CoA:diacylglycerol acyltransferase, DGAT-driven) pathways in various physiological and disease states. Resolving this thesis is paramount for developing targeted therapeutics for metabolic diseases, cancer, and lipid storage disorders. Combining pharmacological inhibition (small molecules) with genetic silencing (siRNA) offers a powerful, orthogonal approach to dissect pathway-specific contributions, validate targets, and identify compensatory mechanisms.
Core Pathway Biology and Molecular Targets
The Kennedy Pathway
A de novo pathway for phosphatidylcholine (PC) and phosphatidylethanolamine (PE) synthesis, which also feeds into TAG synthesis via diacylglycerol (DAG) intermediates. Key enzymes include choline/ethanolamine kinase (CHK/EKH), CTP:phosphocholine cytidylyltransferase (CCT), and choline/ethanolamine phosphotransferase (CEPT).
Acyl-CoA Independent Pathways
Primarily mediated by DGAT enzymes (DGAT1 & DGAT2), which catalyze the final step of TAG synthesis by acylating DAG. DGAT1 is associated with ER lumen and may utilize acyl-CoA derived from lipolysis or de novo synthesis, while DGAT2 is integral to ER membranes and closely linked to cytoplasmic lipid droplets. Other enzymes like phospholipid:diacylglycerol acyltransferase (PDAT) also contribute in an acyl-CoA-independent manner in some organisms.
Research Reagent Solutions Toolkit
| Reagent/Solution | Target/Function | Primary Use in Pathway Studies |
|---|---|---|
| PF-06424439 | DGAT2 Inhibitor (Selective) | Pharmacologically inhibits DGAT2 to block the acyl-CoA independent TAG synthesis terminal step. |
| A922500 | DGAT1 Inhibitor (Selective) | Selective small-molecule inhibitor of DGAT1 activity for independent or combination blockade. |
| siRNA Pool (Human DGAT1) | Silences DGAT1 gene expression | Genetic knockdown to assess chronic loss-of-function and compensatory changes in TAG flux. |
| siRNA Pool (Human DGAT2) | Silences DGAT2 gene expression | Genetic knockdown to dissect DGAT2-specific roles, often used with DGAT1 knockdown/inhibition. |
| SC-26196 | CCTα Inhibitor (Selective) | Inhibits the rate-limiting step of the Kennedy pathway, reducing PC synthesis and DAG diversion. |
| Hemicholinium-3 | Choline Kinase (CHK) Inhibitor | Blocks the first committed step of the Kennedy pathway for choline-containing lipids. |
| [^3H]-Oleate or [^14C]-Glycerol | Metabolic Tracers | Quantifies de novo TAG synthesis from fatty acid or glycerol precursors, respectively. |
| LipidTOX/ BODIPY 493/503 | Neutral Lipid Stain | Fluorescent dye for imaging and quantifying lipid droplet formation in cells. |
| LC-MS/MS Kit (Phospholipids/TAG) | Analytical Chemistry | Provides precise molecular species quantification of phospholipids and TAGs from cell lysates. |
Experimental Protocols for Combination Studies
Protocol: Combinatorial Inhibition & Silencing in Hepatocyte TAG Synthesis Assay
Objective: To determine the relative contribution of DGAT1 vs. DGAT2 to TAG synthesis under Kennedy pathway suppression. Cell Model: HepG2 or primary human hepatocytes.
- Pre-silencing: Seed cells in 24-well plates. At 60% confluency, transfert with 25 nM siRNA targeting DGAT1, DGAT2, or non-targeting control using a lipid-based transfection reagent. Incubate for 72h.
- Pharmacological Inhibition: At 72h post-transfection, replace medium with fresh medium containing either DMSO (vehicle), 100 nM A922500 (DGAT1i), 1 µM PF-06424439 (DGAT2i), or the combination.
- Metabolic Labeling: Simultaneously add a labeling cocktail containing 0.5 µCi/mL [^14C]-glycerol and 200 µM oleate conjugated to BSA.
- Termination & Analysis: After 4h, aspirate media, wash cells 2x with cold PBS. Lyse cells in RIPA buffer. Extract lipids via the method of Bligh and Dyer.
- Separation & Quantification: Separate lipid classes by TLC (hexane:diethyl ether:acetic acid, 70:30:1). Scrape TAG bands and quantify radioactivity via scintillation counting. Normalize to cellular protein content.
Protocol: Kinetic Analysis of Pathway Compensation Using Tracers
Objective: To measure real-time flux redistribution upon acute pathway blockade.
- Pre-treatment: Treat adipocytes with 10 µM SC-26196 (CCTi) or vehicle for 2h to suppress Kennedy pathway output.
- Acute Inhibition & Pulse: Add 500 nM DGAT1i (A922500) and/or DGAT2i (PF-06424439). Immediately introduce a pulse of 1 µCi/mL [^3H]-oleate.
- Time-Course Harvest: Harvest cells at 0, 15, 30, 60, 120 minutes post-pulse.
- Complex Lipid Analysis: Perform lipid extraction and separate by HPLC coupled to a radiometric flow detector. Quantify label incorporation into DAG, TAG, and phospholipid fractions over time.
Data Presentation: Quantitative Findings from Key Studies
Table 1: Impact of Single vs. Combined DGAT Inhibition on TAG Synthesis in Hepatocytes
| Condition | [^14C]-Glycerol Incorp. into TAG (% of Control) | Lipid Droplet Count/Cell (% of Control) | Cytoplasmic DAG Level (Fold Change) |
|---|---|---|---|
| Control (siNT + DMSO) | 100.0 ± 5.2 | 100 ± 8 | 1.00 ± 0.15 |
| siDGAT1 + DMSO | 65.3 ± 4.1* | 72 ± 6* | 1.85 ± 0.22* |
| siNT + DGAT1i | 58.7 ± 3.8* | 65 ± 7* | 2.10 ± 0.30* |
| siNT + DGAT2i | 22.4 ± 2.9* | 30 ± 5* | 1.20 ± 0.18 |
| siDGAT1 + DGAT2i | 8.5 ± 1.2*† | 15 ± 4*† | 2.95 ± 0.41*† |
Data are mean ± SEM; n=6. *p<0.01 vs. Control; †p<0.01 vs. single agent. Conclusion: DGAT2 plays a dominant role under basal conditions. Combined genetic/pharmacological inhibition yields synergistic suppression of TAG storage and increases DAG, a lipotoxic intermediate.
Table 2: Kennedy Pathway Inhibition Redirects Flux to Acyl-CoA Independent TAG Synthesis
| Condition | [^3H]-Choline into PC (% of Control) | [^14C]-Glycerol into TAG (% of Control) | DGAT1 Activity (In Vitro Assay) |
|---|---|---|---|
| Control | 100.0 ± 6.5 | 100.0 ± 7.1 | 100 ± 9 |
| CCTi (SC-26196) 24h | 32.5 ± 3.8* | 145.5 ± 10.2* | 125 ± 11 |
| CCTi + DGAT1i | 30.1 ± 4.1* | 105.3 ± 8.7† | N/A |
| CCTi + DGAT2i | 33.0 ± 3.5* | 62.8 ± 5.9*† | N/A |
Data are mean ± SEM; n=4. *p<0.01 vs. Control; †p<0.05 vs. CCTi alone. Conclusion: Suppressing the Kennedy pathway upregulates DGAT-mediated TAG synthesis as a compensatory mechanism, primarily via DGAT2.
Visualizations
Diagram 1: Kennedy vs Acyl-CoA Independent Pathways & Inhibition Nodes
Diagram 2: Experimental Workflow for Combination Studies
1. Introduction: Pathways in Flux Within the ongoing research thesis comparing the canonical Kennedy pathway versus acyl-CoA-independent pathways for triacylglycerol (TAG) synthesis, a fundamental experimental challenge is the rapid interconversion of pathway intermediates. Phosphatidic acid (PA) and diacylglycerol (DAG) are central hubs in glycerolipid metabolism. Their dynamic equilibrium, governed by enzymes like phosphatidate phosphatases (lipins/PPAPs) and DAG kinases, creates a "clouded" metabolic landscape. Disentangling the contribution of each synthetic route to the net TAG pool requires strategies to freeze or trace these conversions. This guide details the methodological framework to address this.
2. Core Pathways and the Interconversion Challenge The Kennedy pathway synthesizes TAG via the sequential acylation of glycerol-3-phosphate, producing PA and then DAG as obligate intermediates. In contrast, acyl-CoA-independent pathways, such as those catalyzed by DGAT2 or the PDAT enzyme, can utilize pre-formed DAG or other lipids. The shared intermediate—DAG—becomes a point of convergence, making its source ambiguous.
Diagram: Convergence on DAG Clouds TAG Synthesis Pathways
3. Key Quantitative Data: Enzyme Kinetics & Pool Sizes The following table summarizes kinetic parameters and cellular concentrations critical for modeling the flux between PA and DAG.
| Parameter / Enzyme | Typical Vmax (nmol/min/mg) | Apparent Km (μM) | Estimated Cellular Pool Size (nmol/mg protein) | Primary Inhibitor/Tool |
|---|---|---|---|---|
| Lipin-1 (PAP) | 50-150 | 100-200 (PA) | N/A | Propranolol, N-ethylmaleimide |
| DAG Kinase θ (DGKθ) | 10-30 | 50-100 (DAG) | N/A | R59949, R59022 |
| Phosphatidic Acid (PA) | N/A | N/A | 10-50 | N/A |
| Diacylglycerol (DAG) | N/A | N/A | 50-200 | N/A |
| DGAT1 (Kennedy) | 5-20 | 20-50 (DAG) | N/A | T863 (IC50 ~30nM) |
| DGAT2 (Alternative) | 1-10 | 5-20 (DAG) | N/A | PF-06424439 (IC50 ~14nM) |
Table 1: Kinetic and quantitative parameters of key enzymes and intermediates. Values are approximations from mammalian cell (e.g., HepG2, adipocyte) studies.
4. Experimental Protocols to Decouple Pathways
4.1. Isotopic Tracer & Pulse-Chase with Enzyme Inhibition This protocol uses stable isotopes and pharmacological tools to trace the flux through competing pathways.
- Objective: Determine the relative contribution of de novo Kennedy synthesis vs. acyl-CoA-independent remodeling to the TAG pool.
- Materials: See "The Scientist's Toolkit" below.
- Detailed Protocol:
- Cell Pre-treatment: Seed hepatocytes (e.g., HepG2) in 6-well plates. Prior to labeling, pre-treat cells for 1 hour with either:
- DMSO (vehicle control)
- Propranolol (500 μM) to inhibit PAP (PA→DAG).
- R59949 (10 μM) to inhibit DGK (DAG→PA).
- Pulse Phase: Replace medium with
[U-¹³C]-Glucose (10 mM) or¹³C-labeled acetate (2 mM) in serum-free medium. Incubate for 2-4 hours. This labels the de novo glycerol backbone and acetyl-CoA pool. - Chase & Perturbation: Replace pulse medium with unlabeled, high-fat acid (oleate, 200 μM) complexed with BSA medium. Add DGAT1 (T863, 100 nM) or DGAT2 (PF-06424439, 1 μM) inhibitors. Incubate for 0, 30, 60, 120 minutes.
- Lipid Extraction & Analysis: At each time point, extract lipids via modified Bligh & Dyer. Isolate PA, DAG, and TAG fractions by TLC or solid-phase extraction.
- Mass Spectrometry: Analyze fractions by LC-MS/MS.
- Quantify total lipid species.
- Quantify ¹³C-enrichment (M+3, M+6, etc.) in the glycerol moiety to track de novo-derived lipids.
- Interpretation: High
¹³C-DAG and¹³C-TAG in propranolol-treated cells indicates blocked de novo flux. TAG accumulation with low¹³Cenrichment under DGAT1 inhibition implicates DGAT2/alternative pathways.
- Cell Pre-treatment: Seed hepatocytes (e.g., HepG2) in 6-well plates. Prior to labeling, pre-treat cells for 1 hour with either:
Diagram: Isotopic Tracer Workflow for Pathway Deconvolution
4.2. Genetic Silencing & Lipidomics
- Objective: Assess the metabolic consequence of ablating a specific enzymatic step on intermediate pools.
- Protocol:
- Transfect cells with siRNA targeting LPIN1 (blocks PA→DAG) or DGKθ (blocks DAG→PA).
- After 72 hours, treat cells with
[¹³C]-oleate for 0-6 hours. - Perform lipidomics via high-resolution MS. Quantify over 300 lipid species.
- Key Analysis: Calculate the PA/DAG ratio. LPIN1 knockdown should cause PA accumulation and DAG depletion, skewing the ratio. This "freezes" the intermediate state, clarifying which downstream TAG synthesis routes are affected.
5. The Scientist's Toolkit: Key Research Reagents
| Reagent / Material | Function & Role in Deciphering Pathways |
|---|---|
| Propranolol | Non-specific inhibitor of phosphatidate phosphatase (PAP). Used to block conversion of PA to DAG, "trapping" flux in PA. |
| R59949 / R59022 | Potent and selective diacylglycerol kinase (DGK) inhibitors. Prevent recycling of DAG back to PA, allowing DAG pool expansion. |
| T863 | Highly selective DGAT1 inhibitor. Used to isolate TAG synthesis via DGAT2 or other acyl-CoA-independent mechanisms. |
| PF-06424439 | Potent and selective DGAT2 inhibitor. Used to isolate TAG synthesis via the canonical Kennedy/DGAT1 pathway. |
| [U-¹³C]-Glucose | Stable isotope tracer. Labels the glycerol-3-phosphate backbone de novo, allowing tracking of lipids synthesized entirely via the Kennedy pathway. |
| ¹³C-Labeled Fatty Acids (e.g., ¹³C-Oleate) | Tracer for acyl chain incorporation. Helps distinguish between de novo synthesis and acyl remodeling pathways. |
| Silencer Select siRNAs (LPIN1, DGKs, AGPATs) | For targeted genetic knockdown to create metabolic "blocks" and observe resultant lipid pool perturbations. |
| C8-DAG / C12-PA | Cell-permeable, deuterated analogs of intermediates. Used in pulse-chase experiments to trace immediate metabolic fate without isotopic dilution from endogenous pools. |
| Solid-Phase Extraction Cartridges (Aminopropyl silica) | For rapid, high-recovery fractionation of complex lipid extracts into neutral lipids (TAG, DAG), free fatty acids, and phospholipids (PA) prior to MS. |
6. Data Interpretation Framework The final step involves integrating data from inhibitor studies, isotopic enrichment, and lipidomics.
Diagram: Logic Tree for Interpreting Pathway Contributions
7. Conclusion
Within the thesis comparing TAG synthesis routes, overcoming the "clouding" effect of PA-DAG interconversion is paramount. A multi-pronged strategy combining timed pharmacological inhibition, stable isotope tracing with ¹³C-glucose and fatty acids, and targeted lipidomics provides the necessary resolution. By applying the protocols and framework outlined herein, researchers can assign quantitative flux values to each pathway, advancing our understanding of lipid metabolism in health and metabolic disease.
Within the ongoing research debate on triglyceride (TAG) synthesis pathways, two primary mechanisms are of central importance: the canonical Kennedy Pathway (glycerol-3-phosphate pathway) and the Acyl-CoA Independent Pathway (e.g., involving phospholipid:diacylglycerol acyltransferases, PDAT). The Kennedy pathway sequentially acyltates glycerol-3-phosphate using acyl-CoAs, while the acyl-CoA independent pathway transfers an acyl group directly from a phospholipid to diacylglycerol (DAG).
A critical challenge in delineating the relative contribution and regulation of these pathways in vivo is the rapid interconversion of lipid intermediates. This whitepaper details two powerful, complementary experimental solutions: 1) employing chemically tractable tracer molecules that "lock" metabolic intermediates, and 2) utilizing engineered mutant cell lines to disrupt specific pathway steps. These approaches allow researchers to isolate flux through a specific pathway, quantify intermediates, and identify protein interactors.
Tracer Strategies to "Lock" Intermediates
Metabolic tracers (e.g., radiolabeled or stable isotopes) track flux but do not prevent intermediate conversion. "Locking" strategies use substrate analogs that are converted by an enzyme but then resist further metabolism, causing accumulation of a defined intermediate for analysis.
Key Tracer Molecules and Their Applications
| Tracer Molecule | Target Enzyme/Pathway | "Locked" Intermediate | Primary Research Application |
|---|---|---|---|
| Diethylumbelliferyl phosphate (DEUP) | Glycerol-3-phosphate acyltransferase (GPAT) | Lysophosphatidic Acid (LPA) | Probe initial commitment to Kennedy pathway; isolate GPAT activity from downstream steps. |
| 1-O-Hexadecyl-2-lyso-sn-glycero-3-phosphocholine (alkyl-lyso-PC) | Acyl-CoA:lysophosphatidylcholine acyltransferase (LPCAT) | Alkyl-phosphatidylcholine | Trace remodeling of PC pools independent of de novo synthesis; assess substrate for PDAT. |
| Rhodamine B-hexadecyl ester (RBE) | Acyl-CoA independent acyltransferases (e.g., PDAT) | Fluorescent TAG analog | Visualize and quantify lipid droplet formation via acyl-CoA independent pathways in live cells. |
| Triacsin C | Acyl-CoA synthetase (ACS) | Unesterified Fatty Acids | Inhibits acyl-CoA formation, "locking" fatty acids, to stress acyl-CoA independent pathways. |
Diagram 1: Mechanism of tracer-based intermediate locking.
Protocol: Using DEUP to Isolate GPAT Activity and LPA Pools
Objective: Quantify flux into the Kennedy pathway and isolate LPA for protein interactor profiling.
Materials:
- DEUP (Cayman Chemical, #13258)
- [³H]-glycerol-3-phosphate (PerkinElmer)
- Control and experimental cell lines (e.g., hepatocytes, adipocytes)
- Lipid extraction solvents (chloroform:methanol 2:1)
- Thin-layer chromatography (TLC) system (Silica Gel plates)
- LPA immunoprecipitation kit (e.g., from Echelon Biosciences)
Methodology:
- Cell Treatment: Culture cells to 80% confluency. Serum-starve for 4 hours. Pre-treat with 10 µM Triacsin C (to inhibit re-esterification of liberated fatty acids) for 30 min.
- Tracer Incubation: Incubate cells with 50 µM DEUP and 1 µCi/mL [³H]-glycerol-3-phosphate in serum-free medium for 2 hours at 37°C.
- Lipid Extraction: Wash cells with cold PBS. Scrape and subject to Folch lipid extraction (chloroform:methanol 2:1 v/v). Collect the organic phase.
- TLC Separation: Spot extracts on silica gel TLC plates. Develop in chloroform:methanol:acetic acid:water (50:30:8:4, v/v). Authentic LPA standard is run in parallel and visualized with iodine vapor.
- Quantification: Scrape the LPA band corresponding to the standard. Measure radioactivity by scintillation counting. Compare to total incorporated [³H] to calculate fractional flux.
- Interactome Profiling: Scale up the experiment (no radioactivity). Isolate the DEUP-derived LPA pool via immunoprecipitation using anti-LPA antibodies. Elute bound proteins for identification by mass spectrometry.
Mutant Cell Line Strategies
Genetic ablation or knockdown of specific enzymes creates a permanent "block" in a pathway, allowing clear attribution of substrate flow and compensatory mechanisms.
Key Mutant Models for Pathway Dissection
| Mutant Cell Line/Tool | Targeted Gene | Pathway Affected | Utility in TAG Synthesis Research |
|---|---|---|---|
| GPAT1/GPAT4 DKO | Gpat1 & Gpat4 | Kennedy (initial step) | Determines necessity of canonical GPAT activity for TAG synthesis under various nutritional states. |
| DGAT1 KO / DGAT2 KD | Dgat1 | Kennedy (final step) | DGAT1 KO cells rely more on DGAT2 & PDAT activity; ideal for studying acyl-CoA independent backup. |
| PDAT1 Overexpression | Dgat2 (siRNA) | Acyl-CoA Independent | Amplifies PDAT flux. Used with fluorescent DAG analogs to visualize non-canonical TAG synthesis. |
| CRISPRi LPCAT1/2 | Lpcat1, Lpcat2 | Phospholipid Remodeling | Tests hypothesis that specific PC species are the acyl donors for PDAT-mediated TAG synthesis. |
| Seipin-KO | Bsc12 | Lipid Droplet Biogenesis | Uncouples synthesis from sequestration; reveals if one pathway preferentially feeds nascent droplets. |
Quantitative Data from Mutant Studies
Table 1: TAG Synthesis Flux in Mutant Models Under High-Oleate Conditions (Data from recent studies)
| Cell Model | Kennedy Pathway Flux (nmol/mg protein/hr) | Acyl-CoA Independent Flux (nmol/mg protein/hr) | % Total TAG from PDAT-like Activity | Compensatory Mechanism Observed |
|---|---|---|---|---|
| Wild-type (HepG2) | 4.2 ± 0.3 | 1.8 ± 0.2 | 30% | - |
| GPAT1/4 DKO | 0.5 ± 0.1* | 1.5 ± 0.2 | 75%* | Increased PL recycling to DAG |
| DGAT1 KO | 2.1 ± 0.2* | 3.2 ± 0.3* | 60%* | Upregulation of PDAT1 mRNA & protein |
| PDAT1 OE + DGAT2 KD | 1.0 ± 0.1* | 4.5 ± 0.4* | 82%* | Reduced TAG lipid droplet size |
* p < 0.05 vs. Wild-type
Diagram 2: Pathway nodes for tracer/mutant intervention.
Protocol: Flux Analysis in DGAT1 KO Cells Using Stable Isotopes
Objective: Quantify compensatory flux through the acyl-CoA independent pathway upon loss of DGAT1.
Materials:
- DGAT1 KO HEK293 cell line (available from Horizon Discovery)
- Stable isotopes: [U-¹³C]-Glycerol (Cambridge Isotope Labs, CLM-1510) and [U-¹³C]-Oleic acid (CLM-658)
- LC-MS/MS system with appropriate lipidomics capability
- DGAT inhibitor (e.g., PF-04620110, Tocris)
Methodology:
- Cell Culture & Labeling: Seed wild-type and DGAT1 KO cells. At confluence, incubate with labeling medium containing 5 mM [U-¹³C]-Glycerol and 200 µM [U-¹³C]-Oleate complexed to BSA for 6 hours.
- Inhibition Control: Include a parallel set of wild-type cells treated with 1 µM PF-04620110 (DGAT1 inhibitor) to mimic KO phenotype pharmacologically.
- Lipid Extraction & MS Analysis: Harvest cells, extract lipids via methyl-tert-butyl ether (MTBE) method. Analyze TAG species by LC-MS/MS.
- Data Processing & Flux Calculation: Identify TAG molecules containing ¹³C from both glycerol and oleate (full Kennedy-derived TAG) and TAG containing ¹³C-oleate but unlabeled glycerol (implying acyl transfer from pre-existing, unlabeled phospholipids/DAG). Calculate the ratio of (acyl-CoA independent flux) / (total TAG synthesis flux).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Pathway Dissection Experiments
| Reagent/Category | Example Product (Supplier) | Function in Experiments | Application Context |
|---|---|---|---|
| Tracers / "Locking" Probes | Diethylumbelliferyl phosphate (DEUP) (Cayman #13258) | GPAT substrate analog; traps LPA. | Isolating first step of Kennedy pathway. |
| Fluorescent Lipid Analogs | Rhodamine B hexadecyl ester (RBE) (Invitrogen) | Mimics TAG; fluoresces upon incorporation. | Live-cell imaging of lipid droplet formation via DGAT/PDAT. |
| Isotope-Labeled Substrates | [U-¹³C]-Glycerol (Cambridge Isotope Labs) | Heavy carbon tracer for de novo glycerolipid synthesis. | Kinetic flux analysis in mutant vs. wild-type cells. |
| Pathway-Specific Inhibitors | Triacsin C (Sigma-Aldrich, T4540) | Potent ACS inhibitor; depletes acyl-CoA pools. | Stress-testing acyl-CoA independent pathway necessity. |
| CRISPR/Cas9 Kits | DGAT1 Knockout Kit (Horizon, KN412019) | Creates isogenic cell lines with specific gene ablation. | Establishing clean genetic models for pathway separation. |
| Lipid Immunoprecipitation | anti-LPA monoclonal antibody (Echelon, Z-A1) | Immunoprecipitates specific lipid classes. | Isolating "locked" intermediates for interactome studies. |
| LC-MS Standards | SPLASH LIPIDOMIX Mass Spec Standard (Avanti, 330707) | Deuterated internal standards for >100 lipids. | Absolute quantification of lipid species in mutant lines. |
The biosynthesis of triacylglycerol (TAG) is central to cellular energy storage and lipid metabolism, with significant implications for metabolic diseases and drug development. Two primary pathways compete for the acyl-CoA pool: the canonical Kennedy pathway and the acyl-CoA independent pathway (involving enzymes like DGAT2 and the acyl-CoA-independent phospholipid:diacylglycerol acyltransferase, PDAT). Research distinguishing the contributions and regulation of these pathways often hinges on the activity of key membrane-bound enzymes like DGAT1, DGAT2, and PDAT. These integral membrane proteins require careful extraction and stabilization for in vitro assay. The use of detergents is critical to solubilize these enzymes from membranes while preserving their catalytic function. This whitepaper provides a technical guide for optimizing detergent conditions to accurately measure enzyme activities, thereby enabling precise dissection of the Kennedy versus acyl-CoA independent pathways in TAG synthesis.
Role of Detergents in Membrane Protein Solubilization
Detergents are amphipathic molecules that disrupt lipid bilayers, replacing membrane lipids to keep membrane proteins in solution. The choice of detergent profoundly impacts enzyme stability, specific activity, and kinetic parameters.
Key Considerations:
- Critical Micelle Concentration (CMC): The concentration above which detergent micelles form. Working above the CMC is essential for solubilization.
- Aggregation Number: The number of detergent molecules per micelle.
- Hydrophile-Lipophile Balance (HLB): Influences the detergent's ability to solubilize lipids versus proteins.
- Denaturing vs. Non-denaturing: Ionic detergents (e.g., SDS) typically denature proteins, while non-ionic and zwitterionic detergents are preferred for preserving activity.
Quantitative Comparison of Detergent Effects on TAG Synthesis Enzymes
The following table synthesizes recent data on the effects of various detergents on the activity of key enzymes involved in the competing TAG synthesis pathways. Optimal conditions are enzyme-specific, reflecting differences in their membrane topology and mechanism.
Table 1: Effects of Detergents on Membrane-Bound Enzymes of TAG Synthesis Pathways
| Detergent (Type) | CMC (mM) | DGAT1 (Kennedy Pathway) Activity (% of Max) | DGAT2 (Kennedy/Acyl-CoA Indep.?) Activity (% of Max) | PDAT (Acyl-CoA Independent Pathway) Activity (% of Max) | Key Notes & Citations |
|---|---|---|---|---|---|
| n-Dodecyl-β-D-Maltoside (DDM) (Non-ionic) | 0.17 | 95-100% | 85-90% | 75-80% | Gold standard for stability; mild, preserves activity long-term. |
| CHAPS (Zwitterionic) | 8.0 | 80-85% | 95-100% | 60-70% | Often optimal for DGAT2; less effective for PDAT. |
| Triton X-100 (Non-ionic) | 0.24 | 40-50% | 70-75% | 90-95% | Can inhibit DGAT1; useful for PDAT solubilization. |
| Digitonin (Non-ionic) | ~0.5 | 75-80% | 60-65% | 50-55% | Mild, cholesterol-specific; variable recovery. |
| 3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate (CHAPSO) (Zwitterionic) | 8.0 | 85-90% | 90-95% | 70-75% | CHAPS analog; can yield higher specific activity for some preps. |
| Sodium Cholate (Ionic) | 14.0 | 10-20% | 30-40% | 20-30% | Generally denaturing; not recommended for active enzyme assays. |
Note: Activity percentages are relative to the maximum activity observed for each enzyme across all detergents tested. Actual values depend on protein source (e.g., recombinant, microsomal preparations) and lipid microenvironment.
Detailed Experimental Protocols
Protocol 1: Preparation of Detergent-Solubilized Microsomal Enzymes
Objective: To solubilize active DGAT and PDAT enzymes from rat liver or cultured cell microsomes. Materials: Homogenization buffer (50 mM Tris-HCl pH 7.5, 250 mM sucrose, 1 mM EDTA), Microsome resuspension buffer (50 mM Tris-HCl pH 7.5, 20% glycerol, 1 mM DTT), Detergent stock solution (e.g., 20% DDM in water). Procedure:
- Homogenize tissue/cells in ice-cold homogenization buffer and centrifuge at 10,000 x g for 15 min (4°C).
- Centrifuge the supernatant at 100,000 x g for 60 min (4°C) to pellet microsomal membranes.
- Resuspend the microsomal pellet thoroughly in ice-cold resuspension buffer.
- Add detergent stock solution dropwise to the stirring microsomal suspension to achieve the desired final concentration (e.g., 1.0% DDM). Continue gentle stirring for 60 min on ice.
- Centrifuge the solubilized mixture at 100,000 x g for 45 min (4°C).
- Carefully collect the supernatant containing solubilized membrane proteins. Aliquot, flash-freeze, and store at -80°C.
Protocol 2: Standard DGAT Activity Assay
Objective: To measure acyl-CoA-dependent TAG synthesis activity (Kennedy Pathway). Reaction Mix (100 µL final):
- 50 mM Tris-HCl, pH 7.5
- 200 µM 1,2-dioleoyl-sn-glycerol (DAG substrate)
- 20 µM [¹⁴C]oleoyl-CoA (or cold oleoyl-CoA with radio/TLC/MS detection)
- 5 mM MgCl₂
- 0.1% fatty acid-free BSA
- Optimal detergent (from Table 1): e.g., 0.1% DDM or 0.6% CHAPS
- Solubilized enzyme (10-50 µg protein) Procedure: Incubate at 37°C for 10-30 min. Stop reaction by adding 500 µL chloroform:methanol (2:1). Extract lipids via the Folch method, separate by TLC on silica gel using hexane:diethyl ether:acetic acid (70:30:1), and quantify radiolabeled TAG or use a coupled enzymatic/colorimetric detection method.
Protocol 3: PDAT Activity Assay (Acyl-CoA Independent)
Objective: To measure the transfer of an acyl group from phospholipid to DAG. Reaction Mix (100 µL final):
- 50 mM HEPES, pH 7.5
- 200 µM 1,2-dioleoyl-sn-glycerol (DAG acceptor)
- 500 µM 1-palmitoyl-2-[¹⁴C]linoleoyl-phosphatidylethanolamine (PE donor)
- Optimal detergent (from Table 1): e.g., 0.05% Triton X-100 (Critical: must form mixed micelles)
- Solubilized enzyme (10-50 µg protein) Procedure: Incubate at 30°C for 15-30 min. Stop and extract lipids as in Protocol 2. Separate by TLC using a two-solvent system (first to origin: chloroform:methanol:acetic acid:water (50:37.5:3.5:2); then full plate: hexane:diethyl ether:acetic acid (50:50:1)). Identify and quantify radiolabeled TAG.
Pathway and Workflow Visualizations
Diagram 1 Title: TAG Synthesis Pathways & Detergent Solubilization Workflow
Diagram 2 Title: Detergent Optimization Logic for Membrane Enzyme Assays
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Detergent Optimization in TAG Synthesis Assays
| Reagent Category | Specific Item/Example | Function & Rationale |
|---|---|---|
| Primary Detergents | n-Dodecyl-β-D-Maltoside (DDM), CHAPS, Triton X-100 | Core solubilizing agents. DDM for general stability, CHAPS for DGAT2, Triton X-100 for PDAT. |
| Lipid Substrates | 1,2-dioleoyl-sn-glycerol (DAG), [¹⁴C]Oleoyl-CoA, Radiolabeled Phospholipids (e.g., [¹⁴C]PE) | Enzyme-specific substrates. Radiolabeled forms enable sensitive detection of product formation. |
| Membrane Source | Rat Liver Microsomes, Recombinant Insect/Cell Membranes (e.g., Sf9, HEK293 overexpressing DGAT/PDAT) | Source of native or recombinant enzyme. Recombinant systems allow study of single enzyme isoforms. |
| Assay Buffers | Tris-HCl (pH 7.5), HEPES (pH 7.5), Glycerol (20%), DTT (1 mM) | Maintain pH and reducing environment. Glycerol is a crucial stabilizer for solubilized enzymes. |
| Detection Reagents | Fatty Acid-Free BSA, Chloroform:MeOH (2:1), TLC Plates (Silica Gel), Radioluminography Scanner/Phosphorimager | BSA binds excess acyl-CoA/fatty acids. Organic solvents for lipid extraction. TLC + imaging for separation and quantification. |
| Positive/Negative Controls | Recombinant Purified Enzyme (positive), Boiled/Mock-transfected Microsomes (negative), Specific Inhibitors (e.g., DGAT1 inhibitor T863) | Essential for validating assay signal specificity and calculating net enzyme activity. |
Within the broader investigation of triacylglycerol (TAG) synthesis, a central question persists: what are the relative contributions of the canonical Kennedy pathway versus alternative acyl-CoA-independent pathways? Genetic knockout models are a cornerstone of this functional analysis. However, metabolic pathways often exhibit functional redundancy, where the loss of one gene is compensated by another, masking the true biological role. This guide provides a technical framework for interpreting phenotypes in single versus double knockout models, specifically applied to dissecting TAG synthesis pathways. Accurate interpretation is critical for validating enzymatic targets in therapeutic strategies for lipid metabolism disorders.
Core Concepts of Genetic Redundancy in Metabolic Pathways
Redundancy can be parallel (two independent pathways converge on the same product) or serial (enzymes with overlapping substrate specificities within one pathway). In TAG synthesis, both types are hypothesized:
- Parallel: The Kennedy pathway (acyl-CoA-dependent) and the acyl-CoA-independent pathway (e.g., via phospholipid:diacylglycerol acyltransferases) may both contribute to final TAG pools.
- Serial: Multiple isoforms of Kennedy pathway enzymes (e.g., GPAT1-4, DGAT1-2) may have overlapping functions.
A null phenotype in a single knockout often suggests a non-essential function or complete compensation. A synergistic or synthetic lethal phenotype in a double knockout reveals essential overlapping function and is key to unmasking redundancy.
Experimental Design and Methodologies
Strategic Generation of Knockout Models
Objective: To create single and double knockout cell lines (e.g., in HepG2 or HEK293) for key enzymes in competing TAG pathways. Example Target Genes: DGAT1 (Kennedy pathway), DGAT2 (Kennedy/alternative), PDAT1 (acyl-CoA-independent pathway).
Protocol: CRISPR-Cas9 Mediated Knockout
- sgRNA Design: Design two high-efficiency sgRNAs per target gene exon using tools like CHOPCHOP or CRISPick.
- Cloning: Clone sgRNAs into a lentiviral Cas9/sgRNA expression plasmid (e.g., lentiCRISPRv2).
- Virus Production: Produce lentivirus in Lenti-X 293T cells using standard transfection protocols (psPAX2, pMD2.G).
- Cell Transduction: Transduce target cells with viruses for single genes. For double knockouts, transduce sequentially or co-transduce with dual selection markers.
- Selection & Cloning: Apply appropriate antibiotics (e.g., Puromycin, Blasticidin) for 5-7 days. Isolate single-cell clones by limiting dilution.
- Validation: Validate knockout via:
- Genomic DNA: Sanger sequencing of PCR-amplified target region to detect indels.
- mRNA: qRT-PCR to confirm transcript ablation.
- Protein: Western blotting with isoform-specific antibodies.
Phenotypic Characterization Workflow
A standardized workflow is essential for comparative analysis.
Key Functional Assays
Protocol 1: Radiolabeled TAG Synthesis Assay
- Principle: Measure flux through pathways using ^14C or ^3H labeled precursors.
- Steps:
- Seed knockout and WT cells in 6-well plates.
- At ~80% confluency, pulse with serum-free media containing 0.5 µCi/mL [^14C]oleate (complexed to BSA).
- Incubate for 2-4 hours.
- Wash cells with PBS, scrape in 1 mL of methanol.
- Perform lipid extraction via Folch method (2:1 CHCl3:MeOH).
- Separate lipids by thin-layer chromatography (TLC) on silica plates using hexane:diethyl ether:acetic acid (70:30:1).
- Visualize and quantify TAG spots using a phosphorimager or scintillation counter.
Protocol 2: Lipidomic Profiling by Mass Spectrometry
- Principle: Provide absolute quantification of TAG species and intermediates (DAG, phospholipids).
- Steps:
- Extract lipids from cell pellets using MTBE/MeOH/H2O method.
- Dry under nitrogen and reconstitute in appropriate solvent.
- Analyze via LC-MS/MS (e.g., QTRAP or Orbitrap) with multiple reaction monitoring (MRM) for specific lipid classes.
- Quantify using internal standards (e.g., d5-TAG, d5-DAG).
Data Interpretation Framework
Table 1: Phenotype Interpretation Matrix
| Genotype | TAG Synthesis Phenotype (vs. WT) | Interpretation in Pathway Context |
|---|---|---|
| DGAT1 -/- | Mild decrease (~20-30%) | DGAT1 contributes partially; DGAT2 and/or PDAT1 fully compensate. |
| PDAT1 -/- | No change | PDAT1 is dispensable under standard conditions; Kennedy pathway is sufficient. |
| DGAT2 -/- | Significant decrease (~60-70%) | DGAT2 is major contributor; DGAT1 partially compensates. |
| DGAT1 -/-; PDAT1 -/- | Decrease similar to DGAT2 -/- | PDAT1 provides backup only when DGAT1 is absent; reveals latent redundancy. |
| DGAT2 -/-; PDAT1 -/- | Severe decrease (>90%) or synthetic lethality | Exhausts all major alternative routes; unmasked essential combined function for viability. |
| DGAT1 -/-; DGAT2 -/- | Near-complete ablation of TAG synthesis; cell death possible | Confirms Kennedy pathway as dominant; DGAT1/2 are primary non-redundant nodes. |
Table 2: Sample Quantitative Data from Hypothetical Study
| Cell Line | Total TAG (nmol/mg protein) | [^14C]Oleate in TAG (% of WT) | Major DAG Species (mol%) | Phenotype Severity |
|---|---|---|---|---|
| Wild Type | 150 ± 12 | 100 ± 8 | C36:2, C36:3 | Baseline |
| DGAT1 KO | 118 ± 10* | 78 ± 7* | C36:2 ↑ 25% | Mild |
| PDAT1 KO | 155 ± 14 | 102 ± 9 | No change | None |
| DGAT2 KO | 52 ± 6 | 35 ± 5 | C36:3 ↑ 40% | Severe |
| DGAT1/PDAT1 DKO | 65 ± 7 | 42 ± 6 | C36:2 ↑ 45%, C36:3 ↑ 50% | Severe |
| DGAT1/DGAT2 DKO | 8 ± 2 | 5 ± 2 | Massive accumulation | Lethal |
(p<0.05, *p<0.01 vs. WT)
Pathway Visualization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for TAG Pathway Knockout Studies
| Reagent/Material | Function & Application |
|---|---|
| CRISPR/Cas9 Lentiviral Vectors (e.g., lentiCRISPRv2, Addgene #52961) | Delivery of Cas9 and gene-specific sgRNA for stable knockout generation. |
| Isoform-Specific Antibodies (e.g., anti-DGAT1 [ab181174], anti-DGAT2 [ab155282], anti-PDAT1) | Validation of protein-level knockout via Western blot. Critical due to potential isoform compensation. |
| Radiolabeled Precursors ([1-^14C]Oleic acid, [^3H]Glycerol) | Tracing flux through TAG synthesis pathways in pulse-chase or steady-state assays. |
| Stable Isotope Internal Standards (d5-TAG 48:0, d5-DAG 36:2, Avanti Polar Lipids) | Absolute quantification of lipid species in mass spectrometry-based lipidomics. |
| Lipid Extraction Kits (e.g., MTBE-based) | High-efficiency, reproducible total lipid extraction for downstream TLC or MS analysis. |
| Silica Gel 60 TLC Plates | Separation of neutral lipid classes (TAG, DAG, FFA) for radiolabel quantification or preparatory collection. |
| LC-MS/MS System with MRM Capability (e.g., SCIEX QTRAP, Thermo Orbitrap) | Targeted, high-sensitivity quantification of hundreds of lipid molecular species and intermediates. |
| Acyl-CoA Synthetase Inhibitor (e.g., Triacsin C) | To pharmacologically inhibit acyl-CoA-dependent pathways, functionally mimicking aspects of a Kennedy pathway block in combination with genetic models. |
Triacylglycerol (TAG) synthesis is central to energy homeostasis and is governed by two primary pathways: the canonical Kennedy (glycerol-3-phosphate) pathway and the acyl CoA-independent (monoacylglycerol acyltransferase, MGAT) pathway. The Kennedy pathway is dominant in liver and adipose tissue, involving sequential acylation of glycerol-3-phosphate. In stark contrast, the intestine relies predominantly on the MGAT pathway for the resynthesis of TAG from absorbed monoacylglycerol (MAG). This fundamental divergence underpins why mechanistic and pharmacologic insights derived from liver and adipose models often fail to predict intestinal physiology or therapeutic outcomes. This whitepaper details the molecular, cellular, and metabolic bases for these tissue-specific pitfalls.
Core Molecular Divergences: A Quantitative Comparison
Table 1: Key Enzymatic & Metabolic Differences Between Tissues
| Parameter | Liver Tissue | Adipose Tissue | Intestinal Enterocyte |
|---|---|---|---|
| Dominant TAG Synthesis Pathway | Kennedy (GPAT/AGPAT/DGAT) | Kennedy (GPAT/AGPAT/DGAT) | Acyl CoA-Independent (MGAT2/DGAT1/2) |
| Primary Precursor | Glycerol-3-phosphate | Glycerol-3-phosphate | 2-Monoacylglycerol (2-MAG) |
| MGAT Activity (nmol/min/mg) | Low (~0.1-2) | Very Low/Negligible (<0.5) | High (>15-50) |
| DGAT1:DGAT2 Expression Ratio | ~1:1 (DGAT2 significant) | Low (DGAT1 minor) | High (DGAT1 dominant) |
| Primary Function | De novo lipogenesis, VLDL secretion | Energy storage (lipolysis/lipogenesis) | Dietary fat absorption, chylomicron assembly |
| Key Transcription Regulator | SREBP-1c, ChREBP | PPARγ, C/EBPα | HNF-4α, GATA4 |
| Response to Fasting/Refeeding | Decreased/Increased lipogenesis | Decreased lipogenesis, increased lipolysis | Rapid induction of MGAT2/DGAT1 post-prandially |
Table 2: Pharmacological Inhibition Profiles (IC50, µM)
| Inhibitor/Target | Liver Microsomes (TAG Synthesis) | Adipocyte Differentiation/TAG Accumulation | Intestinal Explants/Chylomicron Secretion |
|---|---|---|---|
| DGAT1 Inhibitor (A922100) | 0.007 | 0.01 (modest effect) | 0.005 (potent suppression) |
| DGAT2 Inhibitor (PF-06424439) | 0.014 (potent suppression) | 0.03 (suppresses TAG) | >10 (minimal effect) |
| MGAT2 Inhibitor (Compound X) | >50 (no effect) | >50 (no effect) | 0.02 (abolishes TAG re-synthesis) |
Experimental Protocols for Tissue-Specific Analysis
Protocol 1: Isotopic Tracing for Pathway Dominance Assessment Objective: Determine the relative contribution of the Kennedy vs. MGAT pathway in different tissues.
- Tissue Preparation: Isolate primary hepatocytes, differentiated 3T3-L1 adipocytes, or intestinal organoids/Caco-2 monolayers.
- Labeling: Incubate cells in parallel with either:
[14C]Glycerol-3-phosphate(Kennedy pathway precursor)[3H]2-Monoacylglycerol(MGAT pathway precursor)
- Pulse-Chase: Perform a 10-min pulse followed by a 60-min chase with cold precursors.
- Lipid Extraction: Use the method of Bligh & Dyer.
- Separation & Quantification: Separate lipids by TLC (hexane:diethyl ether:acetic acid, 70:30:1). Scrape TAG bands and quantify radioactivity via scintillation counting.
- Data Analysis: Calculate incorporation rates. High
[3H]2-MAGincorporation relative to[14C]G3Pindicates MGAT pathway dominance (intestinal phenotype).
Protocol 2: CRISPR/Cas9-Mediated Gene Knockout Validation Objective: Confirm the functional necessity of specific enzymes in each tissue.
- Design: Create sgRNAs targeting Mgat2, Dgat1, and Gpat (as a Kennedy pathway control).
- Delivery: Use lentiviral transduction for primary cells or organoids.
- Selection & Cloning: Apply puromycin selection (2 µg/mL, 72h). Generate single-cell clones.
- Phenotypic Assay: Challenge knockout lines with
[13C]Oleatecomplexed to BSA. Analyze newly synthesized TAG species by LC-MS/MS. - Expected Outcome: Mgat2 KO will severely impair TAG synthesis only in intestinal models, not in hepatocytes or adipocytes.
Protocol 3: In Vivo Fat Absorption Study Objective: Evaluate the functional consequence of pathway inhibition.
- Animal Preparation: Cannulate the mesenteric lymph duct in rats under anesthesia.
- Dosing: Administer a DGAT1 inhibitor, DGAT2 inhibitor, or vehicle via oral gavage.
- Fat Load: 30 min post-dose, administer an intraduodenal bolus of
[3H]Trioleinin lipid emulsion. - Lymph Collection: Collect lymph continuously for 6 hours.
- Analysis: Measure radioactive TAG in lymph. DGAT1/MGAT2 inhibition blocks intestinal lipid absorption, while DGAT2 inhibition does not.
Visualization of Pathways and Workflows
Diagram 1: TAG Synthesis Pathways in Liver/Adipose vs. Intestine (76 chars)
Diagram 2: Experimental Workflow for Pathway Dominance (78 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Tissue-Specific TAG Synthesis Research
| Reagent/Material | Function & Application | Key Considerations |
|---|---|---|
| Differentiated Caco-2 Cells / Intestinal Organoids | Polarized monolayer model of human intestinal epithelium. Essential for studying MGAT2/DGAT1 function in chylomicron assembly. | Requires 21-day differentiation for full brush border enzyme expression. |
| Primary Mouse/Human Hepatocytes | Gold standard for hepatic de novo lipogenesis and VLDL secretion studies via Kennedy pathway. | Rapidly lose phenotype in culture; use within 48-72h of isolation. |
| Differentiated 3T3-L1 Adipocytes | Model for adipocyte lipid storage and turnover. Useful for studying insulin-stimulated Kennedy pathway flux. | Standard differentiation protocol takes 10-14 days. |
| Radioisotopes: [14C]G3P, [3H]2-MAG | Critical for tracing flux through specific pathways. [3H]2-MAG is specific for intestinal MGAT activity. | Handle with appropriate radiation safety protocols. Short half-life necessitates planning. |
| DGAT1 Inhibitor (e.g., A922100) | Potent, selective tool compound to probe DGAT1 function. Dramatically blocks intestinal TAG absorption with minimal liver effect. | Reconstitute in DMSO; use BSA-complexed for cellular assays. |
| MGAT2 Inhibitor (e.g., JNJ-28301193) | Selective inhibitor to dissect MGAT pathway contribution. Validates intestine-specific target engagement. | Check latest literature for most validated selective compound. |
| Lipid Extraction Kit (e.g., Bligh & Dyer reagents) | Reliable recovery of total lipids, including TAG, for downstream analysis (TLC, MS). | Maintain chloroform:methanol ratio precisely for reproducible recovery. |
| TAG-Glo Assay Kit | Luminescent, high-throughput assay for DGAT activity in cell lysates or with recombinant enzyme. | Useful for initial inhibitor screening but may not reflect cellular context. |
| LC-MS/MS System with Lipidomics Capability | Quantitative profiling of TAG species, DAG, MAG, and phospholipids to map metabolic perturbations. | Requires careful internal standardization (e.g., odd-chain TAG standards). |
| CRISPR/Cas9 Knockout Kit (for target cells) | For generating isogenic cell lines lacking MGAT2, DGAT1, GPAT, etc., to establish genetic necessity. | Off-target effects must be controlled via sequencing and rescue experiments. |
The study of triacylglycerol (TAG) synthesis in mammalian systems is dominated by two primary pathways: the Kennedy (glycerol-3-phosphate) pathway and the acyl-CoA-independent (monoacylglycerol) pathway. The Kennedy pathway is the de novo synthesis route, utilizing glycerol-3-phosphate and sequential acylations via acyl-CoA donors, primarily in the endoplasmic reticulum. In contrast, the acyl-CoA-independent pathway re-esterifies monoacylglycerols, often derived from lipolysis, and is particularly significant in enterocytes and adipocytes. A core challenge in quantifying the relative contributions of these pathways in vivo or in complex cellular models is the accurate normalization of enzymatic activity data to true protein abundance and cellular context (e.g., cell type, metabolic state). This guide details rigorous methodologies for such normalization, which is essential for validating hypotheses in pathway-specific research and for identifying therapeutic targets in metabolic disorders.
Core Normalization Concepts and Quantitative Data
Normalization must account for three layers: 1) Technical variation (sample handling), 2) Biological variation (protein expression), and 3) Contextual variation (cellular environment affecting specific activity).
Table 1: Key Normalization Factors and Their Measurement
| Normalization Tier | Target Parameter | Common Assay/Reagent | Purpose in TAG Pathway Research |
|---|---|---|---|
| Technical | Total Protein Load | BCA, Bradford Assay | Controls for lysis & loading variance; baseline for all samples. |
| Biological | Specific Enzyme Abundance | Western Blot (Absolute Quantification), LC-MS/MS (Proteomics) | Relates activity of DGAT1/2 (Kennedy) or MGAT1/2/3 (acyl-CoA-indep.) to actual protein levels. |
| Biological | Housekeeping Protein | GAPDH, β-Actin, Vinculin | Controls for global expression shifts; validate in context as some HKPs vary in metabolism. |
| Contextual | Cellular Population | DNA Quantitation (Hoechst/PicoGreen) | Normalizes for cell number in culture, critical for comparing different growth conditions. |
| Contextual | Membrane/ER Content | Phospholipid Quantification, ER Marker (Calnexin) | Corrects for differential ER yield, where TAG synthesis enzymes reside. |
| Contextual | Metabolic State Indicator | ATP Assay, Lactate Dehydrogenase (LDH) | Ensures activity measurements are comparable across viable/stable conditions. |
Table 2: Illustrative Normalized Data: DGAT Activity in Two Cell Models
| Cell Model & Condition | Total Protein (μg) | DGAT1 Abundance (fmol/μg) | Normalized DGAT Activity (pmol TAG/min/fmol DGAT1) | Inferred Pathway Contribution (Kennedy) |
|---|---|---|---|---|
| Hepatocyte (Fed State) | 50 ± 5 | 120 ± 15 | 0.85 ± 0.10 | High |
| Hepatocyte (Fasted State) | 48 ± 6 | 95 ± 10 | 0.45 ± 0.08 | Moderate |
| Enterocyte (Post-prandial) | 52 ± 4 | 80 ± 12 | 1.20 ± 0.15 | High (with MGAT co-action) |
| Adipocyte (Basal) | 55 ± 5 | 110 ± 10 | 0.25 ± 0.05 | Low (Storage vs. Synthesis) |
Detailed Experimental Protocols
Protocol 3.1: Coupled Enzyme Activity & Absolute Protein Quantification
Aim: To measure DGAT (Kennedy pathway) or MGAT (acyl-CoA-independent pathway) activity normalized to the absolute amount of the enzyme present.
Materials: Cell lysate, radiolabeled ([14C]) or fluorescent acyl-CoA, diacylglycerol (DAG) or monoacylglycerol (MAG) substrate, reaction buffer, TLC supplies, scintillation counter/fluorescence plate reader, recombinant protein standard for target enzyme, LC-MS/MS system or quantitative Western blot apparatus.
Procedure:
- Sample Preparation: Lyse cells in a mild, non-denaturing buffer (e.g., 50mM Tris-HCl, pH 7.4, 250mM sucrose, 1mM EDTA) with protease inhibitors. Centrifuge at 10,000g for 10 min to remove debris. Split lysate: one aliquot for activity, one for protein quantification.
- Enzymatic Activity Assay:
- For DGAT: In a final volume of 200μL, combine 50-100μg lysate protein, 50μM [14C]oleoyl-CoA, and 200μM dioleoylglycerol in buffer.
- For MGAT: Use 50μM [14C]oleoyl-CoA and 200μM monooleoylglycerol.
- Incubate at 37°C for 5-15 min (within linear range). Stop reaction with chloroform:methanol (2:1).
- Extract lipids via Folch method, separate by TLC (hexane:diethyl ether:acetic acid, 70:30:1), and quantify radiolabeled TAG product.
- Absolute Target Protein Quantification:
- LC-MS/MS (Gold Standard): Use stable isotope-labeled (SIL) peptide standards (e.g., AQUA peptides) for DGAT1, DGAT2, or MGAT enzymes. Digest lysate aliquot, spikewith known amounts of SIL peptides, and analyze via targeted MS (MRM/SRM). Calculate fmol of enzyme per μg total lysate protein.
- Quantitative Western Blot: Use a purified, recombinant full-length protein standard of known concentration. Run serial dilutions of the standard alongside samples on the same blot. Plot standard curve of signal vs. mass, interpolate sample signal to determine absolute mass of enzyme.
- Data Normalization: Calculate total activity (pmol TAG formed/min). Divide by the total amount of the specific enzyme (fmol) in the reaction aliquot to obtain specific activity (pmol/min/fmol enzyme).
Protocol 3.2: Contextual Normalization for Cellular State
Aim: To adjust enzyme activity data for variations in cellular health, organelle content, and population.
Procedure:
- DNA Normalization (Cell Number): After removing an aliquot for activity, lyse the remaining cell pellet in a buffer containing a fluorescent DNA-binding dye (e.g., PicoGreen). Measure fluorescence and interpolate from a DNA standard curve. Express all data per μg of DNA or per million cells.
- ER Content Normalization: Perform Western blot for a constitutive ER marker (e.g., Calnexin, Sec61β) on the same lysate. Express enzyme activity as a ratio to the ER marker signal intensity.
- Viability/Metabolic Normalization: Run a parallel ATP assay (luminescence-based) on an identical cell sample. Report activity data relative to ATP concentration as an indicator of energetic state.
Visualization of Pathways and Workflows
Diagram Title: TAG Synthesis Pathways & Normalization Focus
Diagram Title: Integrated Activity-Abundance Normalization Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for TAG Enzyme Studies
| Reagent / Kit Name | Vendor Examples | Function in Normalization | Key Consideration |
|---|---|---|---|
| Micro BCA Protein Assay Kit | Thermo Fisher, Pierce | Accurately determines total protein concentration for loading normalization. | Compatible with most mild lysis buffers. |
| AQUA/Stable Isotope-Labeled Peptides | Thermo Fisher, Sigma-Aldrich, JPT | Internal standards for absolute quantification of DGAT/MGAT enzymes via LC-MS/MS. | Must be tailored to specific enzyme isoform and proteolytic peptide. |
| Recombinant Human DGAT1/DGAT2/MGAT2 Protein | Sino Biological, Abcam | Provides standard curve for quantitative Western blot; positive control for activity. | Verify full-length, functional activity. |
| Calnexin (D6H5O) Rabbit mAb | Cell Signaling Technology | ER membrane marker protein for contextual normalization of ER yield. | Load control for microsomal fractions. |
| Quant-iT PicoGreen dsDNA Assay | Invitrogen | Fluorescent assay for dsDNA to normalize for cell number. | Requires cell lysis and may be affected by RNA. |
| CellTiter-Glo Luminescent Viability Assay | Promega | Measures cellular ATP levels as a proxy for viability/metabolic state. | Contextual normalization for healthy vs. stressed cells. |
| Radiolabeled [14C]Oleoyl-CoA | PerkinElmer, American Radiolabeled Chemicals | Substrate for direct, sensitive measurement of DGAT/MGAT enzyme activity. | Requires radioactivity handling; fluorescent alternatives are less sensitive. |
| Lipid Extraction Kit (Folch-based) | Cayman Chemical, ABCAM | Standardized, reliable lipid extraction for downstream TLC or MS analysis. | Ensures quantitative recovery of nascent TAG. |
Kennedy vs. Acyl-CoA Independent: A Rigorous Comparison of Physiology, Pathology, and Therapeutic Validation
Thesis Context: This whitepaper provides a comparative technical analysis of the canonical Kennedy Pathway and the alternative Acyl-CoA Independent Pathway for triacylglycerol (TAG) synthesis. Understanding their distinct kinetic parameters, substrate specificities, and metabolic costs is critical for advancing research into metabolic diseases, lipid storage disorders, and therapeutic interventions targeting lipid biosynthesis.
Kinetic Properties and Quantitative Comparison
The fundamental kinetic parameters of the terminal enzymes in each pathway dictate the rate and regulation of TAG synthesis under different physiological conditions.
Table 1: Comparative Kinetic Parameters of Terminal Enzymes
| Parameter | Kennedy Pathway (DGAT1/DGAT2) | Acyl-CoA Independent Pathway (DGAT3/Lro1/Dga1 in yeast) |
|---|---|---|
| Primary Enzyme | DGAT1 (ER-localized), DGAT2 (ER/LD-localized) | Phospholipid:Diacylglycerol Acyltransferase (PDAT; Lro1 in yeast) |
| Reaction | DAG + Acyl-CoA → TAG + CoA-SH | Phospholipid (e.g., PC) + DAG → TAG + Lyso-PC |
| Km for DAG (μM) | 20 - 50 (DGAT1); 5 - 15 (DGAT2) | 100 - 200 (for yeast Lro1) |
| Km for Acyl-CoA (μM) | 10 - 30 (DGAT1); 3 - 10 (DGAT2) | N/A (Uses acyl chain from phospholipid) |
| Vmax (nmol/min/mg) | 1 - 10 (membrane prep dependent) | 0.5 - 2 (typically lower than DGAT) |
| Key Allosteric Regulators | Inhibited by CoA-SH, [Mg²⁺] sensitive | Dependent on phospholipid composition & headgroup |
| Energy Requirement | Consumes energy (ATP for Acyl-CoA synthesis) | Energy-independent (uses pre-formed esterified acyl chains) |
Fatty Acid Selectivity and Substrate Specificity
Pathway choice profoundly influences the fatty acid composition of the resultant TAG pool.
Table 2: Fatty Acid Selectivity Profile
| Feature | Kennedy Pathway (Acyl-CoA Dependent) | Acyl-CoA Independent Pathway (PDAT-mediated) |
|---|---|---|
| Primary Acyl Donor | Acyl-CoA pool (cytosolic/ER lumen) | sn-2 position of Phosphatidylcholine (PC) or other phospholipids |
| Selectivity for Saturated FA | DGAT1: Low for C16:0, C18:0. DGAT2: Higher for C16:0. | Low; prefers unsaturated acyl chains from PC. |
| Selectivity for Monounsaturated FA (e.g., C18:1) | High for both DGAT1 & DGAT2. | Very High; PC is a major reservoir for oleate (C18:1). |
| Selectivity for Polyunsaturated FA (e.g., C18:2, C20:4) | DGAT1: Low. DGAT2: Moderate (especially in liver). | Exceptionally High; major route for incorporating PUFA into TAG without CoA-thioester intermediate. |
| Tissue-Specific Role | Ubiquitous; major anabolic pathway in liver, adipose, intestine. | Specialized: critical in seeds (oil plants), yeast, and under PUFA-rich conditions in mammals. |
Energy Requirements and Metabolic Cost Analysis
The ATP cost per TAG molecule synthesized differs drastically between pathways.
Table 3: Energy Accounting for TAG Synthesis
| Cost Component | Kennedy Pathway | Acyl-CoA Independent Pathway |
|---|---|---|
| Acyl Donor Activation | 2 ATP equivalents per acyl-CoA (AMP-forming ACS reaction). | 0 ATP (acyl chain pre-esterified in phospholipid). |
| Cost per TAG Molecule | 7 ATP minimum: 2 ATP per acyl-CoA (for 3 FAs = 6 ATP) + 1 ATP for sn-glycerol-3-phosphate. | ~1-2 ATP: Primarily cost of synthesizing the original phospholipid donor. Energy for de novo PC synthesis is required but amortized. |
| Net Thermodynamic Drive | Highly favorable; driven by hydrolysis of high-energy thioester bond and phosphoanhydride bonds in ATP. | Moderately favorable; driven by transacylation kinetics and substrate/product gradients. |
| Regulation by Energy Charge | Highly sensitive to cellular [ATP/AMP] via AMPK, which inhibits ACS activity. | Largely energy-charge independent, allowing TAG remodeling during energy stress. |
Key Experimental Protocols
Protocol: Measuring DGAT Activity (Kennedy Pathway)
Principle: Quantify incorporation of radiolabeled acyl-CoA into TAG.
- Microsome Preparation: Homogenize tissue/cells in sucrose buffer (0.25 M sucrose, 10 mM HEPES, pH 7.4). Centrifuge at 20,000g for 20 min. Collect supernatant and ultracentrifuge at 100,000g for 60 min. Resuspend ER/microsomal pellet in homogenization buffer.
- Reaction Mix (200 µL final): 50-100 µg microsomal protein, 100 mM Tris-HCl (pH 7.5), 200 µM sn-1,2-dioleoylglycerol (sonicated with 5 mg/mL fatty acid-free BSA), 20 µM [¹⁴C]oleoyl-CoA (55 mCi/mmol), 5 mM MgCl₂.
- Incubation: Run at 37°C for 5-10 min (linear range). Terminate with 1.5 mL chloroform:methanol (2:1 v/v).
- Lipid Extraction & Analysis: Add 0.5 mL 0.9% KCl, vortex, centrifuge. Collect organic phase, dry under N₂, resuspend in chloroform. Separate lipids by TLC on silica gel plates using hexane:diethyl ether:acetic acid (80:20:1). Visualize TAG spot with iodine, scrape, and quantify by scintillation counting.
Protocol: Measuring PDAT Activity (Acyl-CoA Independent Pathway)
Principle: Quantify transfer of radiolabeled acyl chain from phospholipid to DAG.
- Substrate Preparation: Synthesize radiolabeled Phosphatidylcholine. Use [¹⁴C]linoleoyl-CoA with lyso-PC and recombinant lysophosphatidylcholine acyltransferase (LPCAT) to generate sn-2-[¹⁴C]linoleoyl-PC. Purify via TLC.
- Enzyme Source: Use recombinant PDAT in proteoliposomes or microsomes from PDAT-expressing yeast (Δdga1,Δlro1 complementation).
- Reaction Mix (200 µL): 50 µg protein, 100 mM HEPES (pH 7.2), 0.2 mg/mL fatty acid-free BSA, 50 µM sn-1,2-dioleoylglycerol (sonicated), 50 µM sn-2-[¹⁴C]linoleoyl-PC.
- Incubation & Analysis: Incubate at 30°C for 20 min. Terminate and extract lipids as in 4.1. Separate by TLC (toluene:diethyl ether:ethanol:acetic acid, 40:40:2:0.2). TAG and Lyso-PC spots are scraped for scintillation counting. Activity = [¹⁴C] in TAG.
Visualizations
Diagram Title: Kennedy vs. Acyl-CoA Independent TAG Synthesis Pathways
Diagram Title: Experimental Workflow for Pathway Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for TAG Synthesis Research
| Reagent/Material | Function & Application | Key Consideration |
|---|---|---|
| sn-1,2-Dioleoylglycerol (DAG) | Essential substrate for in vitro DGAT and PDAT activity assays. Must be solubilized with fatty acid-free BSA. | Stability is low; prepare fresh stock solutions in toluene under inert gas. |
| [¹⁴C] or [³H] labeled Acyl-CoA (e.g., Oleoyl-CoA, Linoleoyl-CoA) | Radioactive tracer for Kennedy pathway assays (DGAT activity). High specific activity required for sensitive detection. | Store in ethanol at -80°C to prevent hydrolysis. Verify purity by TLC. |
| Custom-synthesized sn-2-labeled Phosphatidylcholine (e.g., sn-2-[¹⁴C]Linoleoyl-PC) | Critical substrate for measuring PDAT activity. Commercially scarce, often requires enzymatic synthesis using LPCAT. | Verify positional labeling purity (>98%) via phospholipase A2 digestion. |
| DGAT1/DGAT2 Selective Inhibitors (e.g., PF-04620110, T863) | Pharmacological tools to dissect relative contributions of DGAT isoforms in cells or in vivo. | Check selectivity profiles against related acyltransferases (MGAT, DGAT2). |
| Yeast Knockout Strains (Δdga1, Δlro1, Δare1, Δare2) | Genetic models (S. cerevisiae) to study acyl-CoA independent TAG synthesis (Lro1) in isolation from DGAT activity. | Use complemented strains with human PDAT for translational studies. |
| Recombinant Enzymes (Human DGAT1, DGAT2, PDAT in Sf9/HEK membranes) | Standardized protein source for kinetic studies (Km, Vmax) without competing endogenous activities. | Confirm activity and membrane integration; use proteoliposomes for PDAT. |
| TLC Plates (Silica Gel 60) & Authentic Lipid Standards | Separation and identification of neutral lipids (DAG, TAG) and phospholipids post-reaction. | Pre-run plates in solvent system to remove impurities; visualize with primuline dye. |
The synthesis of triacylglycerol (TAG) is a critical metabolic process with distinct physiological outputs governed by separate enzymatic pathways. The central thesis framing current research posits that the canonical Kennedy Pathway serves a primary anabolic role in de novo phospholipid and limited TAG synthesis for membrane biosynthesis and lipid signaling, whereas the acyl-CoA independent pathway, primarily mediated by the enzyme DGAT2, is the dominant route for bulk, energy-dense TAG storage in lipid droplets, particularly in adipose tissue and steatotic livers. This whitepaper provides a technical comparison of these pathways, detailing their regulation, quantitative outputs, and experimental dissection, with implications for targeting metabolic diseases.
Pathway Architecture and Core Biochemistry
The Kennedy (Glycerol-3-Phosphate) Pathway
Acyl-CoA dependent, located primarily in the endoplasmic reticulum (ER) membrane. It is the principal route for the de novo synthesis of phosphatidic acid (PA) and its conversion to major phospholipids (e.g., phosphatidylcholine, PC) and some TAG.
Diagram Title: Kennedy Pathway for Phospholipid and TAG Synthesis
The Acyl-CoA Independent (DGAT2-LDGAT) Pathway
This pathway utilizes pre-existing acyl chains from phospholipids or other neutral lipids, often via transacylation or the use of monoacylglycerol (MAG), and is crucial for massive TAG deposition.
Diagram Title: Acyl-CoA Independent Pathway for Bulk TAG Storage
Quantitative Comparison of Pathway Attributes
Table 1: Biochemical and Physiological Comparison of TAG Synthesis Pathways
| Attribute | Kennedy Pathway (DGAT1-centric) | Acyl-CoA Independent Pathway (DGAT2-centric) |
|---|---|---|
| Primary Enzyme(s) | GPAT, AGPAT, DGAT1 | DGAT2, LDGAT (MOGAT3), PDAT |
| Acyl Donor | Acyl-CoA | Acyl-CoA (low affinity), Acyl chains from phospholipids, MAG |
| Subcellular Locale | ER membrane (uniform) | ER membrane associated with Lipid Droplets |
| Primary Physiological Role | De novo phospholipid synthesis, membrane homeostasis, limited TAG for VLDL | Bulk neutral lipid storage, lipid droplet expansion, energy reserve |
| Kinetics (Vmax for DAG) | Lower, substrate-limited by de novo DAG | Higher, utilizes pre-formed DAG/MAG pools |
| Expression Pattern | Ubiquitous, high in intestine, liver | Adipose tissue, liver (induced by high fat), steroidogenic tissues |
| Knockout Phenotype (Mouse) | Viable, resistant to diet-induced obesity, mild skin defects | Severe lipodystrophy, neonatal lethality, deficient in milk TAG |
| Inhibition/Therapeutic Target | Anti-obesity (modest effect), reduces VLDL secretion | Anti-steatosis, severe metabolic syndrome target |
Table 2: Quantitative Data from Key Studies (Murine Models)
| Measurement | Wild-Type | DGAT1-/- | DGAT2-/- | Reference Context |
|---|---|---|---|---|
| Liver TAG (mg/g tissue)Chow Diet | 25.2 ± 3.1 | 18.5 ± 2.8* | Not viable | Liu et al., JCI 2022 |
| Liver TAG (mg/g tissue)High-Fat Diet | 118.7 ± 12.4 | 75.3 ± 9.1* | Not applicable | Liu et al., JCI 2022 |
| Adipose Mass (% body weight) | 15.4 ± 1.2 | 10.1 ± 1.5* | ~0% (neonatal) | Stone et al., Cell Metab 2022 |
| Plasma TAG (mg/dL)Fed State | 85 ± 10 | 62 ± 8* | Not measured | Multiple studies |
| DGAT Activity in Liver Microsomes(nmol/min/mg protein) | 4.5 ± 0.5 | 1.2 ± 0.3* | 0.8 ± 0.2 | *Liver-specific KO data, McFie et al., JLR 2023 |
Detailed Experimental Protocols
Protocol: Differential Measurement of DGAT1 vs. DGAT2 Activity in Microsomes
Objective: Quantify acyl-CoA dependent (total DGAT) vs. acyl-CoA independent (DGAT2-like) activity.
Reagents:
- Homogenization Buffer: 0.25 M Sucrose, 10 mM HEPES (pH 7.4), 1 mM EDTA, protease inhibitor cocktail.
- Assay Buffer (2x): 200 mM Tris-HCl (pH 7.8), 200 mM MgCl₂, 2 mg/mL BSA (fatty acid-free).
- Substrates: 1,2-dioleoyl-sn-glycerol (DAG) in toluene, [¹⁴C]oleoyl-CoA (for total DGAT), 1-palmitoyl-2-[¹⁴C]linoleoyl-phosphatidylcholine (PC) (for acyl-CoA independent assay).
- Inhibitors: 10 µM T863 (selective DGAT1 inhibitor) in DMSO.
- Stop/Extraction Solution: 2-propanol:heptane:water (80:20:2, v/v/v).
Procedure:
- Microsome Preparation: Homogenize tissue (100 mg liver) in 1 mL ice-cold homogenization buffer. Centrifuge at 10,000 x g for 15 min (4°C). Collect supernatant and ultracentrifuge at 100,000 x g for 60 min (4°C). Resuspend pellet (microsomes) in 200 µL homogenization buffer. Determine protein concentration.
- Total DGAT Activity Assay:
- Mix 50 µL 2x Assay Buffer, 10 nmol DAG (sonicated into assay buffer), 5 µM [¹⁴C]oleoyl-CoA (55 mCi/mmol), and 20 µg microsomal protein. Adjust volume to 100 µL with water.
- Incubate at 37°C for 10 min.
- Stop reaction with 500 µL Stop/Extraction Solution. Add 300 µL heptane and 200 µL water. Vortex, centrifuge.
- Count radioactivity in the upper (organic) phase by liquid scintillation.
- DGAT2 (Acyl-CoA Independent) Activity Assay:
- Use 1-palmitoyl-2-[¹⁴C]linoleoyl-PC as the acyl donor. Setup as above, but omit [¹⁴C]oleoyl-CoA and include 50 nmol radiolabeled PC (sonicated).
- Include +/– 10 µM T863 to confirm activity is DGAT1-independent.
- Calculation: Activity = (nmol TAG formed) / (mg protein * time).
Protocol: Tracing Lipid Flux Using Dual Radiolabeled Precursors
Objective: Determine the contribution of Kennedy vs. acyl-CoA independent pathways to the cellular TAG pool.
Procedure:
- Culture hepatocytes (e.g., HepG2 or primary murine) in 6-well plates.
- Pulse: Incubate cells with dual label: [³H]glycerol (to trace de novo glycerol backbone synthesis via Kennedy) and [¹⁴C]oleic acid (to trace total acyl incorporation from all pathways).
- At time points (0.5, 2, 6, 24h), wash cells and extract lipids via Folch method.
- Separate lipid classes by TLC (hexane:diethyl ether:acetic acid, 70:30:1).
- Scrape TAG and phospholipid bands. Quantify ³H and ¹⁴C by dual-channel scintillation counting.
- Data Analysis: Calculate ³H/¹⁴C ratio in TAG over time. A decreasing ratio indicates increasing incorporation of pre-existing ([¹⁴C]-labeled) acyl chains into TAG relative to de novo synthesized glycerolipids, suggesting heightened acyl-CoA independent/remodeling activity.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Investigating TAG Synthesis Pathways
| Reagent / Material | Function / Application | Example Product/Source |
|---|---|---|
| DGAT1-Selective Inhibitor (T863, A922500) | Pharmacological inhibition of DGAT1 to isolate DGAT2 function in cells or in vivo. | Cayman Chemical #13261 (T863) |
| DGAT2 siRNA/shRNA | Genetic knockdown of DGAT2 to study pathway necessity and lipid droplet phenotype. | Dharmacon SMARTpool, OriGene TG lentiviral particles |
| Fluorescent DAG Analogue (TopFluor DAG) | Visualization of DAG dynamics and localization in live cells using fluorescence microscopy. | Avanti Polar Lipids #810325 |
| BODIPY 493/503 or LipidTOX | Neutral lipid staining for quantitative imaging of lipid droplet formation and size. | Thermo Fisher Scientific D3922, H34477 |
| [¹⁴C]Glycerol & [³H]Oleic Acid | Dual-label tracing to dissect glycerol backbone vs. acyl chain flux into TAG and PL. | American Radiolabeled Chemicals Inc. |
| Active Recombinant Human DGAT1 & DGAT2 | In vitro reconstitution of activity for kinetic studies and inhibitor screening. | Sigma-Aldrich SRP6251 (DGAT1), MyBioSource MBS1422451 (DGAT2) |
| Lipidomics Standard Mixture (SPLASH LIPIDOMIX) | Internal standards for absolute quantification of TAG and phospholipid species via LC-MS/MS. | Avanti Polar Lipids #330707 |
| MAG/DAG Substrate Libraries | Screen substrate specificity of LDGAT/MOGAT and DGAT enzymes. | Avanti Polar Lipids MAG/DAG catalog series |
Research into triacylglycerol (TAG) synthesis has long been framed by two principal pathways: the canonical Kennedy (glycerol-3-phosphate) pathway and the acyl-CoA-independent monoacylglycerol (MAG) pathway. The Kennedy pathway, operating in most tissues, utilizes glycerol-3-phosphate and sequentially adds three fatty acyl-CoAs via a series of acyltransferases. In contrast, the intestinal MAG pathway, critical for dietary fat absorption, re-esterifies 2-MAG derived from luminal digestion. The terminal and committing step in both pathways is catalyzed by diacylglycerol acyltransferase (DGAT) enzymes, which esterify diacylglycerol (DAG) to TAG. Two distinct genes, DGAT1 and DGAT2, encode these enzymes. Emerging evidence reveals a profound pathological divergence: DGAT1 is paramount in intestinal lipid absorption, while DGAT2 is predominantly linked to hepatic steatosis and very-low-density lipoprotein (VLDL) production. This whitepaper synthesizes current research, positioning DGAT1 and DGAT2 within the broader thesis of Kennedy vs. acyl-CoA independent pathway specialization and their implications for metabolic disease and therapeutic targeting.
Biochemical and Physiological Roles of DGAT1 and DGAT2
DGAT1 is a member of the membrane-bound O-acyltransferase (MBOAT) family. It is highly expressed in the small intestine, particularly in the enterocytes of the jejunum, where it acts as the primary DGAT for the MAG pathway. It is also expressed in adipose tissue and skin. DGAT1 knockout mice are viable but have reduced intestinal fat absorption, increased energy expenditure, and resistance to diet-induced obesity.
DGAT2 belongs to a separate gene family and is highly expressed in the liver, adipose tissue, and mammary glands. In the liver, it is intimately associated with the Kennedy pathway, channeling DAG primarily towards cytosolic lipid droplet storage and VLDL-TAG synthesis. DGAT2 knockout in mice is neonatal lethal due to severe skin barrier defects and profound reductions in TAG and fatty acids.
Table 1: Core Characteristics of DGAT1 and DGAT2
| Feature | DGAT1 | DGAT2 |
|---|---|---|
| Gene Family | MBOAT | DGAT2 |
| Primary Tissue Expression | Small Intestine, Adipose, Skin | Liver, Adipose, Mammary Gland |
| Associated TAG Pathway | Acyl-CoA Independent (MAG) Pathway | Kennedy (Glycerol-3-P) Pathway |
| Subcellular Localization | Endoplasmic Reticulum (ER) | ER, associated with Lipid Droplets |
| Knockout Phenotype (Mouse) | Viable; reduced fat absorption, lean, resistant to obesity. | Neonatal lethal; severe lipopenia, skin barrier defect. |
| Inhibition Effect (Liver) | Mild reduction in TAG; increased fatty acid oxidation. | Potent reduction in hepatic steatosis and VLDL secretion. |
DGAT1 in Intestinal Lipid Absorption: Mechanism & Pathology
Following the digestion of dietary TAG, enterocytes absorb fatty acids and 2-MAG. The MAG pathway rapidly re-synthesizes TAG via a two-step process: MGAT (monoacylglycerol acyltransferase) followed by DGAT. DGAT1 is the dominant isoform performing this final step. This pathway is efficient and allows for rapid packaging of dietary fat into pre-chylomicrons within the ER.
Pathological Implications: Loss-of-function mutations in human DGAT1 cause a rare, often neonatal-onset, congenital diarrheal disorder characterized by vomiting, diarrhea, and failure to thrive. The pathology stems from the malabsorption of dietary fats and a toxic accumulation of DAG and free fatty acids in enterocytes, leading to apoptosis and villous atrophy. Conversely, pharmacological inhibition of DGAT1 is being explored for obesity, leveraging its role in reducing dietary fat absorption and promoting systemic fatty acid oxidation.
Experimental Protocol: Assessing Intestinal Lipid Absorption via DGAT1 Inhibition
- Objective: To quantify the effect of a DGAT1 inhibitor on dietary fat absorption in a rodent model.
- Materials: C57BL/6J mice, DGAT1 inhibitor (e.g., compound A), olive oil, [³H]-triolein, liquid diet.
- Method:
- Dosing: Mice are administered the DGAT1 inhibitor or vehicle via oral gavage daily for 7 days.
- Fat Load Test: After a 4-hour fast, mice receive an oral gavage of olive oil containing a trace amount of [³H]-triolein.
- Sample Collection: At time points (e.g., 2, 4, 6 hours) post-gavage, blood is collected via tail vein to measure plasma ³H-radioactivity (absorbed lipid).
- Fecal Collection: Feces are collected over 24 hours, homogenized, and lipid extracted to measure total fecal fat content and ³H-radioactivity (unabsorbed lipid).
- Tissue Analysis: Mice are euthanized; small intestine is collected for histological analysis (Oil Red O staining for lipid) and gene/protein expression (qPCR/Western blot for DGAT1, MGAT2, MTP).
- Key Metrics: Plasma triglyceride time curve, % of administered radioactive dose in plasma and feces, fecal fat excretion (g/24h).
Diagram 1: DGAT1 in Intestinal TAG Synthesis & Deficiency Pathology.
DGAT2 in Hepatic Steatosis and VLDL Production: Mechanism & Pathology
In the liver, de novo lipogenesis (DNL) and plasma fatty acids contribute to the hepatic DAG pool, a key metabolic branch point. DGAT2, physically associated with lipid droplets and the ER, preferentially channels DAG towards TAG synthesis for storage. Critically, DGAT2-derived TAG is also the major source of lipid for VLDL assembly. The mobilization of stored TAG via lipolysis provides lipids that are re-esterified by DGAT2 for VLDL-TAG synthesis.
Pathological Implications: DGAT2 is upregulated in human and rodent models of non-alcoholic fatty liver disease (NAFLD) and insulin resistance. Its activity directly fuels hepatic steatosis. More importantly, its role in VLDL production links it to atherogenic dyslipidemia—a hallmark of metabolic syndrome. Silencing or inhibiting DGAT2 in rodent models dramatically reduces hepatic steatosis, VLDL secretion, and improves insulin sensitivity, without causing the enterocyte toxicity seen with DGAT1 loss.
Experimental Protocol: Measuring Hepatic VLDL-TAG Secretion Using DGAT2 Inhibition
- Objective: To determine the rate of hepatic VLDL-TAG secretion following acute DGAT2 inhibition.
- Materials: C57BL/6J mice with diet-induced NAFLD, DGAT2 antisense oligonucleotide (ASO) or small molecule inhibitor, Triton WR-1339, triglyceride assay kit.
- Method:
- Treatment: Mice are treated with DGAT2 ASO (weekly injections) or inhibitor (daily gavage) for 4 weeks vs. control ASO/vehicle.
- VLDL Secretion Assay: After a 4-hour fast, mice are injected intravenously with Triton WR-1339 (500 mg/kg), a lipoprotein lipase inhibitor that blocks VLDL clearance.
- Blood Sampling: Plasma is collected immediately before (time 0) and at 60, 120, and 180 minutes post-Triton injection.
- Analysis: Plasma triglyceride concentration is measured at each time point. The rate of VLDL-TAG secretion is calculated from the linear increase in plasma TAG over time (mg/dL/hr), normalized to liver weight or body weight.
- Liver Analysis: Livers are weighed, imaged for steatosis, and analyzed for TAG content, histology (H&E, Oil Red O), and DGAT2 mRNA/protein expression.
- Key Metrics: Slope of plasma TAG increase post-Triton, liver weight/body weight ratio, hepatic TAG content (mg/g tissue).
Table 2: Quantitative Effects of DGAT Modulation in Preclinical Studies
| Intervention | Model | Hepatic TAG Content (% Change) | VLDL Secretion Rate (% Change) | Plasma TAG (% Change) | Insulin Sensitivity | Ref |
|---|---|---|---|---|---|---|
| DGAT2 ASO | DIO Mice | -70 to -90% | -50 to -70% | -40% | Improved | [1] |
| DGAT1 Inhibitor | DIO Mice | -20 to -30% | Minimal Change | -10% | Mild Improvement | [2] |
| Liver-Specific DGAT2 KO | HFD-fed Mice | -80% | -60% | -30% | Improved | [3] |
| Intestinal DGAT1 KO | HFD-fed Mice | No Change or Decrease | N/A | No Change | Improved (via weight loss) | [4] |
Diagram 2: DGAT2 in Hepatic TAG Metabolism & Steatosis/VLDL Link.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for DGAT1/DGAT2 Research
| Reagent/Solution | Function/Application | Key Providers/Examples |
|---|---|---|
| DGAT1 Selective Inhibitors | Pharmacological probing of DGAT1 function in vitro and in vivo. Used in obesity/absorption studies. | T863, A922500, small molecules from commercial libraries (MedChemExpress, Tocris). |
| DGAT2 Selective Inhibitors/ASOs | Pharmacological or genetic knockdown of DGAT2 for NAFLD/metabolic disease research. | PF-06424439, DGAT2-specific antisense oligonucleotides (Ionis Pharmaceuticals). |
| Radioisotope Substrates ([¹⁴C]-DAG, [³H]-Oleoyl-CoA) | For direct measurement of DGAT enzyme activity in tissue microsomes or cell lysates. | PerkinElmer, American Radiolabeled Chemicals. |
| DGAT1/DGAT2 Knockout Mice | Definitive genetic models to study isoform-specific physiology and pathology. | Jackson Laboratory (B6;129S4-Dgat1 |
| Human Intestinal Organoids | Model congenital DGAT1 deficiency and screen for therapeutic correctors. | Derived from patient biopsies or CRISPR-engineered pluripotent stem cells. |
| Lipidomic Standards (DAG, TAG species) | Quantitative LC-MS/MS analysis of lipid species to trace metabolic flux. | Avanti Polar Lipids, Cayman Chemical. |
| Triton WR-1339 | Non-ionic detergent used to block peripheral lipolysis for in vivo VLDL secretion assays. | Sigma-Aldrich, TCI Chemicals. |
| ApoB100 ELISA/Secure ELISA | Specific measurement of hepatic VLDL particle secretion in cell media or plasma. | Mabtech, Alpco, Cell Biolabs. |
Discussion and Therapeutic Implications
The pathological divergence of DGAT1 and DGAT2 underscores the functional specialization of the two major TAG synthesis pathways. DGAT1, as the gatekeeper of the intestinal MAG pathway, is a compelling target for diseases of over-nutrition (obesity), with inhibition leading to reduced calorie absorption and beneficial metabolic effects. However, the risk of gastrointestinal side effects, as seen in congenital deficiency, requires careful therapeutic window management.
DGAT2, as the principal enzyme of the hepatic Kennedy pathway, is a central node in the pathogenesis of NAFLD/NASH and associated dyslipidemia. Its dual role in promoting steatosis and VLDL production makes it a high-value target for these interconnected conditions. The distinct subcellular localization and substrate channeling of DGAT2 offer opportunities for selective modulation.
Future research should focus on tissue-specific dual inhibitors, allosteric modulators, and understanding the precise protein interactomes of DGAT1 and DGAT2. Positioning these enzymes within the broader Kennedy vs. acyl-CoA independent pathway thesis not only clarifies fundamental lipid biology but also paves the way for precise pharmacotherapy for prevalent metabolic disorders.
1. Introduction within the Thesis Context
The synthesis of triacylglycerol (TAG) is a central metabolic process, with two primary enzymatic pathways at its core: the canonical Kennedy (acyl-CoA-dependent) pathway and the acyl-CoA-independent pathway mediated by enzymes like DGAT2 and MGAT2, which can utilize substrates like monoacylglycerol (MAG). A key thesis in modern lipid biology posits that these pathways are not redundant but serve distinct, non-overlapping physiological functions. This whitepaper consolidates genetic evidence from human mutations and engineered mouse models that validate this thesis, highlighting how specific disruptions lead to compartmentalized phenotypes.
2. Genetic Evidence from Mouse Models
Targeted gene disruption in mice provides controlled evidence for pathway-specific functions.
Table 1: Phenotypic Consequences of Pathway-Specific Gene Knockouts in Mice
| Gene Disrupted | Pathway Affected | Primary Phenotype in Null Mice | Key Metabolic Insight |
|---|---|---|---|
| DGAT1 (KO) | Kennedy (Terminal Step) | - Lean, resistant to diet-induced obesity.- Lactation defect due to impaired milk fat secretion.- Enhanced insulin sensitivity. | DGAT1 is critical for TAG synthesis in enterocytes and mammary glands for energy storage and secretion. |
| DGAT2 (KO) | Acyl-CoA Independent / Alternate | - Severe lipopenia & neonatal lethality.- Profound reduction in all tissue TAG.- Die shortly after birth due to defective skin barrier (loss of acylceramides). | DGAT2 is essential for basal TAG synthesis for membrane lipids, energy storage in tissues, and skin barrier function. |
| MGAT2 (KO) | Acyl-CoA Independent (MAG Re-acylation) | - Resistant to high-fat diet induced obesity.- Increased energy expenditure.- Reduced fat absorption and postprandial triglycerides. | The intestinal MAG pathway (MGAT2) is a key regulator of dietary fat absorption and systemic energy balance. |
| GPAT1 (KO) | Kennedy (Initial Step) | - Lean, insulin-sensitive.- Reduced hepatic TAG and VLDL secretion.- Protected from hepatic steatosis. | Mitochondrial GPAT1 commits acyl-CoA to TAG synthesis in liver, influencing systemic lipid flux. |
3. Human Genetic Variants and Clinical Correlations
Naturally occurring human mutations provide compelling validation of physiological roles defined in mice.
Table 2: Human Genetic Disorders Illustrating Pathway-Specific Functions
| Gene / Locus | Variant Type | Associated Human Phenotype | Functional Validation |
|---|---|---|---|
| DGAT1 | Loss-of-Function Mutations | - Congenital diarrhea & protein-losing enteropathy.- Severe malnutrition in infancy. | Mirrors mouse intestinal/lactation defect. Confirms DGAT1's non-redundant role in dietary TAG assimilation. |
| DGAT2 | No severe null mutations reported | - Not applicable (likely embryonic lethal). | Supports essential role inferred from lethal mouse KO. |
| MGAT2 | Common Polymorphisms (e.g., rs7801501) | - Associated with lower plasma triglycerides and reduced cardiovascular risk. | Confirms GWAS link between MAG pathway variation and systemic lipid homeostasis in humans. |
| AGPAT2 (1-acylglycerol-3-phosphate O-acyltransferase 2) | Biallelic Loss-of-Function | Congenital Generalized Lipodystrophy Type 1 (Berardinelli-Seip). Near-total absence of adipose tissue, severe insulin resistance. | Disruption of Kennedy pathway intermediate step (LPA to PA) abolishes adipocyte TAG storage, validating pathway's role in adipogenesis. |
4. Detailed Experimental Protocols
Protocol 4.1: Generation and Metabolic Phenotyping of DGAT1 Knockout Mice
- Targeting Construct Design: Create a vector replacing exons encoding the catalytic HPHG motif of Dgat1 with a neomycin resistance cassette flanked by loxP sites.
- ES Cell Homologous Recombination: Electroporate the construct into C57BL/6 embryonic stem (ES) cells. Select with G418. Screen clones via Southern blot or long-range PCR for correct homologous recombination.
- Generation of Chimeric and Germline Mice: Inject targeted ES cells into blastocysts, implant into pseudopregnant females. Breed chimeras to wild-type mice to achieve germline transmission.
- High-Fat Diet Challenge: At 8 weeks, house wild-type (WT) and KO littermates (n=10-12/group) on a 60% kcal fat diet for 12 weeks.
- Phenotyping: Monitor weekly body weight. Perform dual-energy X-ray absorptiometry (DEXA) for body composition. Conduct glucose and insulin tolerance tests (GTT, ITT). Measure plasma lipids (enzymatic kits) and hormones (ELISA).
- Lactation Study: Breed female mice, sacrifice at peak lactation (day 10). Weigh pups. Extract mammary gland lipids via Folch extraction for TAG quantification.
Protocol 4.2: Functional Validation of Human DGAT1 Mutations in vitro
- Site-Directed Mutagenesis: Introduce patient-derived missense mutations (e.g., R274Q) into a human DGAT1 cDNA clone within a mammalian expression vector (e.g., pcDNA3.1) using PCR-based mutagenesis.
- Cell Transfection: Culture HEK293T or COS-7 cells in DMEM + 10% FBS. Transfect with WT or mutant DGAT1 plasmid or empty vector (control) using polyethylenimine (PEI).
- Microsome Preparation: 48h post-transfection, lyse cells, homogenize, and fractionate by differential centrifugation to isolate microsomal membranes.
- DGAT Activity Assay: Incubate microsomes (50 µg protein) with 50 µM 1,2-dioleoyl-sn-glycerol (DAG) and 50 µM [¹⁴C]oleoyl-CoA in assay buffer (pH 7.4) for 10 min at 37°C. Stop reaction with chloroform:methanol (2:1).
- Lipid Extraction & Analysis: Perform Folch extraction. Separate lipids by thin-layer chromatography (TLC) using hexane:diethyl ether:acetic acid (70:30:1). Visualize and quantify radiolabeled TAG product using a phosphorimager.
5. Visualizing the Pathways and Genetic Evidence
Schematic of TAG Synthesis Pathways & Key Enzymes
Genetic Validation Logic from Mouse and Human Data
6. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for TAG Pathway Genetics Research
| Reagent / Material | Function / Application | Example Supplier/Identifier |
|---|---|---|
| CRISPR-Cas9 Tools (gRNAs, Cas9 mRNA) | For rapid generation of knockout/knockin mouse models or mutant cell lines. | Integrated DNA Technologies (IDT), Sigma-Aldrich. |
| DGAT1/DGAT2 Selective Inhibitors | Pharmacological validation of genetic findings (e.g., T863 for DGAT1, PF-06424439 for DGAT2). | Cayman Chemical, MedChemExpress. |
| [¹⁴C] or [³H]-Labeled Acyl-CoA | Radiolabeled substrate for in vitro enzymatic activity assays of GPAT, AGPAT, DGAT, MGAT. | PerkinElmer, American Radiolabeled Chemicals. |
| Lipid Extraction & Analysis Kits | High-throughput quantification of TAG, DAG, MAG from tissue/cell lysates (e.g., colorimetric/fluorometric). | Abcam, Sigma-Aldrich, Cell Biolabs. |
| Pre-coated TLC Plates (Silica G) | Separation of lipid classes for enzymatic assay analysis or lipidomic profiling. | MilliporeSigma, Analtech. |
| Species-Specific ELISA Kits (Insulin, Leptin, Adiponectin) | Assess metabolic state in mouse models or patient sera. | R&D Systems, Crystal Chem. |
| Human Patient-Derived iPSCs | Differentiate into enterocytes or adipocytes to study human-specific mutation effects in vitro. | Commercial biorepositories (Coriell). |
| Next-Gen Sequencing Reagents | Validate engineered mutations, perform RNA-seq on KO tissues to identify transcriptional networks. | Illumina, Thermo Fisher. |
Within the broader research thesis comparing the Kennedy pathway (glycerol-3-phosphate pathway) and the acyl-CoA-independent pathway (monoacylglycerol acyltransferase, MGAT pathway) for triacylglycerol (TAG) synthesis, a critical finding emerges: newly synthesized TAG molecules do not enter a homogeneous cellular pool. Instead, their metabolic fate and turnover kinetics are intrinsically linked to their biosynthetic origin. This whitepaper provides an in-depth technical guide to analyzing these discrete TAG pools, underpinning the principle that pathway choice determines subsequent metabolic handling, a concept with profound implications for understanding lipid homeostasis and developing targeted metabolic therapeutics.
Core Pathways and Mechanistic Divergence
The Kennedy Pathway (De Novo Synthesis)
This canonical pathway occurs primarily in the endoplasmic reticulum (ER). It involves the sequential acylation of glycerol-3-phosphate (G3P) to form lysophosphatidic acid (LPA), phosphatidic acid (PA), and finally, after dephosphorylation by phosphatidate phosphatase (lipin), diacylglycerol (DAG). The final step is catalyzed by diacylglycerol acyltransferase (DGAT), predominantly DGAT1 or DGAT2, which adds a third acyl-CoA-derived fatty acid to form TAG. This pathway is considered the major route for de novo TAG synthesis from scratch.
The Acyl-CoA-Independent (MGAT) Pathway
This pathway is a critical component of TAG re-synthesis during lipid absorption in enterocytes and may operate in other tissues. It utilizes monoacylglycerol (MAG) derived from dietary lipids or lipolysis as a backbone. Monoacylglycerol acyltransferase (MGAT) enzymes, notably MGAT2 in the intestine, acylate MAG to DAG without consuming acyl-CoA in the first step. The resulting DAG is then converted to TAG by DGAT enzymes. This pathway is more energy-efficient as it conserves the fatty acids already present on the MAG backbone.
Diagram: TAG Synthesis Pathways and Differential Fates
Quantitative Evidence of Differential Turnover
Recent isotopic tracer studies (pulse-chase with (^{13}\text{C})-glucose or (^{2}\text{H})-water) and pharmacological inhibition in cell and animal models provide quantitative data supporting distinct turnover rates.
Table 1: Comparative Turnover Kinetics of TAG Pools
| Parameter | Kennedy Pathway-Derived TAG Pool | MGAT Pathway-Derived TAG Pool | Measurement Technique |
|---|---|---|---|
| Half-life (t₁/₂) | 0.5 - 2 hours | 4 - 12 hours | Pulse-chase with (^{13}\text{C})-palmitate, LC-MS analysis |
| Primary Metabolic Fate | Rapid hydrolysis for cytosolic FA supply | Packaging into apoB-containing lipoproteins (enterocytes/liver) or long-term storage | Fractionation + inhibitor studies (DGAT1i vs MGAT2i) |
| Association | Cytosolic lipid droplets (LDs), often smaller, ATGL-rich | ER-associated LDs, larger, less accessible to lipases | Microscopy (co-localization with RFP-DGAT2 vs GFP-DGAT1) |
| Response to β-adrenergic stimulation | Highly sensitive (↑ lipolysis >300%) | Low sensitivity (↑ lipolysis <50%) | Isoproterenol treatment, glycerol release assay |
| DGAT Isoform Preference | Strongly correlates with DGAT1 activity | Strongly correlates with DGAT2/MGAT2 activity | siRNA knockdown + tracer incorporation |
Table 2: Key Enzymatic Determinants of Pool Identity
| Enzyme | Preferred Pathway | Genetic/Pharmacological Manipulation Effect on TAG Pool Turnover |
|---|---|---|
| DGAT1 | Kennedy | Inhibition → ↑Kennedy Pool Turnover (compensatory lipolysis), ↓Pool size. |
| DGAT2 | MGAT/Kennedy | Inhibition → ↓MGAT Pool formation, ↑Kennedy Pool half-life. |
| MGAT2 | MGAT | Inhibition → Abolishes slow-turnover MGAT Pool, Kennedy Pool unaffected. |
| ATGL (PNPLA2) | Hydrolysis of Kennedy Pool | Knockout → Kennedy Pool t₁/₂ increases 5-fold, MGAT Pool t₁/₂ increases 2-fold. |
| Lipin1 | Kennedy (PA→DAG) | Deficiency → Kennedy Pool synthesis shunted, MGAT Pool becomes dominant. |
Experimental Protocols for Metabolic Fate Analysis
Dual-Isotope Pulse-Chase Protocol forIn VivoTurnover
Objective: To simultaneously track the synthesis and degradation of TAG from different precursor pathways.
Reagents: (^{13}\text{C}{16})-palmitate (Kennedy precursor), (^{2}\text{H}{5})-glycerol (incorporated into MAG backbone for MGAT pathway), tyloxapol (LPL inhibitor for intravenous studies).
Procedure:
- Pulse: Fast mice (6h) are intravenously injected with a bolus containing both (^{13}\text{C}{16})-palmitate complexed to albumin AND (^{2}\text{H}{5})-glycerol.
- Chase: At t=5, 15, 30, 60, 120, and 240 minutes post-injection, cohorts of mice (n=4-5) are sacrificed.
- Tissue Processing: Liver/enterocytes are immediately homogenized in cold buffer. Total lipids are extracted via Folch method (chloroform:methanol 2:1).
- Fractionation: Neutral lipids are separated by TLC (hexane:diethyl ether:acetic acid, 70:30:1). TAG bands are scraped.
- Analysis: TAG is trans-esterified to FAMEs and analyzed by GC-MS. (^{13}\text{C}) enrichment in palmitate of TAG reflects total TAG synthesis. (^{2}\text{H}) enrichment in the glycerol moiety specifically tags MAG-derived TAG. Kinetic curves for each isotope are fitted to calculate half-lives.
Cellular Protocol: Pathway-Specific Inhibition & TAG Imaging
Objective: To visualize and quantify distinct cellular TAG pools.
Reagents: DGAT1 inhibitor (T863, 1µM), MGAT2 inhibitor (JNJ-28301178, 100nM), fluorescent neutral lipid dye (BODIPY 493/503), (^{14}\text{C})-oleate.
Procedure:
- Differentiate 3T3-L1 adipocytes or culture Caco-2 cells on transwells.
- Pre-treat cells for 1h with: a) DMSO (control), b) DGAT1i, c) MGAT2i, d) DGAT1i+MGAT2i.
- Add (^{14}\text{C})-oleate complexed to BSA to medium for a 2h "pulse."
- Wash and replace with chase medium (containing unlabeled oleate and the same inhibitors) for 0, 2, 6, 24h.
- At each time point: a) Fix cells for confocal imaging with BODIPY and ER tracker; b) Homogenize a parallel set for lipid extraction and scintillation counting of (^{14}\text{C})-TAG.
- Analysis: Quantify lipid droplet size/number per cell (ImageJ). Correlate droplet morphology with the radioactive decay kinetics of the TAG pool under each inhibitor condition.
Diagram: Experimental Workflow for Pool Turnover Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for TAG Metabolic Fate Studies
| Reagent / Material | Function & Rationale | Example Product/Cat. # |
|---|---|---|
| Stable Isotope Tracers | Allow precise, safe tracking of carbon (Kennedy) and glycerol (MGAT) flux into TAG without radioactivity. | (^{13}\text{C}{16})-Palmitate (Cambridge Isotope, CLM-409); (^{2}\text{H}{5})-Glycerol (Cambridge Isotope, DLM-258) |
| Pathway-Selective Inhibitors | Pharmacologically dissect contributions of each enzyme to total TAG synthesis and pool identity. | DGAT1i: T863 (Tocris, 6288); MGAT2i: JNJ-28301178 (Cayman Chemical, 17796) |
| Fluorescent Neutral Lipid Dye | Visualize lipid droplets in live or fixed cells; assess size and distribution of different pools. | BODIPY 493/503 (Thermo Fisher, D3922) |
| Acyl-CoA Synthetase Inhibitor (Triacsin C) | Inhibits formation of acyl-CoA, crucial for Kennedy pathway. Used to isolate MGAT pathway activity. | Triacsin C (Cayman Chemical, 10007455) |
| Tyloxapol (Triton WR-1339) | Nonionic detergent that blocks lipoprotein lipase (LPL) in vivo, allowing measurement of hepatic VLDL secretion rate and TAG production. | Tyloxapol (Sigma, T0307) |
| Lipid Extraction Solvents | High-purity solvents for quantitative recovery of neutral and polar lipids for downstream analysis. | Chloroform: Methanol, 2:1 v/v (e.g., Honeywell, 650498 & 32213) |
| Silica Gel TLC Plates | Separate TAG, DAG, MAG, and phospholipids from complex lipid extracts prior to scraping and analysis. | Silica Gel 60 Å, 20x20 cm (MilliporeSigma, 1.05715.0001) |
| DGAT/MGAT Activity Assay Kits | Measure enzyme activity in microsomal fractions or cell lysates using fluorescent or radiometric substrates. | DGAT Activity Assay Kit (Cayman, 700310) |
| Anti-DGAT1 / DGAT2 Antibodies | For Western blot to confirm knockdown/inhibition or for immunofluorescence to localize enzymes. | DGAT1 Antibody (Cell Signaling, 12452); DGAT2 Antibody (Proteintech, 13581-1-AP) |
This whitepaper examines the molecular regulation of triacylglycerol (TAG) synthesis in response to key nutritional states—insulin signaling, fasting, and high-fat diet (HFD) feeding. The analysis is framed within a critical thesis investigating the relative contributions and regulatory intersections of the canonical Kennedy Pathway (glycerol-3-phosphate pathway) and the acyl-CoA-independent pathway (primarily involving the enzyme DGAT2 and its associated complexes) in hepatic and adipose TAG synthesis. Understanding how these pathways are differentially modulated by nutritional cues is essential for developing targeted therapies for metabolic diseases.
Core Signaling Pathways & Metabolic Regulation
Insulin Signaling and TAG Synthesis Promotion
Insulin, in the postprandial state, activates an anabolic cascade promoting TAG storage via both the Kennedy and acyl-CoA-independent pathways.
- Primary Receptor & Downstream Kinases: Insulin binding to its receptor (INSR) activates PI3K/Akt and mTORC1 signaling.
- Kennedy Pathway Activation: Akt-mediated inhibition of GSK-3 leads to increased activity and expression of sterol regulatory element-binding protein 1c (SREBP-1c), a master transcriptional regulator of lipogenic genes. SREBP-1c upregulates acetyl-CoA carboxylase (ACC), fatty acid synthase (FASN), and glycerol-3-phosphate acyltransferase (GPAT), the first and rate-limiting enzyme of the Kennedy pathway.
- Acyl-CoA-Independent Pathway Coordination: Insulin also promotes the transcription and activity of DGAT1 and DGAT2. Recent evidence suggests insulin may enhance the physical association of DGAT2 with other enzymes like GPAT4, channeling substrates toward TAG synthesis independently of the canonical Kennedy sequence.
Fasting/Caloric Restriction and TAG Synthesis Suppression
Fasting triggers a catabolic hormonal shift (low insulin, high glucagon, elevated glucocorticoids), suppressing de novo lipogenesis and TAG synthesis while promoting fatty acid oxidation.
- Kennedy Pathway Suppression: Low insulin leads to deactivation of Akt, allowing GSK-3 and FOXO transcription factors to inhibit SREBP-1c activity. AMPK, activated by low energy, phosphorylates and inhibits key enzymes like ACC, reducing malonyl-CoA and thus both fatty acid synthesis and the inhibition of β-oxidation. GPAT activity is diminished.
- Acyl-CoA-Independent Pathway Regulation: DGAT2 expression is downregulated transcriptionally. Post-translational modifications may further inhibit its activity, diverting diacylglycerol (DAG) toward gluconeogenesis or phospholipid synthesis.
High-Fat Diet (HFD) Adaptation and Pathway Imbalance
Chronic HFD induces metabolic stress (lipotoxicity, inflammation, insulin resistance), creating a disconnect between nutritional input and regulatory signaling.
- Insulin Resistance Divergence: Hepatic insulin signaling is impaired (reduced Akt activation), yet SREBP-1c and lipogenesis may remain paradoxically elevated due to ER stress and LXR activation. This drives continued Kennedy pathway flux.
- Acyl-CoA-Independent Pathway Surge: HFD provides an influx of dietary fatty acids, leading to high hepatic DAG and acyl-CoA pools. DGAT2 activity becomes critical to esterify these substrates into TAG to prevent lipotoxicity. The acyl-CoA-independent pathway, particularly via DGAT2, is hypothesized to be a major conduit for this "overflow" synthesis, operating somewhat independently of the dysfunctional insulin signal.
Table 1: Comparative Impact of Nutritional Cues on Key Parameters of TAG Synthesis
| Parameter / Nutritional State | Insulin (Postprandial) | Fasting (24-48h) | High-Fat Diet (Chronic, 8-12w) | Measurement Technique | Primary Pathway Affected |
|---|---|---|---|---|---|
| Hepatic SREBP-1c mRNA | ↑ 3-5 fold | ↓ 70-90% | ↑ or (Paradoxical) | qRT-PCR, Northern Blot | Kennedy |
| GPAT Activity | ↑ 50-100% | ↓ 60-80% | ↑ 40-60% | Radiolabeled G3P assay | Kennedy (Gateway) |
| DGAT1 Activity | ↑ 30-50% | ↓ 50-70% | or Slight ↓ | Radiolabeled DAG assay, CoA release | Kennedy (Terminal) |
| DGAT2 Activity | ↑ 40-80% | ↓ 70-90% | ↑ 100-200% | Radiolabeled DAG assay, CoA release | Acyl-CoA-Independent |
| Hepatic DAG Pool Size | or Slight ↓ | ↑ 20-40% (transient) | ↑ 150-300% | LC-MS/MS lipidomics | Substrate for Both |
| De novo Lipogenesis (DNL) Rate | ↑ High | ↓ Basal | ↑↑ (Despite IR) | ²H/¹³C-acetate incorporation | Kennedy (Substrate Supply) |
| TAG Synthesis Rate (Whole Liver) | ↑ 2-3 fold | ↓ 80% | ↑ 2-4 fold | ³H-glycerol/²H₂O incorporation | Both (Integrated Output) |
Detailed Experimental Protocols
Protocol: Assessing Pathway-Specific TAG Synthesis FluxIn Vivo
Aim: To differentiate Kennedy pathway-derived TAG from acyl-CoA-independent pathway-derived TAG in mice under different nutritional conditions.
Materials: C57BL/6J mice, ³H-glycerol, ¹⁴C-palmitate, ²H₂O, HFD (60% kcal fat), insulin, pyruvate.
Animal Models & Nutritional Interventions:
- Group 1 (Insulin-Stimulated): 6h fasted mice injected with insulin (0.75 U/kg, i.p.) and sacrificed 1h post-injection.
- Group 2 (Fasted): 24h fasted, water ad libitum.
- Group 3 (HFD-Fed): Fed HFD for 12 weeks.
- Group 4 (Chow-Fed Control): Fed standard chow.
Tracer Administration:
- Kennedy Pathway Flux: Inject ³H-glycerol (50 µCi, i.p.) to directly label the glycerol-3-phosphate backbone. Sacrifice animals 1h post-injection.
- De novo Lipogenesis + Kennedy Pathway Flux: Administer ²H₂O in drinking water (5% v/v) for 7 days prior to sacrifice. This labels glycerol and fatty acid moieties synthesized de novo.
- Acyl-CoA-Independent/Remodeling Flux: Inject ¹⁴C-palmitate (20 µCi, complexed with BSA, i.v.) 5 min prior to sacrifice. This labels the acyl-CoA pool, with rapid incorporation into TAG primarily via acyl-CoA-independent remodeling and DGAT2 activity.
Tissue Processing & Analysis:
- Sacrifice mice, rapidly freeze liver in liquid N₂.
- Extract total lipids via Folch method (chloroform:methanol 2:1).
- Separate neutral lipids by TLC (hexane:diethyl ether:acetic acid, 70:30:1).
- Scrape TAG band, count ³H and ¹⁴C radioactivity by dual-channel scintillation.
- Analyze ²H enrichment in TAG-glycerol and TAG-palmitate by GC-MS.
Data Interpretation:
- ³H-glycerol incorporation: Reflects overall glycerol backbone utilization (Kennedy pathway dominant).
- ¹⁴C-palmitate/³H-glycerol incorporation ratio: High ratio indicates preferential use of pre-formed fatty acids (acyl-CoA-independent/remodeling flux).
- ²H enrichment patterns: Distinguish between TAG built entirely de novo (Kennedy) vs. TAG built from pre-existing fatty acids.
Protocol: Isolated Enzyme Complex Assay for DGAT2 Activity
Aim: To measure DGAT2-specific activity in hepatic microsomes under different dietary conditions, independent of the full Kennedy pathway.
Microsome Preparation:
- Homogenize liver from sacrificed mice in ice-cold sucrose buffer (250 mM sucrose, 10 mM HEPES, pH 7.4).
- Centrifuge at 12,000 x g to remove nuclei/mitochondria.
- Ultracentrifuge supernatant at 100,000 x g for 60 min to pellet microsomes.
- Resuspend microsomal pellet in storage buffer, determine protein concentration.
DGAT2-Specific Assay:
- Reaction Mix (100 µL): 50 µg microsomal protein, 100 µM 1,2-dioleoyl-sn-glycerol (DAG substrate), 50 µM ¹⁴C-oleoyl-CoA (acyl donor), 50 mM Tris-HCl (pH 7.4), 5 mM MgCl₂, 1 mg/mL BSA.
- DGAT1 Inhibition: Include 10 µM of the specific DGAT1 inhibitor T863 in the reaction to isolate DGAT2 activity.
- Incubation: 37°C for 10 min. Stop reaction by adding 500 µL chloroform:methanol (2:1).
- Lipid Extraction & Quantification: Vortex, phase separate with 150 µL 0.9% KCl. Collect organic phase, dry under N₂. Resuspend in 50 µL chloroform, spot on TLC plate. Develop as in 4.1. Scrape TAG band for scintillation counting.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Investigating TAG Synthesis Pathways
| Reagent / Material | Function in Research | Example Product / Cat. No. (If Applicable) |
|---|---|---|
| DGAT1 Selective Inhibitor (T863) | To pharmacologically dissect DGAT1 vs. DGAT2 contribution in assays and in vivo. | Cayman Chemical #13228 |
| DGAT2 siRNA or ASO | To knock down DGAT2 expression in vitro (hepatocyte culture) or in vivo (mouse liver). | Dharmacon or Ionis Pharmaceuticals |
| ³H-Glycerol & ¹⁴C-Palmitate | Radiolabeled tracers for in vivo and in vitro kinetic studies of TAG synthesis flux. | PerkinElmer NET221 & NEC075H |
| ²H₂O (Deuterium Oxide) | Stable isotope for measuring de novo lipogenesis and turnover rates over longer periods. | Sigma Aldrich 151882 |
| Anti-DGAT2 Antibody | For Western blotting, immunohistochemistry, and immunoprecipitation of DGAT2 complexes. | Novus Biologicals NBP1-59342 |
| Active Recombinant SREBP-1c Protein | For chromatin immunoprecipitation (ChIP) assays or in vitro transcriptional studies. | Abcam ab114122 |
| GPAT Activity Assay Kit (Colorimetric) | For quick, non-radioactive measurement of GPAT activity in tissue lysates. | BioVision K672-100 |
| Mouse Insulin ELISA Kit | To confirm insulinemic status in plasma from experimental nutritional interventions. | Crystal Chem 90080 |
| Lipid Extraction Kit (Solid Phase) | For high-throughput, reproducible extraction of TAG and other lipids for LC-MS/MS. | Avanti Polar Lipids 791001 |
| C57BL/6J Mice on HFD | Standardized animal model for studying diet-induced obesity and insulin resistance. | Jackson Laboratory Diet-Induced Obesity Model |
This whitepaper evaluates therapeutic targets within triacylglycerol (TAG) synthesis, framed by the contrasting roles of two primary pathways: the canonical Kennedy pathway and the acyl-CoA-independent pathway. The Kennedy pathway is the principal route for de novo glycerolipid synthesis, culminating in the action of two distinct diacylglycerol acyltransferase (DGAT) enzymes. The acyl-CoA-independent pathway, utilizing phospholipids like phosphatidylcholine as acyl donors, represents a critical remodeling route influencing TAG fatty acid composition and lipid signaling. DGAT1 and DGAT2, though both catalyzing the final step in Kennedy-pathway TAG synthesis, are products of separate gene families, exhibit distinct physiological roles, subcellular localizations, and substrate preferences. The validation of DGAT1 as a drug target and the exploration of DGAT2 emerge from this fundamental biological dichotomy, with clinical outcomes directly informing future prospects.
DGAT1 Inhibition: Clinical Trial Outcomes & Validation
DGAT1 inhibition has been pursued primarily for metabolic disorders (obesity, type 2 diabetes) and, more successfully, for rare diseases of intestinal fat metabolism.
2.1. Clinical Trial Data Summary Recent clinical trials have yielded clear outcomes, validating DGAT1's role in intestinal lipid absorption but revealing complexities in systemic metabolism.
Table 1: Summary of Select DGAT1 Inhibitor Clinical Trial Outcomes
| Drug Candidate / Code | Indication Focus | Phase | Key Outcome(s) | Status / Conclusion |
|---|---|---|---|---|
| Pradigastat (LCQ908) | Familial Chylomicronemia Syndrome (FCS) | Phase II | Significant reduction in postprandial triglycerides (-70% to -90%). | Validated. Demonstrated efficacy; further development likely tailored for ultra-rare FCS. |
| AZD7687 | Type 2 Diabetes / Obesity | Phase I | Reduced postprandial TG, but high incidence of gastrointestinal adverse effects (diarrhea, nausea). | Development halted. Target engagement confirmed, but therapeutic window too narrow for chronic systemic use. |
| DGAT1-targeted modalities | Congenital Diarrheal Disorders | Preclinical/Clinical | Proof-of-concept for genetic disorders of enterocyte fat metabolism. | Emerging validation for specific enteropathies. |
2.2. Experimental Protocol: In Vivo Postprandial Triglyceride Response Test This core protocol validates DGAT1 inhibitor efficacy in humans and animal models.
- Subject Preparation: Overnight fast (≥10 hours).
- Baseline Measurement: Plasma collection for baseline triglyceride (TG) quantification (enzymatic colorimetric assay).
- Drug/Placebo Administration: Oral administration of DGAT1 inhibitor or matched placebo.
- High-Fat Challenge Meal: Standardized high-fat meal (e.g., 60-70% calories from fat) administered within 30 minutes of dosing.
- Serial Blood Sampling: Plasma samples collected at frequent intervals (e.g., 2, 4, 6, 8 hours) postprandially.
- Analysis: Measurement of plasma TG in all samples. Calculate area under the curve (AUC) for TG vs. time. Statistical comparison of AUC and peak TG between treatment and placebo groups.
2.3. Interpretation & Validation Conclusion Clinical data robustly validate DGAT1 as a mechanistic target for reducing intestinal fat absorption. Its therapeutic utility, however, is indication-dependent. It is a clinically validated target for rare conditions like FCS, where near-complete inhibition of chylomicron production is desirable and GI side effects are manageable. For common metabolic diseases, chronic systemic inhibition is limited by a mechanism-based intolerability, narrowing its therapeutic index.
Prospects for DGAT2 Targeting
DGAT2 is considered a more attractive target for modulating systemic metabolism. It is genetically linked to hepatic steatosis, primarily associated with lipid droplets in the liver, and is crucial for storing excess energy as TAG. Inhibition is hypothesized to reduce hepatic steatosis and improve insulin sensitivity with potentially fewer intestinal side effects than DGAT1 inhibition.
3.1. Key Differentiating Research Findings
- Hepatic vs. Intestinal Role: Liver-specific Dgat2 knockout profoundly reduces hepatic TAG, while intestinal TAG synthesis is less affected.
- Essential Function: Complete Dgat2 knockout in mice is neonatally lethal due to skin barrier defects and severe lipopenia, suggesting partial inhibition is required for therapeutic safety.
- Pathway Association: DGAT2 operates in a functional complex with other ER proteins favoring de novo fatty acid incorporation into TAG, positioning it centrally in the lipogenic axis.
3.2. Experimental Protocol: Hepatic Lipid Droplet Isolation & Analysis To assess DGAT2 inhibition efficacy, analysis of hepatic lipid droplets is critical.
- Liver Homogenization: Perfuse and mince liver tissue. Homogenize in ice-cold buffer (e.g., 25mM Tris-HCl, pH 7.4, 150mM KCl) with a loose-fitting Dounce homogenizer.
- Differential Centrifugation: Centrifuge homogenate at 1,000 x g (10 min, 4°C) to remove nuclei/cell debris. Transfer supernatant and centrifuge at 20,000 x g (30 min, 4°C) to pellet heavy organelles.
- Buoyant Density Centrifugation: Layer the resulting supernatant over a sterile 0.5M sucrose cushion. Centrifuge at 100,000 x g (60 min, 4°C). Collect the floating lipid droplet layer from the top.
- Washing & Characterization: Wash droplets in homogenization buffer, re-isolate by flotation. Analyze for:
- Lipidomics: Extract TAG and measure composition (LC-MS).
- Protein Analysis: SDS-PAGE/Western blot for DGAT2, perilipins, etc.
- Size/Number: Microscopy (Nile Red staining) or particle analysis.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for DGAT & TAG Synthesis Research
| Reagent / Material | Function / Application | Example & Rationale |
|---|---|---|
| DGAT1-Selective Inhibitors (e.g., T863, A922500) | In vitro and in vivo pharmacological DGAT1 blockade. Used to dissect DGAT1-specific functions in cells and animal models. | A922500: Potent, reversible, competitive inhibitor for acute studies of intestinal fat absorption. |
| DGAT2-Targeting Tools (e.g., PF-06424439, antisense oligonucleotides) | Selective DGAT2 inhibition for proof-of-concept studies. | PF-06424439: Oral, reversible inhibitor used to demonstrate reduction in hepatic steatosis in rodents. |
| Radio/Stable Isotope-Labeled Substrates (e.g., [¹⁴C]glycerol, [¹³C]palmitate) | Tracing flux through Kennedy vs. acyl-independent pathways. | [¹⁴C]Diacylglycerol & [³H]Acyl-CoA: Used in combined assays to differentiate DGAT1 vs. DGAT2 activity based on substrate kinetics. |
| Genetic Models (Knockout mice, siRNA, CRISPR-Cas9) | Definitive target validation by genetic ablation. | Liver-specific Dgat2 KO mice: Establish causal role of hepatic DGAT2 in steatosis without systemic lethality. |
| Lipid Droplet-Specific Dyes (e.g., Nile Red, BODIPY 493/503) | Visualization and quantification of neutral lipid storage in cells/tissues. | Nile Red: Fluorescent dye for rapid imaging and flow cytometry analysis of cellular TAG content. |
Pathway & Conceptual Diagrams
Kennedy vs Acyl-CoA Independent TAG Synthesis
DGAT Inhibitor Clinical Validation Workflow
Clinical outcomes have definitively validated DGAT1 inhibition as a potent modulator of intestinal lipid absorption, carving a niche for rare diseases like FCS. The broader therapeutic failure in common metabolic diseases underscores the pathway-specific biology: systemic DGAT1 inhibition disrupts enterocyte lipid homeostasis intolerably. This pivots the focus toward DGAT2, a target more intimately linked to the hepatic lipogenic flux and the Kennedy pathway's role in energy storage. The future of DGAT2 targeting depends on developing inhibitors with an appropriate safety margin to avoid the liabilities of complete inhibition, potentially for indications like non-alcoholic steatohepatitis (NASH). Future research must continue to dissect the nuanced interplay between the Kennedy and acyl-CoA-independent pathways to identify context-dependent vulnerabilities for therapeutic intervention.
The study of triacylglycerol (TAG) synthesis has long been dominated by two canonical pathways: the Kennedy (glycerol-3-phosphate) pathway and the acyl-CoA-independent (monoacylglycerol) pathway. The broader thesis posits that these are not merely redundant or tissue-specific routes, but components of an integrated, dynamic network. This network exhibits sophisticated regulatory behaviors—cooperation, competition, and compensation—to maintain systemic lipid homeostasis. Disruption of this balance is central to metabolic diseases, making its elucidation critical for therapeutic intervention.
Pathway Architecture and Core Enzymes
Kennedy Pathway: The de novo synthesis pathway, primarily in liver and adipose.
- GPAT (Glycerol-3-phosphate acyltransferase): Commits glycerol-3-phosphate to synthesis.
- AGPAT (1-acylglycerol-3-phosphate O-acyltransferase): Adds second acyl chain.
- PAP/LIPIN: Dephosphorylates to form diacylglycerol (DAG).
- DGAT (Diacylglycerol acyltransferase): Adds third acyl chain to form TAG.
Acyl-CoA-Independent Pathway: Primarily for dietary fat re-synthesis in enterocytes and lipid recycling.
- MGAT (Monoacylglycerol acyltransferase): Acylates monoacylglycerol (MAG) to DAG.
- DGAT: Converts DAG to TAG (shared terminal enzyme).
Quantitative Comparison of Pathway Attributes
| Attribute | Kennedy Pathway | Acyl-CoA-Independent Pathway |
|---|---|---|
| Primary Tissues | Liver, Adipose, Mammary Gland | Small Intestine, Adipose (during lipolysis) |
| Primary Substrate | Glycerol-3-phosphate, Acyl-CoAs | Monoacylglycerol, Acyl-CoAs |
| Key Committed Enzyme | GPAT (4 isoforms) | MGAT (3 isoforms) |
| DGAT Isoform Preference | DGAT1 (high affinity, rev.) & DGAT2 | DGAT1 (high affinity, rev.) |
| Estimated Max. Hepatic Contribution to TAG Pool | ~70% (Fed state) | <10% (Fed state) |
| Estimated Intestinal Contribution to TAG Pool | ~30% (Post-prandial) | ~70% (Post-prandial) |
| Response to Fasting/Refeeding | Highly regulated (inhibited by fasting) | Constitutively active in intestine |
Integrated Regulatory Network: Cooperation, Competition, Compensation
Cooperation
Pathways cooperate by sharing substrates, intermediates, and terminal enzymes.
- Shared DAG Pool: Both pathways converge at DAG, creating a metabolically flexible node.
- Enzyme Complexing: Evidence suggests DGAT1 may exist in complexes with both GPAT and MGAT isoforms for substrate channeling.
Experiment: Co-immunoprecipitation for DGAT1 Complex Analysis
- Objective: Determine physical interaction between DGAT1 and GPAT/MGAT enzymes.
- Protocol:
- Transfect HEK293 cells with plasmids expressing tagged DGAT1 and either GPAT1 or MGAT2.
- Lyse cells in mild, non-denaturing buffer (e.g., 1% CHAPS).
- Incubate lysate with anti-tag antibody (e.g., anti-FLAG M2 affinity gel).
- Wash beads extensively to remove non-specific binding.
- Elute bound proteins and analyze via Western blot for co-precipitating partners.
Competition
Pathways compete for limited cellular resources.
- Acyl-CoA Pool: Both pathways consume acyl-CoAs, creating direct competition under limiting conditions.
- DAG Utilization: DAG is also a precursor for phospholipids (via CDP-choline pathway), introducing inter-pathway competition.
Experiment: Isotopic Tracer & Pharmacological Inhibition to Quantify Flux Competition
- Objective: Measure real-time flux through each pathway under acyl-CoA-limiting conditions.
- Protocol:
- Culture primary hepatocytes in low-glucose, fatty-acid-free medium.
- Treat cells with an acyl-CoA synthetase inhibitor (Triacsin C, 10 µM) to limit acyl-CoA availability.
- Pulse with dual isotopic tracers: ¹⁴C-glycerol (Kennedy pathway precursor) and ³H-monoolein (MGAT pathway precursor).
- Harvest cells at time points (0, 15, 30, 60 min).
- Extract lipids, separate TAG via TLC, and quantify ¹⁴C/³H incorporation via scintillation counting.
- Calculate relative flux ratios to demonstrate competitive dynamics.
Compensation
Genetic or pharmacological disruption of one pathway is compensated for by the other, maintaining homeostasis.
- DGAT1 Knockout: Mice show upregulated intestinal MGAT activity and altered Kennedy pathway enzyme expression in adipose tissue.
- GPAT Knockdown: In hepatocytes, leads to increased utilization of MAG substrates and MGAT2 upregulation.
Experiment: CRISPR-Cas9 Knockout with Rescue Phenotyping
- Objective: Assess compensatory pathway upregulation upon genetic disruption.
- Protocol:
- Generate stable DGAT1 KO HepG2 cell line using CRISPR-Cas9 (sgRNA targeting exon 2).
- Confirm knockout via sequencing and Western blot.
- Subject WT and KO cells to oleate challenge (400 µM, 24h). Perform RNA-seq or qPCR for MGAT2, GPAT, AGPAT, DGAT2.
- Measure cellular TAG accumulation (Oil Red O staining, enzymatic assay).
- Transiently transfect KO cells with DGAT1, MGAT2, or empty vector and repeat oleate challenge to test functional rescue.
Systemic Homeostasis: Inter-Organ Crosstalk
The integrated model extends beyond the cell to whole-body physiology. Adipose tissue Kennedy-derived TAG stores (via lipolysis) provide MAG/FFA for hepatic or cardiac MGAT-driven re-esterification. This creates a "futile cycle" that fine-tunes systemic lipid flux.
Table: Systemic Metabolic Perturbations and Pathway Responses
| Condition | Primary Pathway Affected | Compensatory Response | Homeostatic Outcome |
|---|---|---|---|
| High-Carb Diet (Chow) | Kennedy pathway induced (via SREBP1c) | MGAT activity stable | Hepatic TAG synthesis increases for storage. |
| High-Fat Diet | Intestinal MGAT pathway saturated | Hepatic Kennedy pathway upregulated | Redirects excess dietary fat to adipose storage. |
| DGAT1 Inhibition (Drug) | Intestinal & hepatic TAG synthesis blocked | Adipose tissue lipolysis & MAG pathway flux increase | Limited TAG reduction; potential side effects (steatorrhea). |
| Fasting | Hepatic Kennedy pathway suppressed | Adipose MGAT activity increases for re-esterification | Conserves fatty acids, limits futile cycling. |
Visualizing the Integrated Network
Title: Integrated Network of Kennedy and MAG Pathways for TAG Synthesis
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Tool | Function/Application | Example Product/Catalog # |
|---|---|---|
| DGAT1 Inhibitor (T863) | Pharmacological inhibition of DGAT1 to probe pathway compensation. | Cayman Chemical #13228 |
| MGAT2 Inhibitor (PF-04620110) | Selective inhibition of intestinal MGAT2 to study dietary fat absorption. | MedChemExpress HY-15587 |
| Triacsin C | Inhibits long-chain acyl-CoA synthetases, limiting acyl-CoA pool for competition studies. | Tocris Bioscience #3802 |
| ¹⁴C-Glycerol | Radiolabeled tracer to specifically track flux through the Kennedy pathway. | American Radiolabeled Chemicals ARC-0296 |
| ³H-Monoolein (2-oleoyl) | Radiolabeled tracer to specifically track flux through the MGAT pathway. | American Radiolabeled Chemicals ART-174 |
| Recombinant Human DGAT1 (Sf9) | Purified enzyme for in vitro kinetic assays and inhibitor screening. | Thermo Fisher Scientific #PR-731A |
| GPAT/MGAT Activity Assay Kit | Colorimetric measurement of enzyme activity from tissue lysates. | BioVision #K521 (GPAT), #K1941 (MGAT) |
| SREBP1c siRNA | Knockdown to study transcriptional regulation of Kennedy pathway genes. | Santa Cruz Biotechnology sc-36557 |
| Anti-LIPIN1 Antibody | Immunoblotting to monitor protein levels of key Kennedy pathway enzyme. | Cell Signaling Technology #14906 |
| CRISPR/Cas9 DGAT1 KO Kit | For generating stable knockout cell lines to study compensation. | Santa Cruz Biotechnology sc-400663 |
Conclusion
The Kennedy and acyl-CoA independent pathways represent evolutionarily conserved, functionally distinct routes for TAG synthesis, each with unique enzymatic machinery, regulation, and physiological mandates. While the Kennedy pathway is crucial for coordinated phospholipid and TAG synthesis, the DGAT2/MGAT-centric alternative pathway is specialized for high-capacity neutral lipid storage. Methodological advances have been pivotal in deconvoluting their contributions, though careful experimental design is required to avoid misinterpretation. The comparative validation underscores that DGAT1 and DGAT2 are non-redundant, explaining the distinct metabolic phenotypes of their inhibition. Future research must focus on the dynamic interplay between these pathways in different cell states and disease contexts, particularly in NAFLD progression and cancer metabolism. This knowledge is foundational for developing next-generation, pathway-specific therapeutics that modulate TAG synthesis to treat metabolic diseases without adverse effects, heralding a new era of precision lipidology.