FRET Biosensors: The Definitive Guide to Real-Time Metabolite Detection in Living Cells
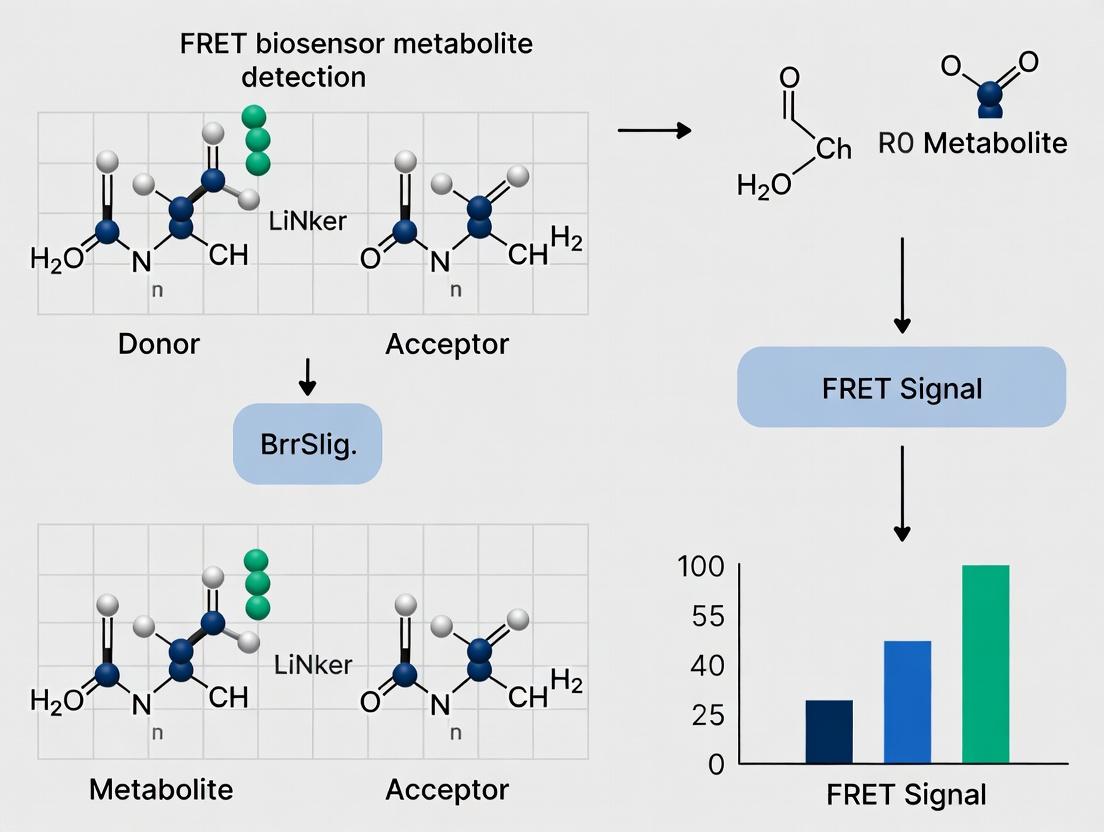
This comprehensive guide explores Förster Resonance Energy Transfer (FRET)-based biosensors, a transformative technology for monitoring metabolite dynamics in live cells.
FRET Biosensors: The Definitive Guide to Real-Time Metabolite Detection in Living Cells
Abstract
This comprehensive guide explores Förster Resonance Energy Transfer (FRET)-based biosensors, a transformative technology for monitoring metabolite dynamics in live cells. We cover the foundational principles of FRET, including the design of genetically encoded indicators with binding domains and fluorescent protein pairs. The article details methodological workflows for implementation, from sensor selection and transfection to quantitative imaging and data analysis. It provides expert troubleshooting advice for common challenges like signal-to-noise ratio and sensor specificity, and critically evaluates validation protocols and comparative performance against other techniques like fluorescent dye-based probes and mass spectrometry. Aimed at researchers and drug development professionals, this resource serves as a practical roadmap for implementing FRET biosensors to uncover metabolic pathways, screen drug candidates, and advance translational research.
What Are FRET Biosensors? Core Principles for Live-Cell Metabolite Imaging
Förster Resonance Energy Transfer (FRET) is a non-radiative process where an excited donor fluorophore transfers energy to a nearby acceptor fluorophore via dipole-dipole coupling. Within the context of FRET biosensor research for metabolite detection, this mechanism serves as a powerful molecular ruler, transducing biochemical events—such as ligand binding, conformational changes, or enzymatic activity—into quantifiable changes in fluorescent emission. This technical guide explores the core physical principles, design strategies for biosensors, and experimental protocols underpinning this critical technology.
Core Principles & Quantitative Framework
The efficiency of FRET (E) is highly sensitive to the distance (r) between the donor and acceptor, described by the Förster equation: E = 1 / [1 + (r/R₀)⁶] where R₀ is the Förster distance at which efficiency is 50%.
Table 1: Key Quantitative Parameters for Common FRET Pairs
| FRET Pair (Donor → Acceptor) | R₀ (nm) | Donor Emission λ (nm) | Acceptor Excitation λ (nm) | Typical Application in Biosensors |
|---|---|---|---|---|
| CFP → YFP (e.g., Cerulean/Venus) | 4.7 - 5.2 | ~475 | ~515 | Ca²⁺, cAMP, kinase activity |
| GFP → mCherry (e.g., Clover/mRuby2) | 5.7 - 6.1 | ~510 | ~587 | Metabolite levels, protease activity |
| Cy3 → Cy5 | 5.0 - 5.5 | ~570 | ~670 | In vitro nucleic acid/protein assays |
| T-Sapphire → mOrange2 | 4.8 | ~495 | ~560 | Ratiometric pH sensing |
FRET Biosensor Design for Metabolite Detection
Metabolite-sensing FRET biosensors typically employ an "affinity clamp" architecture. A metabolite-binding domain is flanked by donor and acceptor fluorescent proteins (FPs). Metabolite binding induces a conformational change that alters the distance/orientation between FPs, modulating FRET efficiency.
Experimental Protocols
Protocol: Ratiometric FRET Measurement in Live Cells
This protocol is for monitoring metabolite dynamics using a genetically encoded FRET biosensor (e.g., a glucose sensor like FLII¹²Pglu-700μδ⁶) expressed in cultured cells.
1. Biosensor Expression:
- Transfect cells with the FRET biosensor plasmid using a standard method (e.g., lipofection, electroporation).
- Allow 24-48 hours for expression. Stable cell lines are preferred for consistency.
2. Microscope Setup:
- Use an inverted epifluorescence or confocal microscope with temperature/CO₂ control.
- Required filter sets:
- Donor excitation / Acceptor emission (FRET channel): Ex: 430-450 nm, Em: 530-550 nm.
- Donor excitation / Donor emission (Donor channel): Ex: 430-450 nm, Em: 470-500 nm.
- Acceptor excitation / Acceptor emission (Acceptor channel): Ex: 500-520 nm, Em: 530-550 nm (controls for acceptor expression and direct excitation).
3. Image Acquisition & Analysis:
- Acquire time-lapse images in all three channels with minimal exposure to reduce photobleaching.
- Calculate the FRET ratio (R) for each pixel/timepoint: R = I_FRET / I_Donor.
- Correct for bleed-through (donor emission in FRET channel) and direct excitation of acceptor using control cells expressing donor or acceptor alone.
- Normalize the ratio (R/R₀) to the baseline period.
Protocol:In VitroFRET Assay for Metabolite Quantification
This protocol uses purified biosensor protein for high-throughput screening of metabolites.
1. Protein Purification:
- Express His-tagged biosensor in E. coli and purify via Ni-NTA affinity chromatography.
- Dialyze into assay buffer (e.g., 20 mM HEPES, pH 7.4, 150 mM KCl).
2. Plate Reader Assay:
- In a black 384-well plate, add 50 μL of purified biosensor (100-200 nM final).
- Add 50 μL of buffer (for standard curve) or sample containing metabolite.
- Incubate for 5-10 minutes at room temperature.
- Read fluorescence using dual excitations/emissions:
- Ex 433 nm / Em 475 nm (Donor).
- Ex 433 nm / Em 528 nm (FRET).
- Calculate the FRET/Donor emission ratio. Generate a standard curve with known metabolite concentrations to interpolate sample values.
Data Analysis & Validation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for FRET Biosensor Research
| Item | Function & Relevance |
|---|---|
| Genetically Encoded FRET Pairs (e.g., mTurquoise2/sYFP2, Clover/mRuby2) | Optimized FP pairs with high quantum yield, photostability, and well-separated spectra for robust FRET. |
| Modular Biosensor Backbones (e.g., pRSET, pcDNA3.1 with flexible linkers) | Vectors for cloning and expressing custom biosensors with donor, acceptor, and sensing domains. |
| FRET Calibration Standards (e.g., linked CFP-YFP constructs with known distances) | Controls for determining microscope-specific R₀ and validating FRET measurement setup. |
| Spectral Unmixing Software (e.g., in NIS-Elements, MetaMorph, or Fiji/ImageJ plugins) | Essential for accurate bleed-through correction and ratiometric calculation from raw image data. |
| Microfluidic Perfusion Systems (e.g., from CellASIC or Ibidi) | Enables precise, rapid changes in extracellular metabolite concentration for dynamic biosensor characterization. |
| Quenched Substrate FRET Peptides (e.g., for caspases, kinases) | Cleavage or phosphorylation changes FRET; used for enzyme activity assays in drug screening. |
Within the broader pursuit of understanding cellular metabolism in health and disease, the development of genetically encoded FRET (Förster Resonance Energy Transfer) biosensors represents a pivotal technological thesis. These tools enable the real-time, subcellular detection of metabolites, ions, and signaling events in living systems, directly informing drug discovery and basic research. This whitepaper deconstructs the core anatomy of these biosensors, focusing on the critical interplay between the binding domain, donor, and acceptor pairs.
Core Architectural Components
A genetically encoded FRET biosensor is a single polypeptide chain integrating three essential modules:
- Sensing/Binding Domain: A protein domain that undergoes a conformational change upon binding the target analyte (e.g., a metabolite, Ca²⁺, kinase activity). This domain is typically sourced from bacterial periplasmic binding proteins or endogenous signaling proteins.
- Donor Fluorophore: A fluorescent protein (e.g., ECFP, mCerulean3, mTurquoise2) that emits light at a specific wavelength when excited.
- Acceptor Fluorophore: A second fluorescent protein (e.g., EYFP, mVenus, mCitrine, cpGFP) with an absorption spectrum that overlaps the donor's emission spectrum. Its fluorescence is excited via FRET from the donor.
The central principle is that the analyte-induced conformational change in the binding domain alters the distance and/or orientation between the donor and acceptor fluorophores, thereby modulating FRET efficiency. This change is measured as a ratio of acceptor-to-donor emission intensity, providing a quantitative, internally controlled signal.
Quantitative Comparison of Common FRET Pairs
The choice of donor-acceptor pair is critical for sensor performance. Key metrics include brightness, photostability, maturation time, and the Förster radius (R₀), the distance at which FRET efficiency is 50%.
Table 1: Characteristics of Common Genetically Encoded FRET Pairs
| Donor Fluorophore | Acceptor Fluoroprotein | R₀ (Å) ~ | Brightness (Relative) | Maturation Rate | pKa | Primary Application |
|---|---|---|---|---|---|---|
| ECFP | EYFP | 49.2 | Moderate | Moderate | 4.7 | Early-generation sensors (e.g., Cameleons) |
| mTurquoise2 | mVenus | 59.5 | High | Fast | 3.1 | High dynamic range, pH-stable sensors |
| mCerulean3 | mCitrine | 53.0 | High | Fast | 3.1 | Improved brightness over ECFP/EYFP |
| mCyRFP1 | mMaroon1 | 64.0 | High | Moderate | 4.5 | Red-shifted, for deep-tissue imaging |
| Clover | mRuby2 | 62.0 | Very High | Moderate | 5.4 | High-brightness, red-shifted pair |
Note: R₀ and brightness values are approximate and can vary based on protein environment and measurement conditions.
Detailed Experimental Protocol: FRET Biosensor CalibrationIn Vitro
The following protocol is essential for characterizing a newly developed biosensor before cellular expression.
Protocol: In Vitro Purification and Titration of a FRET Biosensor
Molecular Cloning & Expression:
- Clone the biosensor gene into a prokaryotic expression vector (e.g., pRSET, pET) with an N-terminal His₆-tag.
- Transform into E. coli BL21(DE3) cells. Induce expression with 0.5 mM IPTG at 16°C for 18-20 hours.
- Harvest cells by centrifugation (5,000 x g, 15 min, 4°C).
Protein Purification:
- Lyse cells using sonication or French press in Lysis Buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 10 mM imidazole, 1 mM PMSF).
- Clarify lysate by centrifugation (20,000 x g, 45 min, 4°C).
- Purify the His-tagged protein from the supernatant using Ni-NTA affinity chromatography. Elute with an imidazole gradient (50-500 mM).
- Perform buffer exchange into Assay Buffer (e.g., 30 mM HEPES pH 7.4, 100 mM KCl) using a desalting column. Determine concentration via absorbance at 280 nm.
Spectroscopic Characterization & Titration:
- Using a fluorometer, obtain the excitation and emission spectra of the purified biosensor in analyte-free buffer. Excite the donor (e.g., at 433 nm for CFP variants) and collect emission from 450-600 nm.
- Prepare a dilution series of the target analyte (metabolite) covering a range from 0 to saturating concentrations.
- For each concentration, incubate a fixed amount of biosensor (e.g., 200 nM) with the analyte for 5 minutes at room temperature.
- Acquire the emission spectrum for each sample upon donor excitation.
- Data Analysis: Calculate the emission ratio (Acceptor emission peak / Donor emission peak). Plot the ratio against analyte concentration and fit the data with a sigmoidal (Hill) curve to determine the apparent dissociation constant (Kd), dynamic range (ΔR/R₀), and operational range.
Signaling Pathway & Biosensor Function Logic
Title: FRET Biosensor Activation Logic
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for FRET Biosensor Development & Use
| Reagent / Material | Function & Purpose |
|---|---|
| High-Fidelity DNA Polymerase (e.g., Q5) | For error-free PCR during biosensor vector construction and mutagenesis. |
| HEK293T/HeLa Cell Lines | Standard mammalian cell lines for initial biosensor characterization due to high transfection efficiency. |
| Polyethylenimine (PEI) Transfection Reagent | Cost-effective chemical transfection method for plasmid DNA delivery into mammalian cells. |
| Glass-Bottom Imaging Dishes | Provide optimal optical clarity for high-resolution live-cell fluorescence microscopy. |
| Phenol Red-Free Imaging Medium | Cell culture medium without fluorescent compounds that interfere with emission detection. |
| Ionomycin/A23187 (Ca²⁺ sensors) | Calcium ionophore used as a positive control to saturate Ca²⁺-sensitive biosensors (e.g., Cameleon). |
| Digitonin/Mild Detergents | For cell permeabilization in calibration protocols to introduce controlled analyte concentrations. |
| Recombinant Protein Ladder & SDS-PAGE Gels | For assessing the purity and molecular weight of purified biosensor protein. |
| Imidazole (for His-tag purification) | Competitive eluent for purifying His-tagged biosensor proteins from Ni-NTA resin. |
| CO₂-Independent Medium | For extended live-cell imaging sessions outside a controlled CO₂ incubator. |
Experimental Workflow for Cellular Validation
Title: Cellular FRET Biosensor Validation Workflow
The continued refinement of these core components—through engineering of brighter, faster-maturing fluorophores and more sensitive, specific binding domains—directly advances the central thesis of FRET-based metabolite detection research. This enables the precise dissection of metabolic fluxes in vivo, accelerates the screening of metabolic modulators, and ultimately provides a dynamic window into the pathophysiology targeted by next-generation therapeutics.
Fluorescence Resonance Energy Transfer (FRET) biosensors represent a cornerstone technology for the real-time, subcellular quantification of metabolites in living cells and tissues. This technical guide focuses on the detection of six critical target metabolite classes—glucose, ATP, cAMP, glutamate, lipids, and ions—within the broader thesis that advancing FRET-based detection is pivotal for elucidating metabolic signaling networks and accelerating drug discovery. These biosensors typically consist of a sensing domain specific to the metabolite, flanked by a pair of fluorescent proteins (e.g., CFP/YFP). Metabolite binding induces a conformational change that alters FRET efficiency, providing a quantifiable ratiometric signal.
Table 1: Key Physiologic and Biosensor Performance Metrics for Target Metabolites
| Metabolite Class | Key Physiologic Range | Representative FRET Biosensor(s) | Reported Kd / Dynamic Range | Typical Cellular Compartment |
|---|---|---|---|---|
| Glucose | 3-10 mM (blood) | FLII12Pglu-700μδ6 | Kd: ~3.9 mM | Cytosol |
| ATP | 1-10 mM (cytosol) | ATeam1.03, QUEEN-2m | Kd: ~3.3 mM (ATeam) | Cytosol, Mitochondria |
| cAMP | 0.1-10 μM (basal/peak) | Epac1-camps, ICUE3 | Kd: ~9.5 μM (Epac1) | Cytosol, Microdomains |
| Glutamate | 1-10 mM (synaptic cleft) | iGluSnFR, GluSnFR | Not Applicable (Single FP) | Extracellular, Synaptic |
| Lipids (PIP3) | Low nM - μM | AktPH-FRET, PIP3 Biosensor | N/A (PH domain binding) | Plasma Membrane |
| Ions (Ca²⁺) | ~100 nM (resting), >1 μM (active) | YC3.6, TN-XXL | Kd: ~250 nM (YC3.6) | Cytosol, ER, Nucleus |
| Ions (H⁺ / pH) | pH 4.5-7.4 (lysosome-cytosol) | pHluorin-based sensors | pKa tuned to range | Lysosome, Golgi, Cytosol |
Table 2: Comparison of FRET Pair Properties for Common Biosensor Constructs
| FRET Pair (Donor->Acceptor) | Excitation (nm) | Emission (Acceptor, nm) | Benefits | Common Use |
|---|---|---|---|---|
| CFP->YFP (e.g., Cerulean->Venus) | ~433 | ~528 | Established, high FRET efficiency | cAMP, Ca²⁺, Protease activity |
| GFP->RFP (e.g., Clover->mRuby2) | ~472 | ~605 | Reduced spectral crosstalk, photostability | Multiplexing, deep-tissue imaging |
| BFP->GFP (e.g., mTagBFP->GFP) | ~399 | ~510 | Large Stokes shift | Specialized multiplex applications |
| Teal->Orange (e.g., mTFP1->mOrange) | ~462 | ~562 | Bright, photostable | High-signal environments |
Experimental Protocols for FRET Biosensor Imaging
Protocol 1: Live-Cell FRET Imaging for Cytosolic Metabolites (e.g., Glucose, ATP)
- Cell Preparation: Plate cells (e.g., HEK293, HeLa) on 35mm glass-bottom dishes. At 60-80% confluency, transfect with plasmid encoding the FRET biosensor (e.g., FLII12Pglu-700μδ6 for glucose) using a suitable transfection reagent (e.g., Lipofectamine 3000). Incubate for 24-48 hours.
- Imaging Setup: Use an inverted epifluorescence or confocal microscope equipped with a temperature (37°C) and CO₂ (5%) control chamber. Required filter sets: Donor excitation/emission (e.g., CFP: Ex 430/25, Em 470/24), Acceptor emission (e.g., YFP: Ex 430/25, Em 535/30), and FRET channel (Ex 430/25, Em 535/30). A 40x or 60x oil-immersion objective is recommended.
- Image Acquisition: Acquire time-lapse images sequentially through donor, FRET, and acceptor channels. Use minimal exposure time and light intensity to avoid phototoxicity and photobleaching. Include a positive control (e.g., saturating glucose) and negative control (e.g., glucose-free medium).
- FRET Ratio Calculation: After background subtraction, calculate the FRET ratio (R) for each time point (t) and region of interest (ROI) as: R(t) = IntensityFRET(t) / IntensityDonor(t). Normalize ratios to the baseline (R/R₀) for presentation.
Protocol 2: Calibration of Ion Biosensors (e.g., Ca²⁺, pH) In Situ
- Generation of Calibration Curve: For Ca²⁺ biosensors (e.g., YC3.6), treat cells at the end of the experiment with ionophores to clamp intracellular Ca²⁺ at known concentrations. A standard protocol: Apply 10 μM ionomycin in calibration buffers containing high-Ca²⁺ (e.g., 10 mM CaCl₂ to saturate sensor) and zero-Ca²⁺ (e.g., 10 mM EGTA).
- Image Acquisition: Capture donor and FRET channel images at each clamped condition.
- Data Analysis: Plot the calculated FRET ratio (R) against the known free [Ca²⁺] for each buffer. Fit the data to the Hill equation: [Ca²⁺] = Kd * ( (R - Rmin) / (Rmax - R) )^(1/n), where Rmin and Rmax are the minimum and maximum ratio values, Kd is the dissociation constant, and n is the Hill coefficient. Apply this curve to convert experimental ratio values to estimated [Ca²⁺].
Visualization: Pathways and Workflows
FRET Biosensor Detection of Metabolic Signaling
FRET Biosensor Imaging Workflow
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Reagents for FRET-Based Metabolite Detection Research
| Item | Function & Explanation |
|---|---|
| FRET Biosensor Plasmids | Genetically encoded constructs (e.g., from Addgene) containing the sensing domain and donor/acceptor FP pair. The core research material. |
| High-Quality Cell Culture Media | Defined, phenol-red-free medium to minimize background autofluorescence during live-cell imaging. |
| Transfection Reagents (e.g., Lipofectamine, PEI) | For efficient, low-toxicity delivery of biosensor plasmids into mammalian cell lines. |
| Ionophores & Calibration Kits (e.g., Ionomycin, A23187) | Essential for in situ calibration of ion and metabolite biosensors to convert ratio to concentration. |
| Metabolite Analogs & Modulators (e.g., 2-DG, Forskolin, Thapsigargin) | Pharmacological tools to manipulate cellular metabolite levels for control experiments and validation. |
| Mounting Media with Index-Matching Properties | For fixed-sample imaging, reduces light scattering and improves signal-to-noise ratio. |
| Imaging Chamber with Environmental Control | Maintains cells at 37°C and 5% CO₂ during long-term live imaging, preserving physiological relevance. |
| Immersion Oil (Type F or similar) | High-quality, non-fluorescent oil matching the refractive index of the objective lens and coverslip. |
The study of cellular metabolism using FRET (Förster Resonance Energy Transfer) biosensors represents a paradigm shift from static snapshots to dynamic, living system analysis. Traditional fixed-cell immunoassays (e.g., immunohistochemistry) and destructive endpoint assays (e.g., LC-MS of lysed samples) provide single-time-point data, averaging signals across cell populations and destroying spatial context. Within the thesis framework of advancing metabolite detection, the core advantage of live-cell FRET biosensing lies in its capacity to deliver quantitative, real-time, and spatially resolved kinetic data of metabolite fluxes within single cells, unveiling heterogeneity and transient dynamics invisible to conventional methods.
Core Advantages: A Quantitative Comparison
The following table summarizes the comparative advantages of live-cell FRET imaging over fixed and destructive assays.
Table 1: Comparative Analysis of Metabolite Detection Methodologies
| Feature | Live-Cell FRET Biosensor Imaging | Fixed-Cell Assays (e.g., IHC) | Destructive Assays (e.g., LC-MS, ELISA) |
|---|---|---|---|
| Temporal Resolution | Seconds to minutes. Continuous monitoring over hours/days. | Single time point. Requires sample fixation at predetermined endpoint. | Single time point. Sample destruction prevents longitudinal study. |
| Spatial Resolution | Subcellular compartmentalization. Can target cytosol, nucleus, organelles (e.g., mito- or nucleo-specific biosensors). | Cellular/subcellular, but artifacts from fixation/permeabilization possible. | None (bulk analysis) or limited (subcellular fractionation is laborious and prone to cross-contamination). |
| Data Type | Kinetic traces of metabolite concentration ([Metabolite] vs. Time). Quantitative ratio-metric (R) data. | Static, semi-quantitative intensity at fixation moment. | Absolute quantitative concentration from a lysate pool. |
| Cellular Context | Live, functioning cells. Measures dynamics in intact physiology. | Fixed, dead cells. Potential for epitope masking or alteration. | Lysed cells. No cellular integrity or spatial information. |
| Throughput Potential | Medium to High (with automated microscopy & multi-well plates). | High (for endpoint screening). | Very High (for population-average biochemistry). |
| Key Advantage for Metabolism Research | Reveals metabolic flux & heterogeneity. Directly observes transient spikes, oscillations, and cell-to-cell variability in metabolite levels. | Provides histological context. Useful for correlating metabolite presence with morphology or marker expression at an endpoint. | High sensitivity & specificity. Gold standard for absolute, validated quantification of metabolite pools. |
| Primary Limitation | Requires biosensor development/validation. Phototoxicity/bleaching during long-term imaging. | No dynamic data. Possible fixation artifacts. Antibody specificity required. | No dynamic or single-cell spatial data. Population averaging masks heterogeneity. |
Experimental Protocol: Real-Time FRET Imaging of Glucose Dynamics
This protocol details a key experiment demonstrating the core advantages, using a genetically encoded FRET biosensor for glucose (e.g., FLII12Pglu-700μδ6).
A. Materials & Cell Preparation
- Biosensor: HEK293T or relevant cell line transfected with plasmid encoding the glucose FRET biosensor.
- Imaging Setup: Inverted epifluorescence or confocal microscope with environmental chamber (37°C, 5% CO₂), appropriate filter sets for CFP (Donor) and YFP (Acceptor) emission, and a high-sensitivity camera.
- Perfusion System: For rapid buffer exchange.
- Imaging Buffer: Hanks' Balanced Salt Solution (HBSS) or physiological saline.
- Stimuli: High glucose buffer (e.g., 20mM), glycolysis inhibitor (e.g., 2-Deoxy-D-glucose, 2-DG, 50mM).
B. Image Acquisition Protocol
- Calibration: Acquire images of cells expressing biosensor alone (donor channel: CFP ex/CFP em) and acceptor alone (YFP ex/YFP em) for spectral bleed-through correction.
- Baseline Acquisition: Mount culture dish on microscope. Perfuse with low/no-glucose imaging buffer. Acquire time-lapse images of donor and FRET (acceptor) channels every 30 seconds for 5-10 minutes to establish a stable baseline ratio (R = FRET intensity / Donor Intensity).
- Stimulation: Rapidly switch perfusion to buffer containing 20mM glucose. Continue time-lapse acquisition for 15-20 minutes. Observe the decrease in FRET ratio (for FLII12Pglu-700μδ6) as glucose levels rise.
- Inhibition/Depletion Challenge: Switch to buffer containing 50mM 2-DG to inhibit glycolysis and deplete intracellular glucose. Continue acquisition to observe the ratio recovery/increase.
- Control: Perform parallel experiment on untransfected cells to account for autofluorescence changes.
C. Data Analysis
- Ratio Calculation: For each time point and cell/Region of Interest (ROI), calculate the background-subtracted, bleed-through-corrected FRET ratio (R).
- Normalization: Normalize ratios to the initial baseline average (R/R₀) to pool data from multiple cells.
- Kinetic Plotting: Plot normalized ratio (R/R₀) vs. time for individual cells to visualize heterogeneity, or average for population response.
- Spatial Mapping: Generate pseudocolor ratio images at key time points (baseline, peak glucose, post-2-DG) to visualize spatial gradients within a single cell.
Visualizing the Workflow and Signaling Context
Diagram 1: FRET Biosensor Real-Time Imaging Workflow
Diagram 2: FRET Biosensor Mechanism & Metabolic Context
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for Live-Cell FRET Metabolite Sensing
| Item | Function & Relevance | Example/Notes |
|---|---|---|
| Genetically Encoded FRET Biosensor Plasmid | Core reagent. Encodes the metabolite-specific sensing protein. Must be validated for specificity and dynamic range. | FLII12Pglu-700μδ6 (Glucose), AT1.03 (ATP), iNap series (NAD+, ATP). |
| Transfection Reagent | Delivers biosensor plasmid into target cells for transient expression. | Lipofectamine 3000, polyethylenimine (PEI), or electroporation systems. |
| Cell Culture Media & Supplements | Maintain cell health during transfection and imaging. Phenol-red free media is used for imaging. | DMEM, Opti-MEM, charcoal-stripped FBS to reduce autofluorescence. |
| Live-Cell Imaging Buffer | Physiologically balanced salt solution for maintaining cell viability during perfusion and imaging. | HBSS or Ringer's solution, often with 20mM HEPES for pH stabilization without CO₂. |
| Metabolic Agonists/Antagonists | To perturb the metabolic network and elicit biosensor response, validating its functionality. | Glucose (agonist), 2-Deoxy-D-glucose (glycolysis inhibitor), Oligomycin (ATP synthase inhibitor). |
| Fluorophore-Specific Filter Sets | Microscope optical filters to isolate donor excitation/emission and FRET (acceptor) emission. | CFP/YFP FRET set: CFP ex: 436/20, emission split with a beamsplitter (e.g., 455DCLP) to CFP em: 480/40 and YFP em: 535/30. |
| Environmental Control System | Maintains physiological conditions (37°C, 5% CO₂, humidity) on microscope stage for long-term live-cell integrity. | Microscope stage-top incubator or full environmental enclosure. |
| Image Analysis Software | For background subtraction, ratio calculation, kinetic trace extraction, and spatial mapping. | Fiji/ImageJ (with Ratio Plus plugin), MetaMorph, CellProfiler. |
This technical guide is framed within the context of a broader thesis focused on advancing metabolite detection research using Förster Resonance Energy Transfer (FRET) biosensors. The ability to monitor metabolites with high spatiotemporal resolution in living cells is pivotal for understanding metabolic flux, signaling dynamics, and for drug discovery. The evolution of FRET biosensor design—from early, simple rationetric probes to sophisticated, circularly permuted variants—represents a critical technological progression that has dramatically enhanced sensitivity, dynamic range, and specificity. This whitepaper details this technical evolution, providing the methodologies and tools essential for researchers and drug development professionals working at the forefront of this field.
The Core Principle of FRET
FRET is a distance-dependent (typically 1-10 nm) physical process where energy from an excited donor fluorophore is non-radiatively transferred to an acceptor fluorophore. The efficiency of FRET (E) is inversely proportional to the sixth power of the distance between the donor and acceptor, making it an exquisitely sensitive molecular ruler. In biosensors, ligand binding or a enzymatic event induces a conformational change in a sensing domain, which alters the distance/orientation between the donor and acceptor, thereby modulating FRET efficiency. This change is detected as a shift in the emission ratio of acceptor to donor fluorescence.
Generational Evolution of FRET Biosensor Architecture
First Generation: Simple Rationetric, Dual-Fluorophore Probes
These early designs directly flanked a single sensing domain with donor and acceptor fluorophores (e.g., CFP and YFP). Binding-induced conformational changes were often small, leading to modest dynamic ranges (typically 10-30% ΔR/R).
Key Experiment Protocol: Measuring cAMP with Early FRET Sensor (e.g., FICRhR)
- Sensor Expression: Transfect mammalian cells with plasmid encoding the CFP-sensing domain-YFP construct.
- Imaging Setup: Use a widefield or confocal microscope equipped with:
- A 440 nm laser or filter for CFP excitation.
- Dual emission filters: 470-500 nm for CFP (donor) and 520-550 nm for YFP (acceptor).
- A dichroic mirror suitable for CFP/YFP FRET pairs.
- Rationetric Imaging: Acquire time-lapse images of donor and acceptor channels simultaneously or sequentially.
- Calibration & Stimulation: Acquire a baseline ratio (R = YFP emission / CFP emission). Apply forskolin (50 µM) to elevate cellular cAMP. Apply a saturating dose of a membrane-permeable cAMP analog (8-Br-cAMP, 500 µM) for maximum response.
- Data Analysis: Calculate ΔR/R = (R - R0) / R0, where R0 is the baseline ratio.
Second Generation: Incorporation of Phosphorylation/Dephosphorylation Sensing
These sensors integrated phosphorylation-specific binding domains (e.g., 14-3-3τ, FHA2) that bound to the sensing domain only upon phosphorylation, amplifying the conformational change. This improved dynamic range for kinase activity sensors.
Key Experiment Protocol: Monitoring ERK Activity with EKAR Sensor
- Cell Preparation: Seed cells expressing the EKAR biosensor (CFP-14-3-3τ-substrate peptide-YFP with a docking domain).
- Serum Starvation: Starve cells in 0.1% FBS media for 12-18 hours to reduce basal ERK activity.
- Imaging: Establish baseline FRET ratio. Stimulate with EGF (100 ng/mL) to activate the MAPK/ERK pathway.
- Inhibition Control: Pre-treat a separate group of cells with the MEK inhibitor U0126 (10 µM) for 1 hour prior to EGF stimulation to block ERK phosphorylation.
- Quantification: Analyze the temporal dynamics of the FRET ratio change, correlating peak amplitude with ERK activation strength.
Third Generation: Circularly Permuted Fluorescent Proteins (cpFPs)
This breakthrough involved creating cpFPs by connecting the original N- and C-termini with a short linker and creating new termini at a location near the chromophore. Ligand binding to a fused sensing domain now directly affects the chromophore environment, causing a large change in fluorescence intensity of a single fluorophore. These are often used in "single-FP, intensiometric" sensors or paired with a second, static FP to create a highly responsive FRET pair.
Key Experiment Protocol: Detecting Glutamate with iGluSnFR3
- Sensor Expression: Express the iGluSnFR3 (a cpGFP-based intensiometric sensor) in neurons via viral transduction or transfection.
- Perfusion Setup: Use a fast perfusion system on an epifluorescence or TIRF microscope. Excite at ~480 nm, collect emission >510 nm.
- Calibration: Perfuse with aCSF (Artificial Cerebrospinal Fluid) for baseline, then apply a saturating dose of glutamate (1 mM) in aCSF to obtain F_max.
- Application: Apply synaptic stimulation (electrical or optogenetic) or puff known concentrations of glutamate (e.g., 10 µM, 100 µM).
- Analysis: Calculate ΔF/F0 = (F - F0) / F0. The kinetics of the fluorescence trace directly report glutamate binding and unbinding.
Modern cpFP-Based FRET Biosensors
The most sensitive contemporary designs combine cpFPs as donors with conventional FPs as acceptors. The cpFP's large intensity change upon binding synergizes with FRET, resulting in exceptionally high dynamic range (>100% ΔR/R).
Key Experiment Protocol: Measuring ATP:ADP Ratio with QUEEN Sensors or PERplexity
- Cytosolic Expression: Express the sensor (e.g., QUEEN-2m, a cpEGFP-mTFP1 FRET pair sandwiching the Mg-binding protein MgtE) in target cells.
- Dual-Emission Imaging: Excite the cpEGFP donor at ~430 nm. Collect emissions at 470-500 nm (donor) and 520-550 nm (acceptor, mTFP1).
- Metabolic Perturbation:
- Inhibit ATP Synthesis: Treat with 2-deoxy-D-glucose (10 mM) and antimycin A (1 µM) to lower ATP:ADP.
- Promote ATP Synthesis: Treat with glucose (25 mM) in energy-depleted cells.
- In Situ Calibration: Permeabilize cells with digitonin (10-20 µM) and perfuse with calibration buffers containing known [ATP] and [ADP] to establish a standard curve for the FRET ratio.
- Data Conversion: Convert the acquired FRET ratio to ATP:ADP ratio using the established calibration curve.
Quantitative Comparison of FRET Biosensor Generations
The following table summarizes key performance metrics for representative biosensors from each generation.
Table 1: Quantitative Comparison of FRET Biosensor Generations
| Generation | Example Sensor | Target | Architecture | Dynamic Range (ΔR/R or ΔF/F0) | Response Time (t1/2) | Key Advantage | Primary Limitation |
|---|---|---|---|---|---|---|---|
| 1st: Simple Rationetric | FICRhR | cAMP | CFP-PKA regulatory domain-YFP | ~15-20% | Seconds to minutes | Simple design, rationetric (reduces artifacts) | Low dynamic range, prone to pH sensitivity |
| 2nd: Phospho-Sensing | EKAR | ERK Kinase Activity | CFP-14-3-3τ-substrate-YFP | ~25-35% | Minutes | Amplified response, good for kinases | Slower due to phosphorylation/docking kinetics |
| 3rd: cpFP Intensiometric | iGluSnFR3 | Glutamate | cpGFP fused to Glu binding protein | ~300-500% (ΔF/F0) | Milliseconds | Very high brightness & dynamic range, fast | Intensiometric (sensitive to artifacts, expression level) |
| Modern: cpFRET | PERplexity (AT1.03) | ATP:ADP Ratio | cpEGFP-MgtE-mTFP1 | >200% (ΔR/R) | Seconds | Extremely high dynamic range, rationetric, quantitative | More complex design, requires careful calibration |
Signaling Pathway and Experimental Workflow Visualizations
Diagram Title: FRET Biosensor Activation via Kinase Signaling
Diagram Title: FRET Biosensor Experimental Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for FRET Biosensor Research
| Item / Reagent | Function / Purpose | Example Product / Note |
|---|---|---|
| Fluorescent Protein (FP) Plasmids | Donor and acceptor fluorophores for sensor construction. | Addgene repositories: mTurquoise2 (donor), cpEGFP variants, mVenus/mCitrine (acceptor). |
| Molecular Biology Kits | Cloning, mutagenesis, and assembly of complex biosensor constructs. | Gibson Assembly Master Mix, Site-Directed Mutagenesis Kits, High-Fidelity DNA Polymerase. |
| Cell Culture Reagents | Maintaining and transfecting mammalian cell lines. | DMEM/F12 media, Fetal Bovine Serum (FBS), Lipofectamine 3000 or PEI for transfection. |
| Imaging Media | Physiologically stable media for live-cell imaging without background fluorescence. | Hanks' Balanced Salt Solution (HBSS) with 20 mM HEPES, pH 7.4. |
| Pharmacological Agonists/Antagonists | To stimulate or inhibit specific pathways for sensor validation. | Forskolin (cAMP), EGF (ERK), Ionomycin (Ca2+), U0126 (MEK inhibitor), Staurosporine (kinase inhibitor). |
| Metabolite Standards | For in vitro and in situ calibration of metabolite sensors. | High-purity ATP, ADP, glutamate, glucose, etc., prepared in calibration buffers. |
| Permeabilization Agent | Allows controlled access of calibration standards to cytosolic sensors. | Digitonin (low concentration, e.g., 10-20 µM) or saponin. |
| Microscope Filter Sets | Specific excitation/emission filters for FRET pairs. | CFP/YFP FRET filter set (e.g., Ex: 430/24, Em: 475/24 & 535/22, Dichroic: 458). |
| Image Analysis Software | For rationetric calculation, time-series analysis, and quantification. | Fiji/ImageJ with RatioPlus plugin, Metamorph, Nikon NIS-Elements, or custom Python/Matlab scripts. |
| Genetically Encoded Biosensor | The final integrated tool for detection. | Commercial sensors available (e.g., Cyto-roGFP for redox), but most are shared via Addgene. |
Implementing FRET Biosensors: A Step-by-Step Protocol from Lab to Discovery
Within the field of FRET-based biosensor research for metabolite detection, selecting the optimal sensor construct is paramount. The choice dictates sensitivity, specificity, temporal resolution, and applicability in complex biological systems. This guide provides an in-depth technical analysis of prominent sensor families, including ATeam, iGLIM, and Snifits, framing their utility within the broader thesis of advancing quantitative, real-time metabolic imaging in live cells for fundamental research and drug development.
Core Sensor Constructs: Mechanisms and Characteristics
ATeam (Adenosine 5'-triphosphate indicator based on Epsilon subunit)
ATeam sensors are intensiometric FRET biosensors for ATP:ADP ratio. They utilize the bacterial F0F1-ATP synthase ε subunit, which undergoes a conformational change upon ATP binding, linked between cyan (CFP) and yellow (YFP) fluorescent proteins.
iGLIM (Improved Green-Light-Induced Dimerizers)
iGLIM is not a metabolite sensor per se but a toolkit for constructing sensors. It employs light-inducible dimerizers (PhyB/PIF) to control the assembly of metabolic enzymes or sensor components with high spatiotemporal precision, enabling user-defined manipulation of metabolic pathways and subsequent detection.
Snifits (Saccharide-Nutrient-Induced FRET-based Indicators)
Snifits are single-wavelength FRET biosensors for sugars like glucose and sucrose. They employ bacterial periplasmic binding proteins (PBPs) that undergo a hinge-twist motion upon ligand binding, coupled to a single fluorescent protein and a quenching dye or a second FP for rationetric measurement.
Quantitative Comparison of Key Sensor Constructs
The following table summarizes the core characteristics of these and related constructs for metabolite detection.
Title: Decision Logic for Selecting a FRET Biosensor Construct
| Sensor Construct | Primary Target | Detection Mode | Dynamic Range (ΔR/R or %) | Affinity (Kd or EC₅₀) | Key Advantages | Primary Limitations |
|---|---|---|---|---|---|---|
| ATeam (AT1.03) | ATP:ADP Ratio | Rationetric FRET (YFP/CFP) | ~1.4 (ΔR/R₀) | N/A (Reports Ratio) | Reports energy charge; multiple affinity variants (e.g., AT1.03YEMK (low), AT1.03NL (high)). | pH sensitive; susceptible to photobleaching; large size. |
| iGLIM | N/A (Toolkit) | Light-Induced Dimerization | N/A | N/A | Unparalleled spatiotemporal control; can be used to build or recruit custom sensors. | Requires exogenous chromophore (phycocyanobilin); complex initial setup. |
| Snifit (e.g., FLIPglu) | Glucose | Rationetric FRET (YFP/CFP) | ~25% (ΔR/R₀) | ~3 μM (FLIPglu-600μM) | High specificity; multiple affinity variants available. | Potentially slow kinetics; may be affected by endogenous binding proteins. |
| QUEEN | ATP | Single FP Intensity | ~5.0 (F/F₀) | ~3.1 mM | Intensiometric, simpler imaging; resistant to pH changes. | No rationetric correction; single-wavelength. |
| SoNar | NAD+/NADH Ratio | Rationetric (YFP/CFP) | ~9.0 (F/F₀) | N/A (Reports Ratio) | Extremely high dynamic range; sensitive to redox status. | Highly oxygen-sensitive; requires careful calibration. |
Experimental Protocols for Key Validations
Protocol 1: Characterizing Sensor ResponseIn Vitro
Objective: Determine the affinity (Kd) and dynamic range of a purified FRET biosensor. Reagents:
- Purified sensor protein.
- Target metabolite stock solution.
- Assay buffer (mimicking physiological conditions, e.g., pH 7.4, 150 mM KCl). Procedure:
- Dilute purified sensor to a working concentration (e.g., 1 μM) in assay buffer in a quartz cuvette.
- Measure baseline fluorescence emission spectra (excite donor: e.g., 433 nm for CFP; collect 450-600 nm).
- Titrate increasing concentrations of metabolite into the cuvette, mixing thoroughly.
- After each addition, measure the full emission spectrum.
- Data Analysis: Calculate the emission ratio (Acceptor/Donor, e.g., YFP/CFP). Fit the ratio vs. metabolite concentration data to a Hill or sigmoidal dose-response curve to extract Kd/EC₅₀ and dynamic range.
Protocol 2: Live-Cell Imaging and Calibration with Ionophores
Objective: Measure cytosolic metabolite levels in adherent mammalian cells. Reagents:
- Cells expressing the biosensor (e.g., via transfection).
- Imaging buffer (Hanks' Balanced Salt Solution, HBSS).
- Calibration compounds: e.g., for ATP sensors, use 2-deoxyglucose (2-DG, 10 mM) & antimycin A (10 μM) to deplete ATP; oligomycin (1-5 μg/mL) to maximize ATP. Procedure:
- Seed cells on glass-bottom dishes and transfert with sensor plasmid.
- 24-48h post-transfection, replace medium with pre-warmed imaging buffer.
- Acquire time-lapse rationetric images (e.g., CFP and YFP channels) on a widefield or confocal microscope.
- Calibration: After baseline recording, perfuse with depletion cocktail (2-DG + antimycin A) to obtain Rmin, then perfuse with oligomycin to obtain Rmax.
- Data Analysis: Convert pixel-by-pixel ratios to metabolite concentration using the formula: [Metabolite] = Kd * ((R - Rmin)/(Rmax - R)).
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function & Rationale |
|---|---|
| pcDNA3.1(+) Vector | Mammalian expression vector; commonly used for cloning and transient expression of sensor constructs in HEK293T or HeLa cells. |
| FuGENE HD Transfection Reagent | Low-toxicity, high-efficiency reagent for delivering plasmid DNA into a wide range of mammalian cell lines for transient expression. |
| CellLight BacMam 2.0 (Invitrogen) | Baculovirus-based system for efficient, uniform sensor delivery to hard-to-transfect cells (e.g., primary neurons, iPSC-derived cells). |
| Recombinant Phycocyanobilin (PCB) | Essential chromophore for activating iGLIM and other phytochrome-based systems; must be supplemented in cell media. |
| Poly-D-Lysine | Coating agent for glass-bottom dishes; enhances adherence of neuronal or other suspension cells for stable imaging. |
| Carbonyl cyanide 4-(trifluoromethoxy)phenylhydrazone (FCCP) | Mitochondrial uncoupler; used in calibration protocols for ATP/NADH sensors to collapse metabolic gradients. |
| Glass-Bottom Dish (35 mm, No. 1.5) | Optimal for high-resolution live-cell microscopy; provides superior optical clarity over plastic. |
| ROI (Region of Interest) & Kinetic Analysis Tool (e.g., in NIS-Elements/Fiji) | Software tools for quantifying fluorescence intensity changes over time from specific cellular compartments. |
Pathway and Workflow Visualizations
Title: Generic PBP-Based FRET Biosensor Mechanism
Title: Core Workflow for Developing and Using a FRET Biosensor
The selection of a FRET biosensor construct—from the rationetric ATeam for energy charge to the versatile iGLIM toolkit and specific Snifit sensors—must be driven by the biological question, required dynamic range, and cellular context. Integrating rigorous in vitro characterization with robust live-cell calibration protocols is essential for generating quantitative, reliable metabolite data. This guide provides a foundational framework for researchers and drug developers to leverage these powerful tools, advancing the thesis that precise metabolic tracking is crucial for understanding disease mechanisms and identifying novel therapeutic interventions.
Within the context of FRET (Förster Resonance Energy Transfer) biosensor research for metabolite detection, the reliable and efficient delivery of genetic constructs into target cells or organisms is foundational. The choice of delivery system—transfection, viral transduction, or the generation of transgenic models—critically influences biosensor expression levels, localization, dynamics, and ultimately, the fidelity of metabolic readings. This guide provides a technical comparison of these core methodologies, detailing protocols and considerations for their application in live-cell metabolic imaging.
Transfection
Transfection involves the introduction of nucleic acids into eukaryotic cells using non-viral, chemical, or physical methods. For FRET biosensor studies, transient transfection is commonly used for rapid screening and characterization.
Experimental Protocol: Lipid-Mediated Transfection of FRET Biosensor Plasmids
Key Reagents & Materials:
- HEK293T or relevant cell line
- FRET biosensor plasmid DNA (e.g., AKAR3 for cAMP, or a custom metabolite sensor)
- High-purity plasmid preparation kit
- Commercial lipid-based transfection reagent (e.g., Lipofectamine 3000)
- Opti-MEM Reduced Serum Medium
- Phenol red-free imaging medium
Procedure:
- Day 1: Seed cells onto poly-D-lysine-coated glass-bottom dishes at 60-80% confluency.
- Day 2 (Transfection): a. Dilute 1.0 µg of biosensor plasmid DNA in 50 µL Opti-MEM. Add 2.0 µL of P3000 enhancer reagent (if using Lipofectamine 3000). b. In a separate tube, dilute 2.0 µL of lipid reagent in 50 µL Opti-MEM. Incubate for 5 min at RT. c. Combine DNA and lipid dilutions, mix gently, and incubate for 15-20 min at RT to form lipid-DNA complexes. d. Add the 100 µL complex dropwise to cells in 1 mL of complete medium. Gently swirl the dish.
- Day 3 (Imaging): 24-48 hours post-transfection, replace medium with phenol red-free imaging medium. Perform FRET imaging on a confocal or epifluorescence microscope equipped with appropriate filters (e.g., CFP excitation, YFP/CFP emission).
Quantitative Comparison of Transfection Methods
| Method | Typical Efficiency (Adherent Cell Lines) | Cytotoxicity | Maximum Insert Size | Primary Cell Suitability | Cost & Throughput |
|---|---|---|---|---|---|
| Cationic Lipids | 70-90% (HEK293) | Moderate | >10 kb | Low to Moderate | Moderate / High |
| Polyethylenimine (PEI) | 60-85% | Moderate-High | >10 kb | Low | Low / High |
| Electroporation | 50-80% | High | >10 kb | High | High / Low-Moderate |
| Calcium Phosphate | 30-50% | Moderate | >10 kb | Very Low | Very Low / Low |
Transfection workflow for FRET biosensor delivery.
Viral Transduction
Viral transduction offers higher efficiency, especially in hard-to-transfect cells (e.g., neurons, primary cells, stem cells), enabling stable biosensor expression.
Experimental Protocol: Lentiviral Transduction for Stable Biosensor Expression
Key Reagents & Materials:
- Lenti-X 293T cells (for packaging)
- Lentiviral transfer plasmid (biosensor), packaging psPAX2, envelope pMD2.G
- PEI transfection reagent
- Polybrene (hexadimethrine bromide)
- Culture medium (with/without serum)
- PBS, 0.45 µm PES filter
Procedure:
- Virus Production (Day 0-3): a. Day 0: Plate Lenti-X cells. b. Day 1: Co-transfect cells with transfer, packaging, and envelope plasmids (ratio ~4:3:1) using PEI. c. Day 2: Replace medium with fresh growth medium. d. Day 3: Harvest viral supernatant, filter through 0.45 µm PES filter. Aliquot and store at -80°C or concentrate via ultracentrifugation.
- Cell Transduction (Day 4): a. Plate target cells. At 30-50% confluency, replace medium with fresh medium containing viral supernatant and 6-8 µg/mL Polybrene. b. Centrifuge plates at 800 x g for 30 min at 32°C (spinoculation) to enhance infection. c. After 24h, replace with fresh medium.
- Selection & Imaging (Day 5+): If using a construct with a selectable marker (e.g., puromycin), begin antibiotic selection 48h post-transduction. Expand resistant pools and perform FRET imaging.
Viral Vector Characteristics
| Vector Type | Packaging Capacity | Integration | Titer Range (TU/mL) | Expression Onset | Biosensor Application |
|---|---|---|---|---|---|
| Adenovirus (AdV) | ~8 kb | No (Episomal) | 10^10 - 10^12 | Rapid (24-48h) | High expression, transient, cytotoxic |
| Lentivirus (LV) | ~8 kb | Yes | 10^7 - 10^9 | Slow (72h+) | Stable expression, diverse cell types |
| Adeno-Associated Virus (AAV) | ~4.7 kb | Rare | 10^11 - 10^13 | Slow (weeks) | In vivo delivery, low immunogenicity |
| Retrovirus (RV) | ~8 kb | Yes | 10^6 - 10^8 | Slow (72h+) | Dividling cells only |
Decision tree for selecting a viral delivery vector.
Transgenic Models
Transgenic animals provide the most physiologically relevant context for FRET biosensor studies, enabling metabolite detection in intact tissues and during development.
Experimental Protocol: Generating a FRET Biosensor Transgenic Mouse Line (Pronuclear Injection)
Key Reagents & Materials:
- Purified, linearized biosensor transgene construct (with tissue-specific promoter).
- Fertilized mouse oocytes (e.g., C57BL/6J strain).
- Microinjection apparatus.
- Pseudopregnant female mice.
- PCR genotyping primers.
Procedure:
- Vector Design: Clone the FRET biosensor sequence downstream of a strong, ubiquitous (e.g., CAG) or tissue-specific promoter. Flank with necessary insulators. Purify and linearize the vector.
- Microinjection: Microinject ~100-200 copies of the linearized construct into the pronucleus of fertilized single-cell embryos.
- Embryo Transfer: Surgically transfer viable injected embryos into the oviducts of pseudopregnant foster females.
- Founder Identification: Genotype pups born (~3 weeks) via tail biopsy PCR using transgene-specific primers. Positive founders (F0) are mosaics.
- Line Establishment: Cross F0 founder with wild-type mice. Screen F1 offspring for germline transmission. Establish stable heterozygous lines.
- FRET Imaging: Isolate primary cells or perform intravital imaging on tissues from positive transgenic animals to measure metabolite dynamics.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in FRET Biosensor Delivery |
|---|---|
| Lipofectamine 3000 | Lipid-based transfection reagent for high-efficiency, transient plasmid delivery to adherent cell lines. |
| Polyethylenimine (PEI) Max | Cationic polymer for cost-effective transfection and viral packaging plasmid delivery in 293T cells. |
| pAAV-hSyn1 | AAV serotype and neuron-specific promoter plasmid for targeted biosensor expression in the brain. |
| psPAX2 / pMD2.G | 2nd generation lentiviral packaging and VSV-G envelope plasmids for producing safe, high-titer virus. |
| Hexadimethrine Bromide (Polybrene) | Positively charged polymer that enhances viral transduction efficiency by neutralizing charge repulsion. |
| Puromycin Dihydrochloride | Antibiotic for selecting mammalian cells stably transduced with lentiviral constructs containing a puromycin resistance gene. |
| CAG Promoter Plasmid | Strong synthetic promoter (CMV enhancer + chicken beta-actin) for driving high-level, ubiquitous biosensor expression in transgenic constructs. |
| CRISPR-Cas9 reagents | For targeted knock-in of FRET biosensor sequences into safe-harbor loci (e.g., Rosa26) in zygotes or stem cells. |
Within the broader thesis on FRET biosensor metabolite detection research, the selection and integration of an appropriate imaging platform are critical. The choice between confocal microscopy, widefield epifluorescence microscopy, and microplate readers dictates the resolution, throughput, quantification accuracy, and ultimately, the biological insights achievable in dynamic live-cell metabolic studies. This guide provides a technical framework for setting up these platforms for robust, quantitative FRET biosensor experiments.
Core Platform Comparison for FRET Biosensor Imaging
Table 1: Quantitative Comparison of Imaging Platforms for FRET Biosensor Research
| Feature | Confocal Microscopy (Laser-Scanning) | Widefield Epifluorescence Microscopy | Multimode Microplate Reader |
|---|---|---|---|
| Spatial Resolution | High (~0.2-0.3 µm lateral) | Moderate (~0.4-0.5 µm lateral) | None (whole-well averaging) |
| Optical Sectioning | Excellent (pinhole eliminates out-of-focus light) | Poor (requires computational deconvolution) | None |
| Acquisition Speed | Slow (limited by scanning) | Very Fast (full-frame capture) | Very Fast (parallel detection) |
| Throughput | Low (single FOV/cell) | Medium (multiple FOVs/well) | Very High (96/384/1536-well plates) |
| Photobleaching/ Phototoxicity | High (focused laser point) | Moderate (widefield illumination) | Low (short exposure, bottom read) |
| Primary FRET Modality | Acceptor Photobleaching, Rationetric Intensity | Fluorescence Lifetime Imaging (FLIM), Rationetric Intensity | Rationetric Intensity |
| Key Metric for Biosensors | High-resolution spatial maps of FRET efficiency | Fast kinetics & lifetime (τ) measurements | High-throughput dose-response & kinetic data |
| Typical Cost | Very High | High-Medium | High |
Experimental Protocols for FRET Biosensor Validation
Protocol 1: Rationetric FRET Measurement on a Widefield/Confocal Microscope
- Cell Preparation: Seed cells expressing the FRET biosensor (e.g., a glucose sensor like FLII12Pglu-700μδ6) in glass-bottom dishes or plates. Allow for adherence and expression (24-48 hrs).
- Microscope Setup:
- Widefield: Use a microscope with a stable light source (LED or metal halide), a dual-band emission filter set (e.g., for CFP/YFP), and a sensitive CCD/CMOS camera.
- Confocal: Configure sequential line-scanning to minimize cross-talk. Set appropriate laser lines (e.g., 405 nm for CFP) and emission detection windows (e.g., 460-500 nm for donor, 520-550 nm for acceptor).
- Image Acquisition: Acquire donor and acceptor channel images sequentially. For kinetics, establish a baseline, then perfuse with stimulus (e.g., metabolite or drug).
- Image Analysis: Background subtract all images. Generate a rationetric image by dividing the acceptor emission image by the donor emission image (FRET ratio = IAcceptor/IDonor). Use regions of interest (ROIs) to quantify ratio changes over time.
Protocol 2: High-Throughput FRET Kinetics on a Plate Reader
- Plate Preparation: Seed biosensor-expressing cells in a black-walled, clear-bottom microplate (e.g., 96-well). Include control wells for background (untransfected cells) and normalization.
- Plate Reader Configuration: Set the instrument for top or bottom dual-emission ratio reads.
- Excitation: Filter for donor excitation (e.g., 430/24 nm for CFP).
- Emission: Configure simultaneous or rapid alternating detection using filters for donor (e.g., 485/20 nm) and acceptor (e.g., 535/25 nm).
- Kinetic Assay: Program a kinetic loop with 1-5 minute intervals. After 3 baseline reads, automatically inject a compound from an onboard injector and continue reading for 60+ minutes.
- Data Processing: Export raw donor and acceptor fluorescence values. Calculate the FRET ratio per well, subtract background, and normalize to the pre-stimulus baseline or control wells. Plot ratio vs. time.
Visualizing the FRET Biosensor Workflow and Signaling Context
Workflow for FRET Biosensor Imaging
FRET Biosensor Principle & Measurement
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for FRET Biosensor Metabolite Detection Research
| Item | Function & Rationale |
|---|---|
| Genetically-Encoded FRET Biosensor Plasmid | Core reagent. Encodes the metabolite-binding protein flanked by donor (CFP, mTurquoise2) and acceptor (YFP, cpVenus) fluorescent proteins. |
| Lipid-Based Transfection Reagent (e.g., PEI, Lipofectamine 3000) | For efficient delivery of biosensor plasmid into mammalian cell lines of interest. |
| Cell Culture Microplates (Black, Clear-Bottom) | Optimized for fluorescence assays. Black walls minimize cross-talk; clear bottom allows high-resolution microscopy. |
| Phenol Red-Free Culture Medium | Phenol red has autofluorescence which interferes with sensitive CFP/YFP detection. |
| Metabolite Agonists/Antagonists & Pharmacological Modulators | Used to perturb metabolic pathways for biosensor validation and experimental assays (e.g., 2-DG for glycolysis, Rotenone for OXPHOS). |
| Ionophores & Control Compounds (e.g., Ionomycin, Forskolin) | Positive controls for biosensors sensitive to Ca²⁺ or cAMP, validating cellular expression and function. |
| Live-Cell Imaging Buffer (Hanks' Balanced Salt Solution, HBSS) | Physiologically buffered saline to maintain cell health during time-lapse imaging outside a CO₂ incubator. |
| Sensitive sCMOS/EMCCD Camera | Critical for widefield/confocal detection of low-light FRET signals with high temporal resolution. |
| Dual-Emission Filter Set (e.g., CFP/YFP) | Enables simultaneous or rapid alternation collection of donor and acceptor emission for accurate ratio calculation. |
| FRET Analysis Software (e.g., ImageJ/FIJI, MetaFluor, CellProfiler) | For background subtraction, ratio calculation, kinetic analysis, and data visualization from imaging datasets. |
Förster Resonance Energy Transfer (FRET)-based biosensors are indispensable tools in modern biochemical research, particularly for the real-time, quantitative detection of metabolites within living cells. The core thesis of this field posits that the spatiotemporal dynamics of metabolites, captured via precise FRET efficiency (E) calculations, are critical for elucidating metabolic pathways, signaling cascades, and drug-target interactions. Quantitative data acquisition via rationetric imaging forms the foundational methodology for this thesis, transforming fluorescent emission ratios into reliable, quantitative metrics of molecular activity and interaction.
Fundamental Principles of Rationetric FRET Imaging
Rationetric FRET imaging involves the simultaneous or sequential acquisition of fluorescence emissions from the donor and acceptor fluorophores within a biosensor. The primary quantitative output is the emission ratio (R), typically acceptor emission divided by donor emission (IA/ID). This ratio is intrinsically corrected for artifacts common in biological imaging, such as variable biosensor expression levels, photobleaching, and changes in sample thickness.
Table 1: Key Advantages of Rationetric vs. Intensity-Based FRET Measurement
| Measurement Type | Primary Output | Key Advantage | Major Vulnerability |
|---|---|---|---|
| Intensity-Based | Donor Quenching or Acceptor Sensitization | Simpler acquisition setup | Artifacts from concentration, excitation intensity |
| Rationetric | Emission Ratio (IA/ID) | Internal control for biosensor concentration, path length | Cross-talk & bleed-through between channels |
| Fluorescence Lifetime (FLIM) | Donor Fluorescence Lifetime (τ) | Absolute measure, concentration-independent | Complex instrumentation, slower acquisition |
Detailed Experimental Protocol for Rationetric FRET Imaging
Microscope Setup & Calibration
- Microscope: An inverted epifluorescence or confocal microscope equipped with a high-sensitivity camera (e.g., sCMOS, EMCCD) or photomultiplier tubes (PMTs).
- Light Source: A stable LED or laser source appropriate for donor excitation.
- Filter Sets: A dedicated filter cube or sequential filter settings are required:
- Donor Channel: Donor excitation / Donor emission filter.
- Acceptor Channel: Donor excitation / Acceptor emission filter (for sensitized emission).
- Acceptor Direct Excitation Channel: Acceptor excitation / Acceptor emission filter (for correction controls).
- Objective: High Numerical Aperture (NA >1.2) oil-immersion objective recommended.
- Environmental Control: Chamber for maintaining 37°C and 5% CO₂ for live-cell imaging.
Image Acquisition Protocol
- Cell Preparation: Plate cells expressing the FRET biosensor onto imaging-grade dishes. Allow for adherence and biosensor expression.
- Focus & Field Selection: Locate a field of healthy, moderately expressing cells.
- Sequential Image Acquisition: a. IDD: Image with Donor excitation, Donor emission filter. b. IDA: Image with Donor excitation, Acceptor emission filter (the FRET channel). c. IAA: Image with Acceptor excitation, Acceptor emission filter. d. IAD (Optional): Image with Acceptor excitation, Donor emission filter.
- Background Subtraction: For each channel, acquire an image from a cell-free region and subtract pixel-by-pixel from the corresponding sample images.
- Time-Series: Repeat sequence over time for dynamic measurements. Minimize light exposure to reduce phototoxicity.
Quantitative FRET Efficiency Calculation: Correction Methods
Raw intensity measurements (I_DA) are contaminated by spectral bleed-through (SBT). Precise E calculation requires correction.
Spectral Correction Coefficients
- Bleed-Through Coefficient (α): The proportion of donor emission detected in the acceptor channel. Calculate using cells expressing donor-only: α = mean(IDA) / mean(IDD).
- Cross-Excitation Coefficient (β): The proportion of acceptor directly excited by the donor excitation light. Calculate using cells expressing acceptor-only: β = mean(IDA) / mean(IAA) under donor excitation conditions.
Corrected FRET Calculation
The corrected FRET signal (Fc) is calculated pixel-by-pixel:
Fc = IDA - (α * IDD) - (β * I_AA)
FRET Efficiency (E) Formulas
FRET efficiency, the fraction of donor molecules transferring energy to an acceptor, can be approximated by:
E ≈ Fc / (Fc + G * I_DD)
Where G is an instrument-specific calibration factor relating donor quenching to acceptor sensitization. It can be determined using a linked donor-acceptor reference standard.
Table 2: Summary of Key Quantitative Parameters and Formulas
| Parameter | Symbol | Definition | Typical Calculation Method |
|---|---|---|---|
| Apparent FRET Ratio | R | IA/ID | IDA / IDD |
| Bleed-Through Coeff. | α | Donor emission in acceptor channel | IDA(donor-only) / IDD(donor-only) |
| Cross-Excitation Coeff. | β | Direct acceptor excitation by donor light | IDA(acceptor-only) / IAA(acceptor-only) |
| Corrected FRET | Fc | SBT-corrected FRET signal | IDA - (α*IDD) - (β*I_AA) |
| FRET Efficiency | E | Fraction of donor energy transferred | Fc / (Fc + G * I_DD) |
| Correction Factor | G | System calibration factor | Derived from reference construct with known E |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for FRET Biosensor Experiments
| Item | Function & Description | Example/Catalog Consideration |
|---|---|---|
| Genetically-Encoded FRET Biosensor | Core reagent; expresses donor and acceptor fluorophores linked by a metabolite-sensitive domain. | e.g., AT1.03 (ATP), iNap (NADH), SoNar (NAD+/NADH). Must be validated for target metabolite. |
| Transfection Reagent / Viral Vector | For delivering biosensor DNA into target cells. | Lipofectamine, FuGENE, or lentiviral/AAV vectors for stable/primary cells. |
| Cell Culture Media & Supplements | Maintain cell health during imaging; some may affect metabolite levels. | Phenol-red free media is essential for imaging. Consider controlled serum or nutrient levels. |
| Reference Control Plasmids | Donor-only and acceptor-only constructs for calculating α and β coefficients. | Critical for quantitative correction. Often created from the original biosensor. |
| Calibration Standards | Linked donor-acceptor constructs or chemical solutions for determining G factor. | e.g., Cerulean-Venus tandem with known fixed distance. |
| Metabolite Modulators | Pharmacological agents or substrates to manipulate intracellular metabolite levels for validation. | e.g., Oligomycin (ATP depletion), H₂O₂ (redox stress), specific metabolic pathway inhibitors. |
| Immersion Oil | High-quality oil matching the objective's refractive index (nd). | Prevents signal loss and spherical aberration. |
Signaling Pathway and Experimental Workflow Diagrams
Diagram 1: FRET Biosensor Metabolite Sensing Mechanism
Diagram 2: Quantitative FRET Data Acquisition & Analysis Workflow
Within the broader thesis on Förster Resonance Energy Transfer (FRET) biosensor metabolite detection research, this whitepaper explores its pivotal applications in modern drug discovery. The integration of high-content screening (HCS) with metabolic pathway profiling via FRET-based sensors represents a paradigm shift, enabling the simultaneous quantification of dynamic metabolic fluxes and phenotypic changes in living cells. This guide details the technical methodologies, experimental protocols, and data analysis frameworks that underpin this integrative approach.
Core Principles: FRET Biosensors for Metabolite Detection
Genetically encoded FRET biosensors are engineered proteins that change their conformation upon binding a specific target metabolite, altering the energy transfer efficiency between donor and acceptor fluorescent proteins. This allows real-time, spatiotemporal quantification of metabolites like glucose, ATP, lactate, glutamate, and cAMP in living cells. This capability is fundamental for profiling metabolic pathway activities in response to pharmacological intervention.
Integrating FRET Metabolite Detection with High-Content Screening (HCS)
HCS automates the acquisition and analysis of multiplexed cellular imaging data. By incorporating FRET biosensors, HCS evolves from morphological assessment to functional metabolic phenotyping. The workflow involves:
- Cell Line Engineering: Stable integration of a FRET biosensor for a key metabolic node (e.g., ATP:AMP ratio via the AMPK pathway sensor).
- Compound Library Exposure: Treatment with small molecules or genetic perturbations in multi-well plates.
- Automated Live-Cell Imaging: Time-lapse acquisition of donor and acceptor emission channels using an HCS microscope.
- Multiparametric Data Extraction: Quantification of FRET ratio (metric for metabolite concentration) alongside conventional HCS parameters (nuclei count, cell viability, mitochondrial morphology).
- Pathway Analysis: Correlating metabolite dynamics with phenotypic outcomes to infer mechanism of action (MoA).
Experimental Protocols
Protocol: HCS with a FRET-Based Glucose Sensor
- Objective: To screen for compounds that modulate cellular glucose uptake in a cancer cell line.
- Materials: See "The Scientist's Toolkit" below.
- Method:
- Seed HeLa cells stably expressing the FRET glucose sensor FLII12Pglu-700μΔ6 in a 96-well collagen-coated imaging plate at 10,000 cells/well. Culture for 24h.
- Using an automated liquid handler, transfer 100 μL of compound library (10 μM final concentration in assay medium) to designated wells. Include controls: DMSO (negative), 100 nM insulin (positive for uptake), and 100 μM cytochalasin B (inhibitor).
- Incubate plate for 2h at 37°C, 5% CO₂.
- Replace medium with low-glucose (2 mM) imaging buffer.
- Mount plate on a pre-warmed (37°C) stage of a high-content imager equipped with environmental control and a 40x objective.
- Acquire a baseline image for 5 minutes (1 frame/min) in CFP and YFP channels (ex: 458nm, em: 475-525nm for CFP; em: 525-575nm for YFP).
- Automatically inject 50 μL of high-glucose (20 mM final) buffer into each well and continue time-lapse imaging for 30 minutes.
- Analysis: For each cell (ROI), calculate the background-subtracted FRET ratio (YFP/CFP intensity). Normalize the ratio to the baseline period (T=0). Plot the kinetic response curve. Calculate the Area Under the Curve (AUC) for the first 15 minutes post-stimulation as the primary quantitative HCS readout for glucose uptake rate.
Protocol: Metabolic Pathway Profiling via Multiplexed FRET Sensors
- Objective: To profile the effect of a lead oncology compound on multiple interconnected metabolic pathways.
- Method:
- In separate assay plates, prepare cell lines expressing distinct FRET biosensors for key metabolites: AT1.03 (ATP), Laconic (lactate), iNAP1 (NADPH).
- Treat all plates with the compound at its IC50 (from viability assays) and a vehicle control.
- Perform simultaneous live-cell FRET imaging for each sensor as per Protocol 4.1, but with sensor-specific imaging settings.
- Extract normalized FRET ratio kinetics for each metabolite.
- Integrate data into a kinetic metabolic model to identify the primary site of disruption (e.g., glycolysis, pentose phosphate pathway, oxidative phosphorylation).
Quantitative Data Presentation
Table 1: Representative HCS Data from a FRET Glucose Uptake Screen of a Kinase Inhibitor Library
| Compound ID | Target Class | Glucose Uptake AUC (Normalized) | p-value (vs. DMSO) | Cell Viability (%) | Mitotracker Intensity (Normalized) |
|---|---|---|---|---|---|
| DMSO | Control | 1.00 ± 0.12 | - | 100 ± 5 | 1.00 ± 0.15 |
| Insulin | Growth Factor | 1.85 ± 0.18 | <0.001 | 102 ± 4 | 1.10 ± 0.12 |
| Cpd-7A | AKT Inhibitor | 0.55 ± 0.09 | <0.001 | 95 ± 6 | 0.65 ± 0.08 |
| Cpd-12F | p38 MAPK Inhibitor | 1.10 ± 0.11 | 0.32 | 98 ± 5 | 0.95 ± 0.10 |
| Cpd-3D | mTOR Inhibitor | 0.72 ± 0.10 | <0.01 | 88 ± 7 | 0.80 ± 0.09 |
Table 2: Metabolic Pathway Profiling Results for Lead Compound Cpd-7A
| Biosensor (Metabolite) | Pathway Monitored | FRET Ratio Δ (10min Post-Treatment) | Interpretation |
|---|---|---|---|
| AT1.03 (ATP) | Energy Charge | -32% | Severe depletion of cellular ATP. |
| Laconic (Lactate) | Glycolysis / Warburg Effect | -45% | Drastic reduction in lactate production. |
| iNAP1 (NADPH) | Pentose Phosphate Pathway / Redox | -5% | Minimal impact on NADPH pool. |
| GluSnFR (Glutamate) | TCA Cycle / Anaplerosis | +15% | Moderate accumulation, suggesting TCA disruption. |
Visualizing Signaling Pathways and Workflows
Title: HCS Integrated FRET Screening Workflow
Title: Key Metabolic Pathways & FRET Sensor Nodes
The Scientist's Toolkit
Key Research Reagent Solutions for FRET-HCS Experiments
| Item | Function in Experiment | Example/Supplier |
|---|---|---|
| Genetically Encoded FRET Biosensor Plasmids | Core detection tool for specific metabolites. | FLII12Pglu-700μΔ6 (Glucose), AT1.03 (ATP), Laconic (Lactate) (Addgene). |
| Lentiviral Packaging System | For stable, homogeneous sensor expression in target cell lines. | psPAX2, pMD2.G packaging plasmids. |
| Collagen-Coated Imaging Microplates | Provide consistent cell adhesion for automated microscopy. | CellCarrier-96 Ultra, PerkinElmer. |
| Live-Cell Imaging Medium | Phenol-red free medium maintaining pH and health during imaging. | FluoroBrite DMEM, Gibco. |
| Automated Liquid Handling System | Ensures precise, high-throughput compound and reagent dispensing. | Bravo, Agilent Technologies. |
| High-Content Imaging System | Automated microscope with environmental control, precise filter sets for CFP/YFP, and software for kinetic acquisition. | ImageXpress Micro Confocal (Molecular Devices), Opera Phenix (Revvity). |
| FRET Image Analysis Software | Calculates ratiometric changes and kinetic parameters from time-lapse images. | MetaXpress (MD), Harmony (PerkinElmer), or custom Python/ImageJ scripts. |
| Pharmacological Modulators (Controls) | Essential for assay validation and data normalization. | Insulin, 2-DG, Rotenone, Oligomycin, Cytochalasin B. |
Within the broader thesis of FRET biosensor metabolite detection research, this case study examines the application of genetically encoded Förster Resonance Energy Transfer (FRET)-based biosensors to quantify real-time glycolytic flux in live cancer cells under therapeutic perturbation. The Warburg effect, or aerobic glycolysis, is a hallmark of cancer, making glycolysis a critical target for oncology drug development. This technical guide details methodologies for utilizing these biosensors to generate pharmacodynamic data, enabling the assessment of drug efficacy and mechanism of action at a metabolic level.
Core Principle: FRET Biosensors for Metabolic Detection
FRET biosensors for metabolites like glucose, lactate, pyruvate, ATP, and NADH consist of a specific ligand-binding domain flanked by a donor fluorescent protein (e.g., CFP, mTFP1) and an acceptor fluorescent protein (e.g., YFP, Venus). Upon binding of the target metabolite, a conformational change alters the distance/orientation between the fluorophores, modulating FRET efficiency. The ratiometric measurement (acceptor/donor emission) provides a quantitative, internally controlled readout of metabolite concentration dynamics, directly reporting on pathway flux.
Experimental Protocols
Biosensor Transfection & Cell Culture
- Protocol: Plate target cancer cells (e.g., HeLa, MCF-7, or patient-derived organoids) in glass-bottom imaging dishes. At 50-70% confluence, transfect with the plasmid encoding the glycolytic FRET biosensor (e.g., HY-cyto for cytosolic NADH, or iGlucoSnFR for glucose) using a lipid-based transfection reagent or electroporation. Incubate for 24-48 hours to allow for expression. Use serum-free, low-fluorescence imaging medium 1 hour prior to experiment.
Live-Cell Imaging & Drug Treatment
- Protocol: Place the dish on a temperature (37°C) and CO2 (5%) controlled confocal or widefield epifluorescence microscope. Use dual-emission photometry or ratio imaging. Establish a 5-10 minute baseline recording of the FRET ratio. Without interrupting imaging, add the therapeutic compound (e.g., 2-DG, Metformin, PI3K/mTOR inhibitors) directly to the medium at the desired concentration. Continue time-lapse imaging for 60-120 minutes. Include vehicle control experiments.
Data Processing & Calibration
- Protocol: For each cell, extract fluorescence intensity over time for donor and acceptor channels. Calculate the background-subtracted FRET ratio (R = FAcceptor / FDonor). Normalize ratios to the pre-treatment baseline (R/R0). To convert ratio to approximate metabolite concentration, perform an in situ calibration at the end of the experiment: perfuse cells with calibration buffers containing ionophores (e.g., nigericin) and metabolite-clamping agents (e.g., glucose oxidase for low glucose) to define Rmin and Rmax, fitting to a sigmoidal or linear model.
Flux Analysis via Metabolic Perturbation
- Protocol: To specifically assess glycolytic capacity or reserve, perform a sequential perturbation assay. After establishing a baseline and/or drug response, inhibit mitochondrial ATP synthase with oligomycin (1-3 µM) to force maximum glycolytic flux. Follow this with the addition of 2-Deoxy-D-glucose (2-DG, 50-100 mM) to completely inhibit glycolysis, confirming biosensor specificity.
Data Presentation: Quantitative Response to Therapeutics
Table 1: Glycolytic Flux Parameters Derived from FRET Biosensor Imaging in HeLa Cells
| Therapeutic Agent (Concentration) | Target | Normalized NADH Ratio (R/R0) at 60 min Post-Treatment | Maximum Flux Response to Oligomycin (% Δ from Baseline) | Time to 50% Max Effect (Minutes) |
|---|---|---|---|---|
| Vehicle Control (DMSO) | N/A | 1.02 ± 0.05 | 185 ± 12% | N/A |
| 2-Deoxy-D-Glucose (50 mM) | Hexokinase / Glycolysis | 0.45 ± 0.08 | 22 ± 5% | 8.5 ± 1.2 |
| Metformin (10 mM) | Mitochondrial Complex I | 1.65 ± 0.15 | 210 ± 18% | 35.0 ± 4.5 |
| PI3K Inhibitor (LY294002, 50 µM) | PI3K Signaling | 0.85 ± 0.06 | 145 ± 10% | 25.0 ± 3.1 |
| mTOR Inhibitor (Rapamycin, 100 nM) | mTORC1 | 0.95 ± 0.07 | 165 ± 15% | >60 |
Table 2: Key FRET Biosensors for Glycolytic Metabolite Detection
| Biosensor Name | Target Metabolite | Dynamic Range (ΔR/R0) | Affinity (Kd) | Primary Application in Cancer Studies |
|---|---|---|---|---|
| FLII12Pglu-700μδ6 | Glucose | ~1.5 | ~7 µM | Glucose uptake & hexokinase activity |
| Laconic | Lactate | ~0.5 | ~0.3 mM | Lactate efflux & MCT transporter activity |
| HY-cyto | NADH/NAD+ Redox | ~0.8 | N/A (Redox) | GAPDH & mitochondrial shuttle activity |
| ATeam | ATP | ~2.0 | ~3.5 mM (ATP) | ATP production & energy charge |
| Pyronic | Pyruvate | ~0.4 | ~0.3 mM | Pyruvate kinase activity & mitochondrial entry |
Visualizing Pathways and Workflows
Title: Glycolytic Pathway & Therapeutic Intervention Points
Title: Live-Cell FRET Imaging Experimental Workflow
Title: FRET Biosensor Mechanism of Action
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for FRET-Based Glycolytic Flux Assays
| Item | Function & Role in Experiment | Example Product/Catalog |
|---|---|---|
| Genetically Encoded FRET Biosensor Plasmids | Core tool for metabolite detection. Must be chosen based on target (Glucose, NADH, Lactate, etc.). | HY-cyto (Addgene #65422), iGlucoSnFR (Addgene #199882), Pyronic (Addgene #100864) |
| Transfection Reagent | For introducing biosensor plasmid into cancer cell lines, particularly hard-to-transfect lines. | Lipofectamine 3000, FuGENE HD, or nucleofection kits for primary cells. |
| Glass-Bottom Imaging Dishes | Provide optimal optical clarity for high-resolution, live-cell microscopy. | MatTek dishes (P35G-1.5-14-C) or ibidi µ-Dishes. |
| Low-Autofluorescence Imaging Medium | Minimizes background fluorescence, essential for sensitive ratiometric measurements. | FluoroBrite DMEM or Hibernate-A Low Fluorescence medium. |
| Pharmacologic Inhibitors/Activators | Used for metabolic perturbation (e.g., forcing glycolytic flux) and as therapeutic test compounds. | Oligomycin (ATP synthase inhibitor), 2-DG (glycolysis inhibitor), specific kinase inhibitors (e.g., LY294002). |
| Calibration Buffer Kits | For in situ calibration of biosensor response to determine absolute metabolite concentration ranges. | Commercially available or custom buffers with ionophores (nigericin, monensin) and metabolite clamping agents. |
| Confocal or Widefield Microscope with FRET Capability | Must have controlled environment (temp, CO2), sensitive cameras, and appropriate filter sets for CFP/YFP FRET pairs. | Systems from Nikon, Zeiss, Olympus, or specialized plate readers like BMG LABTECH PHERAstar. |
| Ratiometric Image Analysis Software | For background subtraction, ratio calculation, time-lapse analysis, and single-cell tracking. | Fiji/ImageJ with RatioPlus plugin, MetaMorph, NIS-Elements AR, or custom Python/MATLAB scripts. |
Optimizing FRET Biosensor Performance: Solving Signal, Noise, and Specificity Challenges
Within the broader thesis on FRET biosensor metabolite detection research, achieving a robust Förster Resonance Energy Transfer (FRET) change is critical for accurate, quantitative live-cell measurements. A low observed FRET change, often reported as a low ΔR/R₀ or ΔF/F, compromises data interpretation and biological insight. This technical guide systematically addresses the three primary technical determinants: biosensor expression levels, its ligand-binding affinity (Kd), and intrinsic dynamic range. Accurate diagnosis and correction of issues in these domains are fundamental to advancing metabolic signaling research and drug discovery applications.
Core Determinants of FRET Response
Biosensor Expression Level
Expression level directly impacts the signal-to-noise ratio (SNR). Insufficient expression yields a signal obscured by cellular autofluorescence and instrument noise. Excessively high expression can lead to aggregation, aberrant subcellular localization, and buffering of the target metabolite, perturbing the very biology under study.
Quantitative Guidelines: Table 1: Expression Level Impact on FRET Signal
| Expression State | Typical Emission Intensity (Donor Channel) | FRET Change (ΔR/R₀) | Primary Risk |
|---|---|---|---|
| Too Low | < 2x background autofluorescence | Very Low, Noisy | Poor SNR, data unusable |
| Optimal | 5-10x background autofluorescence | Maximized | High SNR, minimal perturbation |
| Too High | >50x background autofluorescence | Often Attenuated | Buffering, aggregation, cytotoxicity |
Experimental Protocol: Quantifying Expression Levels
- Transfect/Transfert Cells: Use a consistent protocol (e.g., lipofection, electroporation) with a titrated amount of biosensor plasmid (e.g., 0.5, 1.0, 2.0 µg DNA per 35mm dish).
- Image Acquisition: 24-48 hours post-transfection, acquire donor (e.g., CFP) channel images using identical exposure times and laser powers across samples.
- Background Subtraction: Measure mean intensity in a cell-free region to establish background autofluorescence.
- Analysis: Calculate mean donor fluorescence intensity per cell. Plot against transfection DNA amount to identify the linear range. The optimal expression window is typically where intensity is 5-10x the background without saturating pixels.
Sensor Affinity (Kd)
The biosensor's dissociation constant (Kd) must be matched to the expected physiological range of the target metabolite. A sensor with too low an affinity (high Kd) will be largely unbound under basal conditions and may not respond to subtle changes. A sensor with too high an affinity (low Kd) will be saturated at basal levels, yielding a small FRET change upon stimulation, and will act as a potent buffer.
Quantitative Data: Table 2: Matching Sensor Kd to Metabolite Concentration
| Metabolite Context | Expected [Metabolite] Range | Recommended Kd Range | Rationale |
|---|---|---|---|
| Second Messengers (e.g., cAMP, Ca²⁺) | Nanomolar to low micromolar | ~0.5x to 2x basal level | Detect both basal and peak signals |
| Abundant Metabolites (e.g., Glucose, ATP) | Mid micromolar to millimolar | Within physiological fluctuation range | Avoid saturation at baseline |
| Low Abundance Signaling Lipids | Sub-micromolar | Low nM to µM | Maximize occupancy change |
Experimental Protocol: In Vitro Kd Calibration
- Protein Purification: Express and purify the biosensor protein (e.g., via His-tag).
- Spectrofluorometry: In a cuvette, place a known concentration of biosensor in appropriate physiological buffer.
- Titration: Sequentially add known concentrations of the target metabolite (ligand). After each addition, record full emission spectra (excite donor).
- Data Fitting: Calculate the FRET ratio (R) at each ligand concentration [L]. Fit the data to the Hill equation: R = Rmin + (Rmax - R_min) * ([L]^n / (Kd^n + [L]^n)), where n is the Hill coefficient. This yields the in vitro Kd.
Intrinsic Dynamic Range (ΔR_max)
The dynamic range (ΔRmax = Rmax / Rmin or ΔR/Rmin) is the maximum possible FRET ratio change of the biosensor architecture itself. A poorly designed sensor may have a low ΔR_max due to suboptimal linker lengths, orientation factors (κ²), or inefficient allosteric coupling.
Quantitative Benchmarks: Table 3: Typical Dynamic Ranges of Common FRET Pairs
| FRET Pair (Donor-Acceptor) | Theoretical ΔR_max | Practical Achievable ΔR/R₀ | Notes |
|---|---|---|---|
| CFP-YFP (e.g., Cerulean-Citrine) | High | 20%-50% | Standard pair, pH-sensitive (YFP) |
| CFP-mRuby2 | High | 30%-70% | Improved photostability, less pH-sensitive |
| mTurquoise2-sfGFP | Very High | 50%-100%+ | Bright, stable, high FRET efficiency |
| mNeonGreen-mRuby3 | High | 40%-80% | Green-red pair, avoids CFP limitations |
Experimental Protocol: Measuring Dynamic Range in Live Cells
- Clamp Conditions: Transfert cells with the biosensor.
- Minimum Ratio (R_min): Apply conditions that ensure zero ligand occupancy. This may involve:
- For ions: Use a chelator (e.g., BAPTA-AM for Ca²⁺) or inhibitor of upstream synthesis.
- For metabolites: Use pharmacological inhibitors of synthesis or activators of degradation.
- Maximum Ratio (R_max): Apply conditions that saturate the sensor.
- For ions: Use ionophores (e.g., Ionomycin for Ca²⁺).
- For metabolites: Use membrane-permeable analogs or potent agonists.
- Calculation: Acquire ratio images under clamped conditions. ΔRmax = Rmax / Rmin (or calculate as ΔR/Rmin).
Integrated Diagnostic Workflow
A systematic approach is required to diagnose the root cause of a low FRET change.
Title: Diagnostic Flowchart for Low FRET Change
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for FRET Biosensor Optimization
| Reagent/Material | Function/Application | Example Product/Catalog |
|---|---|---|
| Biosensor Plasmid | Encodes the FRET-based sensor. Use modular backbones for easy swapping of sensing domains and FP variants. | Addgene vectors (e.g., pcDNA3.1, pCAGGS). |
| High-Efficiency Transfection Reagent | For consistent, moderate expression in hard-to-transfect cells (e.g., primary neurons, immune cells). | Lipofectamine 3000, Mirus TransIT-X2. |
| Membrane-Permeable Metabolite Analogs/Modulators | To clamp cellular metabolite levels for dynamic range measurement (Rmin, Rmax). | Forskolin (cAMP), Ionomycin (Ca²⁺), 2-DG (Glucose). |
| Pharmacological Inhibitors/Activators | To perturb metabolic pathways and test sensor response in live cells. | H89 (PKA), Wortmannin (PI3K), Oligomycin (ATP). |
| Recombinant Protein Purification Kit | For in vitro Kd characterization via spectrofluorometry. | His-tag Purification Kit (Ni-NTA). |
| Synthetic Metabolite Ligand | High-purity standard for in vitro titration. | Sigma-Aldrich (e.g., cAMP sodium salt). |
| Imaging Chamber | Provides stable, physiological environment during live-cell imaging. | Lab-Tek Chambered Coverglass. |
| Phenol-Red Free Imaging Medium | Reduces background fluorescence for sensitive FRET measurements. | FluoroBrite DMEM. |
| FRET Reference Plasmids | Controls for expression and instrumental setup (e.g., CFP-YFP tandem dimer). | pmCerulean3-mVenus (Addgene). |
Pathway Diagram: FRET Biosensor Response to Metabolite Flux
Title: FRET Biosensor Metabolite Detection Pathway
Effective troubleshooting of low FRET change requires a methodical investigation of expression, affinity, and dynamic range. By quantifying these parameters using the outlined protocols and consulting the diagnostic flowchart, researchers can pinpoint the limiting factor. Optimizing these core elements is not merely a technical exercise but a foundational step in ensuring that FRET biosensor data accurately reflects underlying biology, thereby strengthening conclusions in metabolic research and accelerating therapeutic discovery.
In live-cell imaging for FRET biosensor-based metabolite detection, the integrity of the biological system is paramount. Photobleaching degrades the fluorescent signal of the biosensor, directly compromising the quantitative accuracy of metabolite measurements. Concurrently, phototoxicity induces cellular stress, altering the very metabolic pathways under investigation and leading to biologically irrelevant results. This guide details the imaging parameters and environmental controls necessary to minimize these artifacts, ensuring faithful reporting of metabolic dynamics in research and drug development applications.
Core Mechanisms and Pathways
Pathways of Photodamage in Live-Cell FRET Imaging
Diagram 1: Photodamage Pathways in FRET Imaging
Critical Imaging Parameters & Quantitative Optimization
The optimization of imaging hardware and acquisition settings is the first line of defense against photodamage.
Table 1: Key Imaging Parameters for Minimizing Photodamage
| Parameter | Principle of Effect on Photodamage | Recommended Practice for FRET Biosensors | Quantitative Trade-off Consideration |
|---|---|---|---|
| Excitation Intensity | Linear increase in photon absorption, quadratic increase in phototoxicity risk. | Use minimum intensity to achieve SNR > 5:1. Use neutral density filters. | 50% reduction in intensity can reduce photobleaching by >70%. |
| Exposure Time | Longer exposure increases total photon dose per frame. | Use shortest exposure without compromising signal. Consider binning vs. exposure. | Doubling exposure time typically doubles photobleaching rate. |
| Temporal Resolution | Higher frame rate increases cumulative dose and out-of-focus exposure. | Sample at the slowest rate acceptable for the metabolic process (e.g., 30-60 sec for glucose dynamics). | Reducing frame rate from 1 Hz to 0.1 Hz reduces dose 10-fold. |
| Spatial Resolution (XY) | Smaller pixel size requires higher intensity for same SNR, increasing dose. | Set pixel size to ~1/3 of optical resolution (e.g., 110-130 nm for high NA). Avoid oversampling. | 2x oversampling increases dose 4x for same field of view. |
| Z-stack Acquisition | Multiple planes multiply dose. Out-of-focus planes still experience exposure. | Use confined Z-stacks, optimal spacing (0.5 μm), or single-plane imaging when possible. | A 10-plane stack delivers 10x the dose of a single plane. |
| Detector Gain/EMCCD | Amplifies signal post-readout, not affecting photodamage. | Increase gain to allow lower excitation intensity. Be mindful of increased noise. | Enables up to 10-50x reduction in excitation power. |
Environmental & Sample Control Protocols
Controlling the cellular microenvironment is crucial to mitigate phototoxicity effects and maintain metabolic viability.
Protocol: Preparation of an Optimized Imaging Medium
Objective: To provide a physiologically stable environment that mitigates oxidative stress during prolonged FRET imaging. Materials: Phenol-red free imaging medium, HEPES buffer (20-25 mM), commercially available oxygen scavenging system (e.g., Oxyrase, 0.3-0.6 U/mL), antioxidant (e.g., ascorbic acid 50-100 μM, Trolox 100-200 μM), serum replacement appropriate for cell type, metabolite substrates (e.g., 5-10 mM Glucose). Procedure:
- Prepare base phenol-red free medium supplemented with 25 mM HEPES for pH stability without CO₂ control.
- Add serum replacement (e.g., 1-5% B-27 for neurons) to support cell health.
- Gently add oxygen scavenging enzyme (Oxyrase) and mix without vortexing to avoid denaturation.
- Add fresh antioxidants (Trolox, Ascorbic Acid) immediately before use.
- Adjust pH to 7.4 using NaOH/HCl.
- Filter sterilize (0.22 μm).
- Note: For hypoxia-sensitive studies, omit oxygen scavengers and use a controlled environmental chamber.
Protocol: System Calibration for Intensity Uniformity and Focus Stability
Objective: To eliminate unnecessary exposure from "hot spots" or focus-drift induced re-imaging. Procedure:
- Field Uniformity Calibration: Image a uniform fluorescent slide (e.g., red, green) at multiple positions. Use software to create a flat-field correction map. Apply this map during live acquisition to ensure even illumination, preventing localized over-exposure.
- Autofocus Stabilization: Prior to experiment, set up hardware-based autofocus (e.g., laser-based infrared, or software-assisted). Define a stability corridor (± 0.5 μm). Use a low-power IR laser pulse at intervals (e.g., every 5-10 min) rather than continuous exposure to minimize added photostress.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Photodamage Mitigation in FRET Imaging
| Item | Category | Function & Rationale |
|---|---|---|
| Phenol Red-Free Medium | Imaging Medium | Eliminates background fluorescence and potential photosensitizer activity. |
| HEPES Buffer | pH Stabilizer | Maintains physiological pH outside a CO₂ incubator, critical for open microscope stages. |
| Oxyrase | Oxygen Scavenger | Enzymatically reduces dissolved O₂, suppressing ROS formation at the source. |
| Trolox | Antioxidant | Water-soluble Vitamin E analog; scavenges ROS in aqueous cellular compartments. |
| Cycloactyl or RO-3306 | Cell Cycle Inhibitor | Halts cell cycle progression for long-term studies, reducing motion artifacts and metabolic heterogeneity. |
| Sirius Dyes or CellMask | Fiducial Markers | Low-bleaching, far-red fiducial markers for drift correction without interfering with CFP/YFP FRET channels. |
| Antifade Mountants (e.g., ProLong Live) | Mounting Medium | For fixed samples, contains free radical scavengers to preserve fluorescence during validation imaging. |
| Genetically Encoded ROS Sensors (e.g., roGFP) | Reporters | Internal control to monitor oxidative stress levels during the imaging experiment itself. |
Integrated Experimental Workflow
Diagram 2: Optimized Workflow for FRET Metabolite Imaging
Detailed Steps:
- Sample Preparation (Prep): Transfer cells expressing FRET biosensor to optimized imaging medium. Equilibrate in a dark, temperature-controlled environment (37°C) for 30 minutes.
- System Calibration (Calibrate): Execute flat-field and focus stability protocols (Section 4.2). Set temperature and gas controls (5% CO₂, if used).
- Parameter Definition (Define): Set acquisition software using minima from Table 1. Use a single control position to establish baseline biosensor response and bleaching rate.
- Data Acquisition (Acquire): Initiate experiment. Use hardware autofocus. Monitor a single control position for signs of phototoxicity (e.g., abnormal morphology, sudden FRET ratio shifts).
- Post-Hoc Validation (Validate): Analyze bleaching kinetics in control cells. Compare final morphology to non-imaged controls. Correlate with ROS sensor data if available.
Data Analysis and Correction Protocol
Protocol: Correcting FRET Ratio Time-Series for Photobleaching
Objective: To mathematically isolate metabolite-dependent FRET changes from artifact signal decay. Prerequisite: Acquire control data from cells under non-stimulating conditions to define pure bleaching kinetics. Procedure:
- Calculate Ratios: For each time point t, compute the background-subtracted FRET ratio R(t) = Iₐᶜᶜᵉᵖᵗᵒʳ / Iᵈᵒⁿᵒʳ.
- Model Bleaching: Fit the control cell ratio data Rᶜ(t) to a double exponential decay: Rᶜ(t) = A₁·exp(-k₁·t) + A₂·exp(-k₂·t) + C.
- Apply Correction: For experimental cells, compute the corrected ratio Rˢᶜ(t) = Rˢ(t) / Rᶜ(t), where Rˢ(t) is the raw experimental ratio. This assumes bleaching is multiplicative and consistent across samples.
- Normalize: Express final data as ΔR/R₀ or % change from baseline for metabolite quantification.
Within the broader thesis on FRET biosensor metabolite detection research, a critical and often underexamined challenge is the inherent perturbation caused by the biosensor itself. This technical guide provides an in-depth analysis of how biosensor expression and function can alter native cellular metabolism and physiology, thereby potentially confounding experimental results. The accurate quantification of metabolites via Förster Resonance Energy Transfer (FRET) biosensors relies on the assumption of minimal cellular disturbance, an assumption that requires rigorous validation. This document outlines the sources of perturbation, methods for their quantification, and experimental strategies for mitigation, ensuring data derived from FRET biosensor research accurately reflects in vivo states.
Biosensor perturbation arises from multiple interrelated factors:
- Metabolic Sequestration: The biosensor acts as an additional binding site for the target metabolite, potentially altering its free concentration, flux, and subcellular distribution.
- Energetic and Resource Burden: Biosensor expression consumes cellular resources (ATP, amino acids, nucleotides) and burdens transcriptional/translational machinery.
- Signaling Interference: The biosensor polypeptide may inadvertently participate in or disrupt protein-protein interaction networks, scaffolding, or signaling pathways.
- Misfolding and Aggregation: Overexpression can lead to protein misfolding, triggering cellular stress responses such as the unfolded protein response (UPR).
- Compartment-Specific Effects: Targeted biosensors (e.g., to mitochondria, nucleus) can disrupt organelle membrane potential, integrity, or transport mechanisms.
Quantitative Assessment of Perturbation Effects
The following table summarizes key metrics and experimental findings for assessing biosensor perturbation, derived from recent literature.
Table 1: Quantifiable Metrics for Biosensor Perturbation Assessment
| Metric Category | Specific Measurement | Experimental Technique | Typical Control/Baseline | Indicative Threshold for Significant Perturbation |
|---|---|---|---|---|
| Cellular Fitness | Doubling Time / Growth Rate | Time-lapse microscopy, cell counting. | Isogenic cells without biosensor. | >20% increase in doubling time. |
| Cellular Fitness | Colony Forming Unit (CFU) Efficiency | Clonogenic assay. | Isogenic cells without biosensor. | <70% of control CFU efficiency. |
| Metabolic State | ATP:ADP Ratio | Luciferase-based assay, HPLC. | Untransfected cells or cells expressing inert control protein. | >15% deviation from control ratio. |
| Metabolic State | Lactate Production / Extracellular Acidification Rate (ECAR) | Seahorse XF Analyzer, biochemical assay. | Control cells. | Sustained >25% change in basal ECAR. |
| Stress Response | CHOP or BiP Expression (UPR markers) | qPCR, immunoblotting. | Cells treated with tunica- mycin (positive control) vs. untreated. | >2-fold upregulation vs. unstressed control cells. |
| Biosensor Artifact | Apparent Metabolite Level vs. Direct Measurement | Compare biosensor FRET ratio with LC-MS/MS measurement of extracted metabolite. | LC-MS/MS value from control cells. | Systematic, concentration-dependent discrepancy. |
| Expression Load | Biosensor Protein Abundance | Quantitative immunoblotting, flow cytometry (if fluorescent). | Endogenous level of a similar-sized abundant protein (e.g., GAPDH). | Expression exceeding 0.1-1% of total cellular protein. |
Experimental Protocols for Perturbation Analysis
Protocol 4.1: Parallel Measurement of Growth Rate and Biosensor Signal
Objective: To correlate cellular proliferation with biosensor expression and function in real-time.
- Seed cells expressing the FRET biosensor and control cells (expressing a non-perturbing fluorescent protein like mCherry) in a 96-well imaging plate.
- Place the plate in a live-cell imager maintained at 37°C and 5% CO₂.
- Acquire phase-contrast images for confluence analysis (every 2 hours) and fluorescence images (FRET channel and donor/acceptor channels) for metabolite sensing (as required by experiment).
- Use image analysis software to segment cells and quantify:
- Doubling Time: From phase-contrast confluence over 48-72 hours.
- Mean Biosensor Expression: From the donor channel intensity (corrected for background).
- FRET Ratio: As per standard calibration.
- Plot growth curves and compare doubling times. Correlate individual cell expression level with its division time.
Protocol 4.2: Direct Comparison of Metabolite Levels via LC-MS/MS
Objective: To validate biosensor readings against an orthogonal, absolute quantification method.
- Sample Preparation: In parallel experiments, subject identical cultures of biosensor-expressing and control cells to the same treatment.
- FRET Measurement: For one set, perform live-cell FRET ratio imaging.
- Metabolite Extraction: At the identical timepoint, rapidly wash the other set of cells with cold saline and quench metabolism with cold (-20°C) 80% methanol/water. Scrape cells, vortex, and incubate at -80°C for 1 hour. Centrifuge at 16,000×g for 15 min at 4°C.
- LC-MS/MS Analysis: Dry the supernatant under nitrogen, reconstitute in appropriate solvent, and analyze using a targeted LC-MS/MS method calibrated with authentic metabolite standards. Normalize metabolite peaks to total protein content or cell count.
- Data Correlation: Plot the biosensor FRET ratio against the LC-MS/MS quantified concentration for the same condition. A linear relationship with a slope of 1 (after sensor calibration) indicates minimal perturbation. Deviations suggest sequestration or interference.
Protocol 4.3: Assessing Unfolded Protein Response (UPR) Activation
Objective: To determine if biosensor overexpression induces endoplasmic reticulum stress.
- Cell Lysis: Harvest biosensor-expressing cells, control cells, and positive control cells (treated with 2 µg/mL tunicamycin for 6 hours). Lyse in RIPA buffer with protease inhibitors.
- Immunoblotting: Separate 20-30 µg of protein by SDS-PAGE and transfer to a PVDF membrane.
- Probing: Probe the membrane with primary antibodies against UPR markers (e.g., anti-BiP/GRP78, anti-CHOP) and a loading control (e.g., anti-β-Actin).
- Quantification: Use chemiluminescence detection and densitometry. Calculate the fold-change in BiP or CHOP protein level normalized to the loading control and relative to the untreated control sample. Significant upregulation indicates biosensor-induced ER stress.
Pathway and Workflow Visualizations
Diagram 1: Biosensor Perturbation Sources & Consequences
Diagram 2: Experimental Workflow for Mitigating Perturbation
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for Perturbation Analysis
| Reagent / Material | Function & Rationale | Example Product/Catalog |
|---|---|---|
| Tunicamycin | Induces ER stress by inhibiting N-linked glycosylation; serves as a positive control for UPR activation in Protocol 4.3. | Millipore Sigma, Cat# 654380 |
| Seahorse XF Glycolysis Stress Test Kit | Provides standardized reagents (glucose, oligomycin, 2-DG) to measure extracellular acidification rate (ECAR), a key indicator of glycolytic flux perturbation. | Agilent Technologies, Part # 103020-100 |
| LC-MS/MS Metabolite Standards | Authentic, isotopically labeled standards are required for absolute quantification of metabolites in Protocol 4.2 to validate biosensor accuracy. | Cambridge Isotope Laboratories (e.g., [13C6]-Glucose), Sigma-Aldrich MSUP. |
| Anti-BiP/GRP78 Antibody | Primary antibody for detecting a key marker of the unfolded protein response via immunoblotting in Protocol 4.3. | Cell Signaling Technology, Cat# 3177S |
| FuGENE HD Transfection Reagent | Low-cytotoxicity transfection reagent ideal for delivering biosensor plasmids with minimal acute stress, allowing for better titration of expression. | Promega, Cat# E2311 |
| Tetracycline-Inducible Expression System | Enables precise, dose-controlled induction of biosensor expression, critical for titrating to the minimal effective level to reduce burden. | Takara Bio, Tet-One Inducible Systems |
| CellTiter-Glo Luminescent Cell Viability Assay | Measures ATP content as a direct indicator of cellular metabolic health and energy charge in perturbation screening. | Promega, Cat# G7570 |
Correcting for Spectral Crosstalk and Bleed-Through in Data Analysis
In Förster Resonance Energy Transfer (FRET)-based biosensor research, accurate metabolite detection is fundamentally limited by spectral crosstalk and bleed-through. These phenomena introduce systematic errors by causing donor emission to leak into the acceptor channel and vice-versa, as well as through direct acceptor excitation by the donor excitation wavelength. This technical guide provides an in-depth framework for the mathematical correction of these artifacts, a critical step for quantifying genuine FRET efficiency and obtaining reliable metabolite concentration data in live-cell imaging and high-throughput screening for drug development.
FRET biosensors translate metabolite concentration changes into quantifiable fluorescence ratio changes. A typical intensiometric FRET sensor consists of a donor fluorophore (e.g., CFP, mTurquoise2) and an acceptor fluorophore (e.g., YFP, cpVenus) linked by a metabolite-binding domain. Upon binding, a conformational shift alters the FRET efficiency. However, the measured signals in the donor (IDD) and acceptor (IAA) channels are contaminated:
- Spectral Bleed-Through (SBT): Donor fluorescence emitted in its characteristic range is also detected in the acceptor emission filter set.
- Direct Acceptor Excitation: The light intended to excite the donor also partially excites the acceptor fluorophore. These contaminants must be corrected to calculate the true, FRET-dependent signal in the acceptor channel (IFRET).
Quantitative Characterization of Crosstalk Coefficients
Correction requires experimentally determining coefficients that define the contamination levels. These are best derived from control samples expressing donor-only or acceptor-only constructs under identical imaging conditions.
Table 1: Experimentally Determined Crosstalk Coefficients
| Coefficient | Symbol | Definition | Typical Experimental Value (CFP/YFP pair) |
|---|---|---|---|
| Donor Bleed-Through | α | IAA(donor) / IDD(donor) | 0.35 - 0.55 |
| Acceptor Direct Excitation | β | IDA(acceptor) / IAA(acceptor) | 0.05 - 0.15 |
| Acceptor Bleed-Through | γ | IAA(acceptor) / IDA(acceptor) | ≤ 0.01 |
| Donor Direct Excitation | δ | IDD(acceptor) / IAA(acceptor) | ≤ 0.01 |
IDD: Intensity in donor channel with donor excitation; IAA: Intensity in acceptor channel with acceptor excitation; IAD: Intensity in acceptor channel with donor excitation.
Protocol 2.1: Determining Coefficients α and β
- Cell Preparation: Plate cells expressing the donor-only construct (e.g., CFP) in one well and the acceptor-only construct (e.g., YFP) in another.
- Image Acquisition: Acquire three images per sample:
- IDD: Donor channel (Donor Ex / Donor Em).
- IAA: Acceptor channel (Acceptor Ex / Acceptor Em).
- IAD: FRET channel (Donor Ex / Acceptor Em).
- Calculation: For donor-only cells, calculate α = mean(IAD) / mean(IDD). For acceptor-only cells, calculate β = mean(IAD) / mean(IAA). Use a robust ROI analysis across ≥ 20 cells.
Mathematical Correction Models
The observed intensities are linear combinations of the pure components: True Donor (D), True Acceptor (A), and FRET-induced Acceptor (F).
Equations for Observed Intensities:
- IDD(obs) = D + δ·A
- IAA(obs) = A + γ·D
- IAD(obs) = F + α·D + β·A
Assuming γ and δ are negligible for well-designed filter sets, the corrected FRET signal (IFRET) is derived:
IFRET = IAD(obs) - α·IDD(obs) - β·IAA(obs)
The standard metric, the corrected FRET Ratio (Rcorrected), is: Rcorrected = IFRET / IDD(obs)
This ratio is proportional to FRET efficiency and thus to metabolite concentration.
Diagram 1: Pathways of signal contribution to detected FRET channel.
Advanced Correction: 3-Cube and Sensitized Emission Methods
For precise quantification, the 3-cube method is standard. It uses images from three filter sets: Donor, Acceptor, and FRET.
Protocol 4.1: The 3-Cube Sensitized Emission FRET Protocol
- Microscope Setup: Configure filter sets for Donor (ExD/EmD), Acceptor (ExA/EmA), and FRET (ExD/EmA). Maintain identical exposure times and illumination intensity.
- Image Acquisition: Capture a z-stack or time-series for all three channels. Ensure perfect image registration.
- Background Subtraction: Subtract mean intensity from a cell-free region from all images.
- Pixel-by-Pixel Calculation: Apply the correction formula
IFRET = IAD - α*IDD - β*IAAto each pixel using pre-determined α and β. - Ratio Calculation: Generate the corrected FRET ratio map:
Rcorrected = IFRET / IDD.
Diagram 2: Workflow for the standard 3-cube sensitized emission FRET analysis.
The Scientist's Toolkit: Key Reagents & Materials
Table 2: Essential Research Reagent Solutions for FRET Crosstalk Correction
| Item | Function & Role in Correction | Example Product/Construct |
|---|---|---|
| Donor-Only Plasmid | Expresses the donor fluorophore alone. Critical for empirical measurement of donor bleed-through coefficient (α). | pCS2-CFP, mTurquoise2-N1. |
| Acceptor-Only Plasmid | Expresses the acceptor fluorophore alone. Critical for empirical measurement of direct excitation coefficient (β). | pCS2-YFP, cpVenus-N1. |
| FRET Biosensor Plasmid | The experimental construct linking donor and acceptor via a metabolite-sensing domain. | AT1.03 (cAMP), iGluSnFR (glutamate). |
| Cell Line with Low Autofluorescence | Minimizes background noise, improving accuracy of coefficient determination and biosensor readout. | HEK293T, HeLa, CHO-K1. |
| Validated Filter Sets | Microscope filters with minimal bleed-through. Optimized for specific fluorophore pairs (e.g., CFP/YFP). | Chroma 89002 set (CFP/YFP/FRET). |
| Image Analysis Software | Enables pixel-math operations for applying correction formulas and ratio image generation. | Fiji/ImageJ, MetaMorph, NIS-Elements. |
Impact on Drug Development Research
Accurate crosstalk correction transforms FRET biosensors from qualitative indicators to quantitative tools. In high-content screening for drug discovery, it allows for:
- Precise EC50/IC50 Determination: Accurate dose-response curves for metabolites under drug modulation.
- Detection of Subtle Modulations: Reliably identifying weak allosteric modulators by small changes in FRET ratio.
- Kinetic Analysis: Correcting for photobleaching differences between channels to obtain true binding/unbinding kinetics. Failure to correct leads to systematic errors where apparent FRET changes may reflect alterations in biosensor expression level rather than metabolite concentration, potentially derailing target validation efforts.
Within the framework of FRET (Förster Resonance Energy Transfer) biosensor research for metabolite detection, achieving accurate absolute concentration measurements is paramount. The choice between in vitro and in situ calibration strategies fundamentally influences data interpretation, sensor performance validation, and biological relevance. This guide details the technical principles, methodologies, and applications of both approaches, providing a roadmap for researchers and drug development professionals.
Core Principles and Definitions
Absolute Concentration Measurement: The quantification of target analyte concentration in meaningful physical units (e.g., µM, nM) within a biological sample, as opposed to relative fluorescence changes.
In Vitro Calibration: The biosensor is characterized in a purified, controlled environment outside the cellular context. A known titration of the target metabolite is applied to the biosensor protein in solution, and the resulting FRET response is recorded to generate a standard curve.
In Situ Calibration: The biosensor is calibrated within its operational cellular environment. This involves manipulating intracellular metabolite levels to known values and measuring the concomitant FRET response, thereby accounting for cellular factors that influence sensor performance.
In Vitro Calibration: Methodology and Protocol
Experimental Protocol
Objective: To determine the in vitro apparent dissociation constant (Kdapp), dynamic range (ΔR), and saturation points of the FRET biosensor.
Key Reagents & Materials:
- Purified FRET biosensor protein.
- High-purity target metabolite standard.
- Assay buffer (mimicking physiological pH and ionic strength, e.g., 20 mM HEPES, 100 mM KCl, pH 7.2).
- Microplate reader or fluorometer capable of dual-emission measurement.
- Black-walled 96- or 384-well plates.
Procedure:
- Sensor Preparation: Dilute purified biosensor to a consistent, low-nanomolar concentration in assay buffer. The concentration must be well below the expected Kd to avoid ligand depletion.
- Metabolite Titration Series: Prepare a serial dilution of the metabolite standard, typically covering a range from zero to saturating concentration (e.g., 0 to 1 mM in 12-16 steps).
- Plate Setup: In each well, combine a fixed volume of biosensor solution with a varying volume of metabolite solution. Adjust with assay buffer to a constant final volume.
- FRET Measurement: Incubate to equilibrium (5-15 min). Excite the donor fluorophore (e.g., CFP at ~433 nm) and measure emission intensities at both donor (e.g., CFP at ~475 nm) and acceptor (e.g., YFP at ~527 nm) channels.
- Data Processing: Calculate the FRET ratio (R = IAcceptor / IDonor) for each well. Normalize ratios to the zero-metabolite value (Rmin) or the saturating-metabolite value (Rmax).
Data Analysis and Standard Curve
The normalized FRET response (Y) is typically fit to the Hill equation: Y = (Rmax - Rmin) * [L]n / (Kdapp + [L]n) + Rmin where [L] is metabolite concentration, and n is the Hill coefficient.
Table 1: Example In Vitro Calibration Parameters for Hypothetical FRET Biosensors
| Biosensor (Target) | Kdapp (µM) | Dynamic Range (ΔR/Rmin) | Hill Coefficient (n) | Reference Buffer |
|---|---|---|---|---|
| FLIP-ATP (ATP) | 5.2 ± 0.3 | 1.8 | 1.1 | 20 mM HEPES, 100 mM KCl, pH 7.2 |
| Sweetie (Glucose) | 850 ± 50 | 2.5 | 1.0 | Intracellular mimic buffer |
| iNap (cAMP) | 0.15 ± 0.02 | 3.0 | 0.9 | PBS, 1 mM Mg2+ |
Diagram 1: In vitro calibration experimental workflow.
In Situ Calibration: Methodology and Protocol
Rationale and Challenges
Cellular milieu can alter biosensor performance due to factors like macromolecular crowding, pH, competing metabolites, and post-translational modifications. In situ calibration aims to define the functional standard curve inside the cell.
CommonIn SituStrategies
A. Perfusion/Sonication with Calibration Buffers (For Ionomic Sensors):
- Protocol: Cells expressing the biosensor are permeabilized (e.g., with digitonin) and perfused with calibration buffers containing known concentrations of the target ion (e.g., Ca2+, Mg2+) and ionophores (e.g., ionomycin for Ca2+). The FRET response at each defined concentration is recorded via live-cell microscopy.
B. Metabolic Clamping (For Metabolite Sensors):
- Protocol: Cellular metabolism is manipulated to clamp intracellular metabolite levels at known steady-states.
- For ATP/ADP sensors: Treat cells with a series of metabolic inhibitors (e.g., oligomycin, 2-deoxyglucose) and uncouplers (e.g., FCCP) to titrate ATP levels from fully depleted to maximal.
- For glucose sensors: Use glucose transport inhibitors and varying external glucose concentrations in conjunction with glycolytic inhibitors.
C. Ratiometric Reference Calibration (e.g., with pHluorin):
- Protocol: Co-express the FRET biosensor with a ratiometric reference sensor (e.g., pHluorin for pH) that is sensitive to a common cellular confounder. This allows for parallel correction and calibration.
Data Interpretation
In situ calibration curves often show a right- or left-shifted Kd and a compressed dynamic range compared to in vitro curves.
Table 2: Comparison of Calibration Strategies
| Feature | In Vitro Calibration | In Situ Calibration |
|---|---|---|
| Environment | Controlled, purified buffer | Complex, living cell |
| Accounts for Cellular Factors | No | Yes |
| Technical Difficulty | Low to Moderate | High |
| Primary Output | Intrinsic sensor parameters (Kdapp, ΔR) | Functional cellular standard curve |
| Best For | Sensor characterization, optimization, & quality control | Absolute quantification in biological experiments |
| Key Assumption | Sensor behaves identically in vitro and in cellulo | Calibration maneuver does not alter other sensor-influencing variables |
Diagram 2: Decision tree for calibration strategy selection.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Reagents for FRET Biosensor Calibration
| Item | Function | Example/Specification |
|---|---|---|
| Purified Biosensor Protein | The core sensing element for in vitro characterization. | His- or GST-tagged, >90% purity, validated activity. |
| Metabolite Standard | High-purity ligand for generating standard curves. | ≥99% purity (HPLC-grade), prepared in appropriate solvent. |
| Physiological Assay Buffer | Mimics intracellular conditions for in vitro tests. | Contains relevant ions (K⁺, Mg²⁺), pH buffer (HEPES, PIPES), and reducing agents (DTT). |
| Permeabilization Agent | Creates pores in cell membrane for in situ clamping. | Digitonin, Streptolysin O, or saponin. |
| Metabolic Modulators | Titrate intracellular metabolite levels in situ. | Inhibitors (Oligomycin, 2-DG), Uncouplers (FCCP), Ionophores (Ionomycin). |
| Ratiometric Reference Sensor | Controls for non-specific cellular effects. | pHluorin (for pH), FRET-based reference sensors. |
| Live-Cell Imaging Medium | Maintains cell health during in situ calibration. | Phenol-red free, with stable pH and physiological nutrients. |
Integrated Workflow for Reliable Absolute Quantification
The most rigorous approach combines both strategies:
- Characterize the biosensor in vitro to establish baseline performance.
- Perform a careful in situ calibration for each cell type or experimental condition when possible.
- Apply the in situ standard curve to convert experimental FRET ratios into absolute concentrations.
- Report both the in vitro Kd and the details of any in situ calibration method used, acknowledging potential limitations.
In FRET-based metabolite detection research, the path to credible absolute concentration data is defined by calibration strategy. While in vitro calibration is essential for sensor development, in situ calibration provides the biologically relevant standard curve necessary for definitive quantitative biology. The choice is not merely technical but foundational to the interpretation of metabolic dynamics in health, disease, and drug response.
Within FRET biosensor metabolite detection research, achieving robust, reproducible, and physiologically relevant signals hinges on the precise optimization of biosensor expression in live cells. This whitepaper provides an in-depth technical guide on two critical pillars of this process: the strategic selection of promoters to control expression levels and the rigorous selection of clonal cell lines. Proper execution of these techniques is fundamental for generating high-quality, quantitative data on metabolite flux and dynamics.
Genetically encoded FRET biosensors are powerful tools for visualizing metabolite concentrations and signaling events in real time. However, inconsistent or excessive expression of the biosensor protein can lead to artifacts, including buffering of the target metabolite, cellular toxicity, and poor signal-to-noise ratios. The optimization of sensor expression through promoter choice and clonal selection is therefore not merely a procedural step but a core experimental determinant of data fidelity. This guide details the methodologies to systematically address this challenge.
Promoter Choice: Tuning Expression Levels
The promoter drives the initial level of biosensor transcription. Selection is based on the desired expression strength, cell type specificity, and experimental timeline.
Common Promoter Options
Diagram Title: Promoter Selection Decision Tree for FRET Biosensors
Table 1: Quantitative Comparison of Common Promoters for Biosensor Expression
| Promoter | Relative Strength (Typical) | Best Use Case | Key Consideration |
|---|---|---|---|
| CMV/CAG | Very High (100%) | Transient transfection for rapid assessment; low-expressing cell types. | High risk of sensor overexpression artifacts and silencing in some cell types. |
| EF1α | High-Moderate (~70-80%) | Generation of stable cell lines; consistent long-term expression. | Often provides reliable, sustained expression with lower toxicity risk than CMV. |
| PGK | Moderate (~50%) | Stable expression where moderate levels are sufficient. | Less prone to silencing than viral promoters in certain stem or primary cells. |
| TRE (Tet-On) | Inducible (Low to High) | Precise temporal control of sensor expression; toxic metabolites. | Requires stable line with rtTA; baseline leakiness must be characterized. |
| Cell-Specific | Variable | In vivo or co-culture studies targeting specific cell populations. | Complexity of delivery and validation; expression level is fixed by native regulation. |
Protocol: Evaluating Promoter Efficacy
Objective: To compare biosensor expression levels and FRET performance driven by different promoters. Materials: See "Scientist's Toolkit" below. Procedure:
- Cloning: Subclone your FRET biosensor cDNA (e.g., AT1.03 for cAMP, iNap for NADH) into mammalian expression vectors containing the promoters to be tested (e.g., pCAG, pEF1α, pPGK).
- Transfection: Seed HeLa or HEK293T cells in a 24-well plate with glass coverslips. At 60-80% confluency, transfert each promoter-biosensor construct using a standardized lipid-based method (e.g., 500 ng DNA, 1.5 µL Lipofectamine 3000 per well). Include an empty vector control.
- Sample Preparation: 24-48 hours post-transfection, wash cells with PBS and fix with 4% PFA for 15 min for initial screening. For live-cell analysis, transfer cells to imaging medium.
- Image Acquisition & Quantification:
- Use an epifluorescence or confocal microscope with appropriate filter sets for donor (CFP), FRET, and acceptor (YFP) channels.
- Acquire images of multiple fields per condition using identical exposure times.
- Quantify Expression Level: Measure the mean YFP (or CFP) fluorescence intensity per cell as a proxy for biosensor concentration. Plot as a histogram.
- Quantify FRET Performance: Calculate the basal FRET ratio (e.g., YFP/CFP emission ratio upon CFP excitation) for each cell. Assess the dynamic range by imaging cells under conditions of maximal and minimal metabolite concentration (e.g., forskolin/IBMX for cAMP sensors, glucose starvation for ATP sensors).
- Analysis: The optimal promoter yields a population of cells with moderate fluorescence intensity and a high, consistent basal FRET ratio with a robust dynamic range. High-intensity cells with a compressed dynamic range indicate overexpression artifacts.
Clonal Selection: Isolating a Uniform Population
Even with an optimal promoter, transfection and random genomic integration create heterogeneous expression. Clonal selection is essential for generating stable, uniform cell lines.
Workflow for Generating Clonal Stable Cell Lines
Diagram Title: Stable Clonal Cell Line Generation Workflow
Protocol: Single-Cell Cloning and Screening by FACS
Objective: To isolate and characterize monoclonal cell lines with uniform and functional FRET biosensor expression. Materials: See "Scientist's Toolkit" below. Procedure:
- Generate Polyclonal Pool: Transfect your chosen promoter-biosensor construct along with a puromycin (or other) resistance plasmid, or use a single vector containing the resistance gene. Begin antibiotic selection (e.g., 2 µg/mL puromycin) 48 hours post-transfection for 5-7 days.
- Prepare for FACS: Harvest the polyclonal pool using trypsin, resuspend in complete growth medium, and filter through a 35-40 µm cell strainer.
- Single-Cell Sorting: Using a FACS sorter with a 100 µm nozzle, gate for viable cells (based on FSC/SSC). Set a gate for cells expressing moderate levels of the biosensor (e.g., the middle 40-60% of YFP-positive signal). Sort one cell per well into a 96-well plate pre-filled with 150 µL of conditioned growth medium.
- Clone Expansion: Grow sorted cells at 37°C, 5% CO₂ for 2-3 weeks, refreshing medium carefully every 3-4 days. Visually identify wells containing single colonies.
- Primary High-Throughput Screening:
- Transfer clones to 24-well plates with glass-bottom inserts for imaging.
- Using an automated microscope, acquire images of the basal FRET/CFP/YFP channels for each clone.
- Key Metrics: Calculate for each clone: (a) the mean biosensor expression (YFP intensity), (b) the uniformity of expression (coefficient of variation, CV, of YFP intensity across cells), and (c) the uniformity of the basal FRET ratio (CV of the ratio across cells).
- Secondary Validation:
- Expand 5-10 top candidate clones (showing moderate mean expression and low CVs).
- Perform full live-cell FRET assays to determine the dynamic range (response to saturating stimulus/inhibitor) and sensitivity (EC50/IC50) of the biosensor in each clone.
- Test response stability over multiple passages (e.g., P5, P10, P15).
- Selection & Banking: Choose the clone that demonstrates optimal uniformity, a robust dynamic range matching the biosensor's expected performance, and stable expression over at least 10 passages. Create a master cell bank.
Table 2: Quantitative Clonal Selection Criteria
| Screening Stage | Key Parameter | Optimal Value | Measurement Tool |
|---|---|---|---|
| Primary (Imaging) | Expression Level (Fluor. Intensity) | Moderate (e.g., 2-5x over autofluorescence) | Mean YFP/CFP intensity per cell |
| Expression Uniformity | Low (CV < 15-20%) | CV of YFP/CFP intensity across clone | |
| Basal FRET Ratio Uniformity | Very Low (CV < 10%) | CV of basal FRET ratio across clone | |
| Secondary (Validation) | Dynamic Range (ΔR/R₀) | High (Matches literature for biosensor) | (Rmax - Rmin) / R_min |
| Sensitivity (EC50/IC50) | Matches expected physiology | Dose-response curve fitting | |
| Stability | Consistent over ≥10 passages | Periodic FRET assay |
The Scientist's Toolkit: Key Reagent Solutions
| Item/Reagent | Function in Optimization | Example Product/Catalog # (Illustrative) |
|---|---|---|
| Mammalian Expression Vectors | Backbone for cloning biosensor under different promoters. | pCAG, pEF1α-IRES-Puro, pLV-TRE, pcDNA3.1 |
| Lipid-Based Transfection Reagent | For transient transfection and initial stable pool generation. | Lipofectamine 3000, Fugene HD, Polyethylenimine (PEI) |
| Fluorescent Protein-Specific Antibodies | Validation of biosensor expression by Western blot. | Anti-GFP (cross-reacts with YFP/CFP), Anti-RFP |
| Selection Antibiotics | For stable pool and clone selection. | Puromycin, Geneticin (G418), Hygromycin B |
| FACS Sorter with Single-Cell Dispenser | Precise isolation of single cells into multi-well plates. | BD FACSAria, Beckman Coulter MoFlo Astrios |
| Conditioned Medium | Supports survival and growth of single cells after sorting. | Filtered supernatant from untransfected, confluent cultures. |
| Automated Live-Cell Imaging System | High-throughput screening of clonal FRET responses. | ImageXpress Micro, Incucyte, Opera Phenix |
| FRET Calibration Standards | Controls for microscope FRET channel sensitivity. | Cells expressing CFP only, YFP only, or linked CFP-YFP. |
| Metabolite Modulators | To test biosensor dynamic range in clones. | Forskolin (cAMP), Oligomycin (ATP), Ionophores (Ca²⁺, pH). |
Validating FRET Biosensor Data: Comparative Analysis with Complementary Techniques
In the rigorous field of FRET (Förster Resonance Energy Transfer) biosensor metabolite detection, establishing robust validation controls is paramount for generating credible, interpretable, and biologically relevant data. While the design of the biosensor itself is critical, the experimental framework for its validation dictates the reliability of the conclusions drawn. This technical guide details the three essential pillars of validation—Specificity, Reversibility, and Dose-Response—within the context of FRET biosensor research, providing researchers with the protocols and analytical tools necessary for definitive characterization.
Specificity: Establishing Causal Linkage
Specificity controls confirm that the observed FRET ratio change is directly and exclusively caused by the target metabolite interacting with the biosensor's sensing domain, and not by artifacts such as pH fluctuations, osmolarity changes, or interference from structurally similar molecules.
Experimental Protocol: Specificity Challenge
Objective: To test biosensor response against the target analyte and a panel of potential interferents.
Procedure:
- Cell Preparation: Plate cells (e.g., HEK293T, HeLa) expressing the FRET biosensor in an imaging-compatible dish. Allow for adherence and expression (12-48 hrs).
- Baseline Acquisition: Acquire fluorescence intensity images for donor (CFP, e.g., 445 nm ex / 480 nm em) and acceptor (YFP, e.g., 515 nm em) channels under standard imaging buffer. Calculate the baseline FRET ratio (Acceptor Intensity / Donor Intensity).
- Analyte Application: Perfuse cells with imaging buffer containing the target metabolite at a physiologically relevant concentration (e.g., 1 mM Glucose for a glucose biosensor). Monitor FRET ratio for 5-10 minutes.
- Interferent Application: After washout and recovery, sequentially apply buffers containing potential interferents at equivalent or higher concentrations. For a cAMP biosensor: Apply cGMP, AMP, adenosine. For a glucose biosensor: Apply mannose, fructose, 2-DG. Universal controls: Apply buffers with altered pH (±0.5 units) or osmolarity (±50 mOsm).
- Data Analysis: Quantify the maximum ΔFRET ratio (% or absolute change) for each condition. A specific biosensor shows a significant response only to the target.
Table 1: Example Specificity Test for a Hypothetical cAMP FRET Biosensor (Epac1-camps). ΔFRET ratio represents mean ± SEM (n=20 cells).
| Compound Applied | Concentration | Mean ΔFRET Ratio (%) | Significant Response (p<0.01) |
|---|---|---|---|
| cAMP | 10 µM | +35.2 ± 1.5 | Yes |
| Forskolin (AC activator) | 50 µM | +32.8 ± 2.1 | Yes |
| cGMP | 100 µM | +1.2 ± 0.8 | No |
| AMP | 1 mM | +0.5 ± 0.3 | No |
| Buffer pH 6.8 | - | -0.9 ± 0.6 | No |
| Buffer pH 7.8 | - | +1.1 ± 0.7 | No |
Reversibility: Confirming Dynamic Range
Reversibility demonstrates the biosensor's ability to return to its baseline FRET state upon removal of the analyte, confirming its utility for monitoring dynamic fluctuations in metabolite levels and indicating a lack of sensor saturation or permanent perturbation.
Experimental Protocol: Pulse-Challenge Assay
Objective: To assess the kinetic on/off rates and full recovery of the FRET signal.
Procedure:
- Baseline & Stimulation: Establish a stable baseline FRET ratio in imaging buffer. Apply a pulse of the target metabolite (via precise perfusion system) for a defined period (e.g., 60 seconds).
- Washout & Monitoring: Rapidly switch back to analyte-free imaging buffer. Continue acquiring donor and acceptor images for a period 3-5x the stimulation length (e.g., 5 minutes).
- Kinetic Analysis: Plot FRET ratio versus time. Fit the recovery phase to an exponential decay model to calculate the off-rate (τ_off).
- Multiple Cycles: For robust sensors, repeat the pulse-challenge cycle 2-3 times to demonstrate consistent performance without photobleaching or desensitization.
Table 2: Reversibility Parameters for Metabolite Biosensors. τ_on and τ_off represent time constants. Data compiled from recent literature.
| Biosensor Target | Sensor Name | τ_on (s) | τ_off (s) | % Recovery after 300s |
|---|---|---|---|---|
| Glucose | FLII12Pglu-700μδ6 | 25 ± 3 | 45 ± 5 | 98.5 ± 0.7 |
| Lactate | Laconic | 15 ± 2 | 120 ± 15 | 95.2 ± 1.2 |
| ATP:ADP Ratio | PercevalHR | 2 ± 0.5 | 5 ± 1 | 99.1 ± 0.5 |
| cAMP | cADDis | 10 ± 1 | 30 ± 4 | 97.8 ± 0.9 |
Dose-Response: Quantifying Affinity and Range
A dose-response curve defines the operational range, apparent affinity (Kd), and dynamic range (ΔRmax) of the biosensor. It is essential for interpreting the magnitude of FRET changes in terms of physiological analyte concentrations.
Experimental Protocol: In-situ Titration
Objective: To determine the relationship between analyte concentration and FRET response in the cellular environment.
Procedure:
- Calibration Setup: Use cells expressing the biosensor. For permeable metabolites (e.g., H2O2), use buffers with incrementally increasing concentrations. For impermeable metabolites or second messengers (e.g., cAMP, Ca²⁺), use ionophores (e.g., ionomycin for Ca²⁺) or membrane-permeable analogs/activators in combination with enzyme inhibitors to clamp intracellular levels.
- Stepwise Application: Perfuse cells with the lowest concentration until a stable FRET plateau is reached (5-10 mins). Move to the next higher concentration without washout (cumulative addition).
- Data Fitting: For each cell, normalize FRET ratio (R) to baseline (R0) or min/max. Plot normalized response (R/R0 or ΔR/R0) against log[Analyte]. Fit the aggregated data to a sigmoidal dose-response curve (e.g., Hill equation):
Response = ΔR_min + (ΔR_max - ΔR_min) / (1 + (K_d / [Analyte])^n_H). - Key Outputs: Extract the apparent Kd (concentration at half-maximal response), Hill coefficient (nH, indicating cooperativity), and dynamic range (ΔR_max).
Table 3: Characterized Dose-Response Parameters for Common FRET Biosensors.
| Biosensor | Target | Apparent K_d | Dynamic Range (ΔR/R0) | Hill Coefficient (n_H) | Physiological Range |
|---|---|---|---|---|---|
| FLII12Pglu-700μδ6 | Glucose | 700 µM | 1.4 | 1.0 | 0-10 mM |
| iATPSnFR1 | ATP | 2.5 mM | 3.8 | 1.2 | 0.1-10 mM |
| HyPer7 | H2O2 | 1.1 µM | 5.0 | 1.0 | 1 nM - 100 µM |
| G-Flamp1 | Glutamate | 12 µM | 0.9 | 1.0 | 1-100 µM |
| Epac1-camps | cAMP | 9.4 µM | 0.35 | 1.0 | 0.1-10 µM |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for FRET Biosensor Validation Experiments.
| Reagent / Material | Function / Purpose |
|---|---|
| Genetically-Encoded FRET Biosensor Plasmid (e.g., FLII12Pglu-700μδ6, Epac1-camps) | The core tool; encodes the donor-acceptor fluorescent protein pair linked by a metabolite-responsive domain. |
| Lipofectamine 3000 or Polyethylenimine (PEI) | Transfection reagents for delivering biosensor plasmid into mammalian cell lines. |
| Live-Cell Imaging Buffer (Phenol Red-free) | Maintains cell health during imaging. Often contains HEPES (pH 7.4), salts, and glucose/serum substitute. |
| Precision Perfusion System (e.g., ValveLink8) | Enables rapid, precise, and repeatable exchange of extracellular solutions for stimulation and washout. |
| Target Metabolite & Analog Library (e.g., cAMP, cGMP, 2-DG, various sugars) | For specificity challenges and dose-response calibration. |
| Pharmacologic Modulators (e.g., Forskolin, IBMX, Ionophores) | To clamp or modulate intracellular levels of metabolites/second messengers for calibration. |
| Inverted Fluorescence Microscope with:- 40x/60x Oil-immersion Objective- Dual- or Multi-band Emission Filter Set (e.g., CFP/YFP)- Stable LED/Laser Light Source- sCMOS Camera | Essential hardware for high-sensitivity, time-lapse FRET ratio imaging with minimal photobleaching. |
| FRET Ratio Image Analysis Software (e.g., ImageJ/FIJI with RatioPlus, MetaFluor, custom Python/Matlab scripts) | To calculate and analyze time-series of acceptor/donor intensity ratios from raw image data. |
Visualizing Experimental Workflows and Principles
Diagram Title: Specificity Validation Workflow for FRET Biosensors
Diagram Title: Key Parameters from a Biosensor Dose-Response Curve
Diagram Title: FRET Biosensor Mechanism Linked to Validation Pillars
In the development and validation of Förster Resonance Energy Transfer (FRET)-based biosensors for metabolite detection, rigorous benchmarking against established analytical methods is paramount. This guide details the technical processes for comparing novel FRET sensor performance against the gold standards of enzymatic assays and mass spectrometry (MS). The reliability of a new biosensor hinges on its correlation with these definitive techniques, which provide the reference data for sensitivity, specificity, dynamic range, and temporal resolution.
Gold Standard 1: Enzymatic Assays
Enzymatic assays are a cornerstone of specific metabolite quantification, relying on the high specificity of enzymes coupled to spectrophotometric or fluorometric readouts.
Core Principle
The target metabolite participates in or is consumed by an enzyme-catalyzed reaction, leading to a stoichiometric change in a cofactor (e.g., NADH/NAD⁺, ATP/ADP) that can be measured optically. The rate or endpoint of this change is directly proportional to the metabolite concentration.
Detailed Protocol: Lactate Dehydrogenase (LDH) Assay for Lactate Quantification
This protocol is commonly used to validate FRET-based lactate biosensors.
Objective: Quantify lactate concentration in cell lysate or medium. Reagents:
- Glycine-Hydrazine Buffer (0.4 M glycine, 0.4 M hydrazine, pH 9.2)
- NAD⁺ solution (27 mM in DI water)
- Lactate Dehydrogenase (LDH, from porcine heart, ~1,000 U/mL)
- Sodium Lactate standard (100 mM)
Procedure:
- Sample Preparation: Deproteinize biological samples (e.g., cell culture medium) by adding perchloric acid (0.6 M final), vortexing, incubating on ice for 10 min, and centrifuging at 12,000 x g for 5 min. Neutralize the supernatant with 2 M KOH/0.3 M MOPS. Centrifuge again to remove KClO₄ precipitate.
- Standard Curve: Prepare a dilution series of sodium lactate standard (e.g., 0, 0.5, 1, 2, 4 mM) in assay buffer.
- Reaction Setup: In a quartz cuvette, mix:
- 950 µL Glycine-Hydrazine Buffer
- 20 µL NAD⁺ solution
- 20 µL sample or standard
- Baseline Reading: Record absorbance at 340 nm (A₃₄₀) for 1 minute.
- Reaction Initiation: Add 10 µL of LDH enzyme solution, mix rapidly.
- Measurement: Monitor A₃₄₀ until it stabilizes (typically 10-30 min). The hydrazine traps the generated pyruvate, driving the reaction to completion.
- Calculation: Calculate ΔA₃₄₀ (Final A₃₄₀ - Initial A₃₄₀). Plot ΔA₃₄₀ of standards against concentration. The lactate concentration in the unknown sample is determined from the linear regression of the standard curve.
Gold Standard 2: Mass Spectrometry (MS)
MS offers unparalleled specificity and the ability to perform multiplexed, untargeted metabolite profiling. Liquid Chromatography-MS (LC-MS) is the most common platform for quantitative metabolomics.
Core Principle
Metabolites are separated by liquid chromatography (LC), ionized (typically by electrospray ionization, ESI), and separated in the mass spectrometer based on their mass-to-charge ratio (m/z). Quantification is achieved by comparing the ion intensity (peak area) of the target metabolite to a spiked, isotopically labeled internal standard.
Detailed Protocol: Targeted LC-MS/MS for ATP, ADP, and AMP Quantification
This protocol validates energy charge measurements from FRET biosensors like ATeam.
Objective: Quantify adenine nucleotides in cell extracts. Reagents & Materials:
- Extraction Solvent: 80% methanol/20% water (v/v), pre-chilled to -80°C.
- Internal Standard Solution: ¹³C₁₀,¹⁵N₅-ATP, ¹³C₁₀,¹⁵N₅-ADP, ¹³C₁₀,¹⁵N₅-AMP in water.
- LC-MS grade water, methanol, acetonitrile.
- HPLC column: Reversed-phase C18 or HILIC column.
Procedure:
- Rapid Metabolite Extraction:
- Aspirate medium from cultured cells and immediately quench metabolism by adding -80°C extraction solvent (1 mL per 10⁶ cells).
- Scrape cells on dry ice and transfer suspension to a pre-cooled microcentrifuge tube.
- Vortex for 30 seconds, incubate at -80°C for 1 hour.
- Centrifuge at 16,000 x g for 15 min at 4°C.
- Transfer supernatant to a new tube. Dry under a gentle nitrogen stream or vacuum concentrator.
- Reconstitute dried extract in 100 µL of LC-MS compatible buffer (e.g., 5 mM ammonium acetate in water).
LC-MS/MS Analysis:
- Chromatography: Use a HILIC column (e.g., SeQuant ZIC-pHILIC) with a gradient from 80% acetonitrile/20% 10 mM ammonium carbonate (pH 9.2) to 50% acetonitrile/50% water. Flow rate: 0.2 mL/min.
- Mass Spectrometry: Operate in negative ion mode (ESI-). Use Multiple Reaction Monitoring (MRM) for highest sensitivity and specificity.
- MS Parameters: Set optimized collision energies and declustering potentials for each analyte. Example MRM transitions:
- ATP: 506 > 159, 506 > 408
- ADP: 426 > 134, 426 > 328
- AMP: 346 > 134, 346 > 97
- Corresponding labeled internal standards.
Data Analysis:
- Integrate peak areas for each analyte and its corresponding internal standard.
- Calculate the ratio (Analyte Peak Area / Internal Standard Peak Area).
- Generate a standard curve for each analyte using pure standards spiked with a constant amount of internal standard. Use linear regression to determine the concentration in the sample.
Benchmarking Workflow & Data Correlation
The validation process involves parallel measurement of biologically relevant samples using the novel FRET biosensor and the gold standard techniques.
Title: FRET Biosensor Validation Benchmarking Workflow
Data Presentation and Comparison
Table 1: Comparative Metrics of Metabolite Detection Techniques
| Feature | FRET Biosensor | Enzymatic Assay | LC-MS/MS |
|---|---|---|---|
| Primary Use | Real-time, live-cell dynamics | Specific, endpoint quantification | Specific, multiplexed profiling |
| Sensitivity | nM - µM range (dependent on probe Kd) | µM range (limited by A₃₄₀) | pM - nM range (highest) |
| Temporal Resolution | Milliseconds to seconds | Minutes to hours | Minutes per sample |
| Spatial Resolution | Subcellular (if targeted) | None (bulk lysate) | None (bulk extract) |
| Multiplexing | Limited (2-3 colors) | No (single analyte per assay) | High (100s of metabolites) |
| Throughput | High (live imaging) | Medium (plate reader) | Low to Medium |
| Key Advantage | Live-cell dynamics | Cost-effective, specific | Definitive identification & quantification |
Table 2: Example Benchmarking Data: Cytosolic ATP Concentration
| Cell Line | FRET Biosensor (ATeam) [mM] | Enzymatic (Luciferase) [mM] | LC-MS/MS [mM] | Correlation (R² vs. MS) |
|---|---|---|---|---|
| HEK293 | 2.8 ± 0.3 | 2.9 ± 0.4 | 3.1 ± 0.2 | 0.94 |
| HeLa | 2.1 ± 0.2 | 2.3 ± 0.3 | 2.4 ± 0.1 | 0.91 |
| Primary Neurons | 1.5 ± 0.4 | 1.6 ± 0.2 | 1.7 ± 0.2 | 0.89 |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for FRET Biosensor Benchmarking
| Item | Function/Description | Example Supplier/Product |
|---|---|---|
| Purified Metabolite Standards | For generating standard curves in enzymatic assays and LC-MS. Critical for absolute quantification. | Sigma-Aldrich (e.g., Sodium Lactate L7022, ATP A2383) |
| Stable Isotope-Labeled Internal Standards (SIL IS) | Spiked into samples for MS analysis to correct for ionization efficiency and matrix effects. Essential for robust quantification. | Cambridge Isotope Laboratories (e.g., ¹³C₁₀,¹⁵N₅-ATP) |
| High-Purity Enzymes for Assays | Catalyze the specific reaction in enzymatic assays. Purity is critical to avoid side-reactions. | Roche (Dehydrogenases, Kinases) |
| LC-MS Grade Solvents | Ultra-pure solvents (water, methanol, acetonitrile) to minimize background ions and contamination in MS. | Fisher Chemical (Optima grade) |
| Metabolite Extraction Kits | Standardized, optimized kits for efficient and reproducible quenching/extraction of metabolites for MS. | Biocrates, Metabolon |
| FRET Biosensor Plasmids | Genetically encoded sensors (e.g., ATeam for ATP, Laconic for lactate). The tool being validated. | Addgene (various deposits) |
| Cell Permeabilization Agents | Used in calibration protocols for FRET biosensors to clamp intracellular [metabolite] to known external levels. | Digitonin, β-escin, Streptolysin O |
Comparison with Single-Fluorescent Protein Biosensors (e.g., GCaMP for Calcium)
The development of genetically encoded biosensors has revolutionized our ability to visualize dynamic biochemical processes in living cells and organisms. Within this field, two dominant architectural paradigms have emerged: single-fluorescent protein (FP) biosensors (e.g., GCaMP for Ca²⁺) and Förster Resonance Energy Transfer (FRET)-based biosensors for metabolite detection. This whitepaper provides a technical comparison of these designs, framed within the broader thesis that FRET-based biosensors offer unique and complementary advantages for quantitative, multiparameter, and ratiometric metabolite sensing, despite the superior brightness and simplicity of single-FP designs for specific ions like calcium. Understanding their respective operational principles, performance metrics, and experimental requirements is crucial for researchers selecting the optimal tool for their biological question.
Operational Principles & Core Architecture
Single-FP Biosensors (Exemplified by GCaMP)
GCaMP is a fusion protein comprising a circularly permuted green fluorescent protein (cpGFP) sandwiched between Calmodulin (CaM) and a CaM-binding peptide (M13). Ca²⁺ binding to CaM induces a conformational change that wraps CaM around M13. This allosterically alters the cpGFP chromophore environment, dramatically increasing its fluorescence intensity.
FRET-Based Biosensors (For Metabolite Detection)
A typical FRET biosensor consists of a donor FP and an acceptor FP linked by a sensing domain specific to a target metabolite (e.g., glucose, cAMP, ATP). Metabolite binding induces a conformational change in the sensing domain, altering the distance and/or orientation between the donor and acceptor, thereby modulating the efficiency of energy transfer. The readout is the emission ratio of acceptor to donor fluorescence.
Quantitative Performance Comparison
Table 1: Key Performance Characteristics of Single-FP vs. FRET Biosensors
| Characteristic | Single-FP Biosensors (e.g., GCaMP6f/7/8) | FRET Biosensors (e.g., for Glucose, cAMP) | Implication for Research |
|---|---|---|---|
| Signal Type | Intensity-based change (ΔF/F0) | Ratiometric (Acceptor/Donor emission) | FRET signals are internally controlled, less sensitive to expression variance, focus drift, and photobleaching. |
| Dynamic Range (Δ) | Extremely high (e.g., GCaMP6f: ~200% ΔF/F; GCaMP8: up to 5200%) | Moderate to High (e.g., 30-200% ΔR/R) | Single-FP sensors excel in detecting small numbers of events; FRET offers quantitative precision. |
| Brightness | Very High (single bright FP) | Lower (signal split between two FPs) | Single-FP sensors are superior for low-expression systems or in vivo imaging where brightness is critical. |
| Spectral Channels | Single excitation/emission | Dual excitation and/or dual emission | FRET requires more complex optics and unmixing but enables multiparameter imaging with other probes. |
| Quantification | Semi-quantitative; sensitive to artifacts | More rigorously quantitative via ratioing | FRET is preferred for precise concentration estimation (e.g., metabolite levels). |
| Temporal Resolution | Very Fast (GCaMP6f: τdecay ~100-200 ms) | Fast, but often limited by kinetics of linker/sensor (τ ~seconds) | Single-FP sensors are optimal for high-speed kinetics (e.g., neuronal spikes). |
| Common Targets | Ions (Ca²⁺, H⁺, Cl⁻), some neurotransmitters | Metabolites (ATP, cAMP, glucose), lipids, kinase activity (Akt, ERK) | Target choice often dictates architecture: conformational sensors for metabolites, allosteric for ions. |
| In Vivo Applicability | Excellent (bright, simple signal) | Good, but more challenging due to spectral requirements | GCaMP dominates neuroscience; improved FRET pairs (e.g., cyan-yellow) enhance in vivo use. |
Table 2: Example Biosensor Specifications (Current Generations)
| Biosensor Name | Target | Type | Dynamic Range (ΔF/F or ΔR/R) | KD / EC50 | Key Reference/Resource |
|---|---|---|---|---|---|
| jGCaMP8m | Ca²⁺ | Single-FP (G) | ~5200% | ~100 nM | Dana et al., Nature 2019 |
| GCaMP7f | Ca²⁺ | Single-FP (G) | ~1100% | ~120 nM | Na et al., bioRxiv 2021 |
| FLII12Pglu-700μδ6 | Glucose | FRET (CFP/YFP) | ~70% ΔR/R | ~700 μM | Takanaga et al., JBC 2008 |
| QUEEN-2m | ATP | Single-FP (cpYFP) | ~12-fold intensity | ~3.3 mM | Yaginuma et al., Sci Rep 2023 |
| ATeam | ATP | FRET (CFP/YFP) | ~2.6-fold ratio | ~3.3 mM | Imamura et al., PNAS 2009 |
| cAMPdiff-FRET | cAMP | FRET (mTurq2/cp174Venus) | ~40% ΔR/R | ~1.8 μM | Klarenbeek et al., Nat Methods 2015 |
Experimental Protocols
Protocol 1: Imaging Intracellular Ca²⁺ Transients with GCaMP
A. Cell Preparation & Transfection
- Plate cells (e.g., HEK293, primary neurons) on imaging-grade dishes.
- Transfect with plasmid encoding GCaMP variant (e.g., pGP-CMV-GCaMP6f) using appropriate method (lipofection, electroporation, viral transduction). Allow 24-48 hrs for expression.
- For neurons, use synapsin promoter for neuron-specific expression.
B. Live-Cell Imaging Setup
- Use an epifluorescence or confocal microscope equipped with a 470-490 nm LED/laser line and a 500-550 nm emission filter.
- Maintain cells at 37°C and 5% CO₂ in physiological buffer (e.g., Hanks' Balanced Salt Solution, HBSS).
- Use a 40x or 60x oil-immersion objective.
C. Data Acquisition & Analysis
- Acquire images at 5-100 Hz, depending on kinetics of interest.
- Apply stimuli (e.g., KCl depolarization, agonist addition) during acquisition.
- Define Regions of Interest (ROIs) over cell bodies/processes.
- Calculate ΔF/F0 = (F - F0) / F0, where F0 is the baseline fluorescence averaged over a stable period.
Protocol 2: Quantifying Metabolite Dynamics with a FRET Biosensor
A. Cell Preparation & Co-transfection
- Plate cells as above.
- Co-transfect with plasmids encoding the FRET biosensor (e.g., ATeam for ATP) and optionally a localization marker. Use a 1:1 molar ratio if using separate donor/acceptor constructs (less common for integrated designs).
B. Dual-Emission Ratiometric Imaging
- Use a microscope capable of simultaneous dual-emission acquisition.
- Excitation: Use a 430-440 nm laser/LED for CFP.
- Emission: Split light onto two cameras or use a beamsplitter: 460-500 nm for donor (CFP) channel, 520-550 nm for acceptor (FRET) channel.
- Maintain environmental control.
C. Data Processing & Ratiometric Calculation
- Acquire time-series images for both channels.
- Perform background subtraction for each channel.
- Apply flat-field correction if necessary.
- Calculate the FRET ratio (R) = IntensityAcceptorChannel / IntensityDonorChannel for each pixel/time point.
- Express changes as ΔR/R0 = (R - R0) / R0, where R0 is the baseline ratio.
- Correct for bleed-through (cross-talk) and direct acceptor excitation by acquiring control cells expressing donor-only and acceptor-only constructs.
Signaling Pathway & Workflow Diagrams
Diagram 1: Core Signaling Mechanisms of Biosensor Types
Diagram 2: Biosensor Selection Workflow for Researchers
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions & Materials
| Item | Category | Function & Application | Example Product/Vector |
|---|---|---|---|
| GCaMP Expression Plasmid | Molecular Biology | Drives expression of single-FP Ca²⁺ sensor in cells. | pGP-CMV-GCaMP8m (Addgene #162375) |
| FRET Biosensor Plasmid | Molecular Biology | Drives expression of integrated donor-sensor-acceptor construct. | pLyn-FLII12Pglu-700μδ6 (Addgene #17866) |
| Genetically Encoded cAMP Sensor | Molecular Biology | FRET-based sensor for cyclic AMP dynamics. | pcDNA3-cAMPdiff-FRET (Addgene #79987) |
| ATeam (ATP FRET Sensor) | Molecular Biology | Quantifies ATP:ADP ratio in living cells. | pCMV-AT1.03 (Addgene #51958) |
| Lipofectamine 3000 | Transfection | Lipid-based reagent for plasmid delivery into mammalian cells. | Thermo Fisher Scientific L3000001 |
| Neurobasal/B-27 Medium | Cell Culture | Optimized medium for primary neuron culture and imaging. | Gibco Neurobasal + B-27 Supplement |
| HBSS (Imaging Buffer) | Buffers & Salts | Physiological salt solution for live-cell imaging experiments. | Hanks' Balanced Salt Solution, Ca²⁺/Mg²⁺ |
| Ionomycin / Iono. Cocktail | Pharmacological Agent | Ca²⁺ ionophore used for sensor calibration (max signal). | MilliporeSigma 407952 |
| EGTA / BAPTA-AM | Chelators | Ca²⁺ chelators used for sensor calibration (min signal). | Thermo Fisher Scientific E1219 |
| 2-Deoxy-D-Glucose & Antimycin A | Metabolic Inhibitors | Used to deplete ATP for calibration of ATP sensors. | MilliporeSigma D6134 & A8674 |
| CellMask Deep Red | Plasma Membrane Stain | Labels cell morphology for ROI definition; spectrally distinct from GFP/YFP/CFP. | Thermo Fisher Scientific C10046 |
| Matrigel / Poly-D-Lysine | Coating Reagents | Coat imaging dishes to improve cell adhesion, especially for neurons. | Corning 354230 / MilliporeSigma A3890401 |
Strengths and Limitations vs. Fluorescent Dye-Based Metabolite Probes
In the context of developing Förster Resonance Energy Transfer (FRET)-based biosensors for metabolite detection, understanding the relative merits of different probe technologies is paramount. This whitepaper provides a technical comparison between genetically-encoded FRET biosensors and synthetic fluorescent dye-based metabolite probes, focusing on their application in live-cell imaging and drug discovery research.
Comparative Analysis of Probe Technologies
The following tables summarize key quantitative and qualitative parameters for each class of probe.
Table 1: Performance and Practical Characteristics
| Characteristic | Genetically-Encoded FRET Biosensors | Synthetic Fluorescent Dye-Based Probes |
|---|---|---|
| Spatial Targeting | Precise (genetically targetable to organelles) | Limited (often reliant on chemical properties) |
| Temporal Resolution | High (seconds to minutes for dynamic imaging) | Variable (seconds to hours; depends on loading/washing) |
| Quantitative Output | Ratiometric (FRET ratio minimizes artifacts) | Often intensity-based (prone to artifact) |
| In Vivo Applicability | Excellent (transgenic organisms possible) | Challenging (delivery and clearance issues) |
| Multiplexing Potential | Moderate (limited by fluorescent protein spectra) | High (broad palette of synthetic dyes available) |
| Typical Development Time | Long (months to years for optimization) | Shorter (weeks to months for synthesis) |
| Cost per Experiment | Low (after initial construct) | High (recurring reagent cost) |
Table 2: Analytical Metrics (Typical Ranges)
| Metric | Genetically-Encoded FRET Biosensors | Synthetic Fluorescent Dye-Based Probes |
|---|---|---|
| Dynamic Range (ΔR/Rmax) | 10% - 50% | 50% - 500%+ |
| Affinity (Kd) | Tunable (nM to mM range possible) | Fixed post-synthesis (nM to μM common) |
| Photostability | Moderate to Low | High (especially with newer dyes) |
| Brightness | Moderate | Very High |
| Cellular Perturbation | Low (native expression possible) | Potentially High (loading concentrations, chemical effects) |
Experimental Protocols
Protocol 1: Live-Cell Ratiometric Imaging with a FRET Biosensor (e.g., for Glucose)
- Cell Preparation: Plate cells expressing the biosensor (e.g., FLII12Pglu-700μδ6) in a glass-bottom dish.
- Imaging Setup: Use a confocal or widefield microscope equipped with dual-emission capability. Set excitation for the donor (e.g., 433 nm for CFP). Collect emission simultaneously at 475 nm (CFP channel) and 527 nm (FRET/YFP channel) using a beam splitter.
- Calibration: Acquire baseline images. Perfuse with calibration solutions: 1) 0 mM glucose + 2-deoxyglucose to obtain Rmin, 2) 30 mM glucose to obtain Rmax.
- Data Acquisition: Perform time-lapse imaging during experimental treatments. Calculate the FRET ratio (R = FRET channel intensity / CFP channel intensity) for each time point.
- Analysis: Normalize ratios: Normalized R = (R - Rmin) / (Rmax - Rmin). Convert to metabolite concentration using the known Kd of the biosensor.
Protocol 2: Intracellular Metabolite Tracking with a Synthetic Dye Probe (e.g., ROS Detection with H2DCFDA)
- Probe Loading: Prepare a 10 mM stock of H2DCFDA in DMSO. Wash cells with serum-free buffer. Load cells with 5-20 μM H2DCFDA in buffer for 30-45 minutes at 37°C in the dark.
- Washing: Thoroughly wash cells 2-3 times with fresh buffer to remove extracellular dye and esterases.
- Imaging Setup: Use a microscope with FITC filter sets (Ex ~488 nm, Em ~525 nm). Note: This is an intensity-based, non-ratiometric probe.
- Data Acquisition: Begin time-lapse imaging immediately after adding experimental stimuli. Minimize light exposure to prevent photobleaching and dye oxidation artifacts.
- Analysis: Quantify mean fluorescence intensity over time within regions of interest. Normalize to baseline (F/F0). Include appropriate controls (untreated, antioxidant-treated) to confirm specificity.
Visualizations
FRET vs Dye Probe Selection Workflow
Core Detection Mechanisms: FRET vs Chemical Reaction
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Relevance |
|---|---|
| Genetically-Encoded Biosensor Plasmids (e.g., from Addgene) | DNA constructs encoding FRET sensors for metabolites like glucose (FLII12Pglu), lactate (Laconic), ATP (ATeam), cAMP (Epac-based). Essential for stable cell line generation. |
| Cell-Permeant Synthetic Dyes (e.g., H2DCFDA, MitoSOX Red, JC-1) | Small-molecule fluorogenic probes for reactive species, mitochondrial potential, or specific ions. Enable measurement in cells without genetic manipulation. |
| Acetoxymethyl (AM) Ester Dyes | Chemical modification rendering polar dyes cell-permeant. Intracellular esterases cleave the AM groups, trapping the active dye inside the cell. |
| Ionophores & Calibration Kits (e.g., ionomycin, nigericin, high-K+ buffers) | Used in conjunction with ratiometric probes (dye or protein-based) to establish Rmin and Rmax for quantitative calibration of intracellular ion concentrations. |
| FRET Reference Standards (e.g., CFP-YFP tandem proteins) | Control constructs with fixed FRET efficiency. Critical for validating microscope FRET capability and correcting for spectral bleed-through. |
| Transfection/Transduction Reagents (e.g., lipofectamine, lentivirus) | For delivering and expressing genetically-encoded biosensors in mammalian cell lines, particularly those resistant to standard transfection. |
| Glass-Bottom Culture Dishes | Provide optimal optical clarity for high-resolution live-cell imaging, minimizing background fluorescence and distortion. |
| Environmental Control Systems (chamber, heater, CO2) | Maintain physiological conditions (37°C, 5% CO2, humidity) on the microscope stage during prolonged live-cell imaging experiments. |
1. Introduction Within the field of FRET biosensor metabolite detection research, achieving a comprehensive understanding of biomolecular interactions demands validation across multiple physical and temporal scales. Förster Resonance Energy Transfer (FRET) provides exquisite spatiotemporal resolution of dynamic processes in living cells but is inherently relative and context-dependent. Orthogonal methods—Surface Plasmon Resonance (SPR), Mass Spectrometry (MS), and Electrophysiology—provide complementary, absolute quantitative data on binding kinetics, stoichiometry, structural identity, and functional consequences. This guide details the strategic integration of these techniques to build a robust, multi-parametric framework for validating and interpreting FRET biosensor data, crucial for both fundamental research and drug development pipelines.
2. Core Principles of Method Integration
2.1 FRET Biosensor Context Genetically encoded FRET biosensors for metabolites (e.g., glucose, ATP, cAMP, glutamate) consist of a sensing domain flanked by donor (CFP, mCerulean) and acceptor (YFP, mCitrine) fluorescent proteins. Metabolite binding induces a conformational change altering FRET efficiency. While ideal for real-time, subcellular tracking, FRET readings require calibration and can be influenced by environmental factors (pH, Cl⁻ concentration). Orthogonal methods ground these observations in quantitative physical parameters.
2.2 Orthogonal Validation Logic
- SPR validates the binding kinetics (ka, kd, KD) and stoichiometry of the isolated sensing domain-metabolite interaction, confirming the sensor's inherent thermodynamic mechanism.
- MS (particularly Native MS or HDX-MS) identifies binding stoichiometry, modification states, and conformational dynamics of the sensor protein, providing structural corroboration.
- Electrophysiology (e.g., patch clamp) assesses the functional output of metabolite-gated ion channel-based FRET sensors or validates sensors in electrophysiologically active cells (e.g., neurons).
3. Detailed Methodologies & Protocols
3.1 FRET Imaging Protocol (Reference Experiment)
- Objective: Measure real-time cytosolic ATP:ADP ratio in HEK293 cells.
- Biosensor: PercevalHR (ATP:ADP ratio sensor).
- Protocol:
- Plate cells on glass-bottom dishes and transfect with PercevalHR cDNA.
- 24-48h post-transfection, acquire images on a ratiometric fluorescence microscope.
- Excitation: 430 nm (CFP excitation). Emission Collection: 470 nm (donor emission) and 535 nm (acceptor emission) using a beamsplitter.
- Calculate ratio R = I535 / I470 after background subtraction.
- Perform in situ calibration: Apply 10 μM oligomycin (inhibits ATP synthase) and 10 mM 2-deoxyglucose (inhibits glycolysis) to deplete ATP, recording Rmin. Apply 10 mM glucose and 5 μM rotenone to maximize mitochondrial ATP production, recording Rmax.
- Normalized ratio = (R - Rmin) / (Rmax - Rmin).
3.2 SPR Protocol for Binding Validation
- Objective: Determine kinetic parameters of the isolated sensing domain (e.g., Snf1 protein domain for sugar sensing) binding to its ligand.
- Protocol:
- Immobilize purified, his-tagged sensing domain on a Ni-NTA sensor chip.
- Flow analyte (metabolite) at increasing concentrations (e.g., 0.1, 0.5, 2.5, 10, 50 μM) in HBS-EP buffer at 30 μL/min.
- Record association (120 s) and dissociation (180 s) phases.
- Regenerate surface with 350 mM EDTA.
- Fit double-referenced sensorgrams to a 1:1 Langmuir binding model using software (e.g., Biacore Evaluation Software) to extract association (ka) and dissociation (kd) rate constants. KD = kd/ka.
3.3 Native Mass Spectrometry Protocol
- Objective: Confirm binding stoichiometry and complex integrity of the FRET biosensor's apo- and holo-forms.
- Protocol:
- Desalt purified biosensor protein into 200 mM ammonium acetate, pH 7.5.
- For holo-form, incubate with 5-fold molar excess of metabolite.
- Introduce sample via nano-electrospray ionization (nano-ESI) into a high-mass Q-TOF or Orbitrap mass spectrometer.
- Use gentle source conditions (low collision energy, ~50 V) to preserve non-covalent complexes.
- Deconvolute mass spectra to determine molecular weights of apo-protein and protein-metabolite complex(es).
3.4 Electrophysiology Integration Protocol
- Objective: Correlate metabolite-induced FRET changes with membrane potential in a voltage-sensitive FRET (VSFP) or ion channel-linked sensor.
- Protocol:
- Co-transfect cells with the metabolite FRET biosensor and a constitutive plasma membrane marker (e.g., mem-mCherry).
- Perform simultaneous dual-optical and whole-cell patch-clamp recording.
- Voltage-clamp at -70 mV. Apply metabolite via a fast perfusion system.
- Record concurrently: a) FRET emission ratio, and b) membrane current or voltage changes.
- Analyze temporal correlation between FRET transition and electrophysiological response.
4. Quantitative Data Synthesis (Tables)
Table 1: Comparative Outputs of Integrated Techniques
| Method | Primary Output | Typical Resolution | Throughput | Sample Context |
|---|---|---|---|---|
| FRET Imaging | Dynamic Ratio Change (ΔR/R0) | Temporal: ms-s; Spatial: μm | Medium-High | Living Cells, Tissues |
| Surface Plasmon Resonance | Binding Kinetics (KD, ka, kd) | -- | Medium | Purified Protein / Domain |
| Native Mass Spectrometry | Molecular Mass, Stoichiometry | Mass Accuracy: < 0.01% | Low | Purified Protein / Complex |
| Patch-Clamp Electrophysiology | Membrane Current (pA) / Potential (mV) | Temporal: ms; Current: pA | Low | Living Cells |
Table 2: Example Integrated Dataset for a cAMP FRET Biosensor (Epac-SH150)
| Parameter | FRET (FLIM) | SPR (Biacore 8K) | Native MS (Q-TOF) | Electrophysiology (Patch Clamp) |
|---|---|---|---|---|
| Measured Output | τ (Donor Lifetime) decrease from 3.5 ns to 2.8 ns | KD = 9.3 ± 1.2 μM, ka = 2.1e5 M⁻¹s⁻¹ | Apo: 75,432 Da; +cAMP: + 329 Da (1:1 complex) | cAMP-induced K⁺ current shift of +15 pA (in relevant cell line) |
| Sample Prep | Live HEK293 cells | Immobilized Epac CBD domain on CMS chip | Epac CBD in ammonium acetate | HEK293 cells co-expressing GIRK channel |
| Key Validation | In-cell cAMP-induced conformational change | High-affinity, specific 1:1 binding confirmed | 1:1 binding stoichiometry confirmed | Functional coupling to downstream effector validated |
5. The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function / Application |
|---|---|
| Genetically Encoded FRET Biosensor Plasmids (e.g., ATeam for ATP, iGluSnFR for glutamate) | Provide the foundational molecular tool for live-cell metabolite imaging. |
| High-Purity, Recombinant Sensor Domain Protein | Essential for in vitro validation via SPR and MS. Requires >95% purity. |
| Series S Sensor Chips (e.g., Ni-NTA, CM5) | SPR chip surfaces for immobilizing his-tagged proteins or via amine coupling. |
| Ammonium Acetate (MS Grade) | Volatile buffer for native MS sample preparation, preserving non-covalent interactions. |
| Ion Channel / Electrophysiology Cell Lines (e.g., HEK293T, CHO, neuronal lines) | Provide a consistent cellular background for combined optical/electrical recordings. |
| Metabolite Agonists/Antagonists & Pharmacological Tools (e.g., oligomycin, forskolin, CNQX) | For sensor calibration, pathway modulation, and experimental controls. |
| Gentle Ag/Ab Immobilization Kits (for SPR) | For oriented immobilization of sensor proteins, minimizing denaturation. |
| Cell-Permeant and -Impermeant Metabolite Analogs | Allow controlled manipulation of intracellular vs. extracellular metabolite pools. |
6. Integrated Workflow and Pathway Visualizations
Title: Orthogonal Validation Cycle for FRET Biosensors
Title: Sequential Integrated Experimental Workflow
Title: Biosensor Mechanism & Orthogonal Validation Points
7. Conclusion The integration of FRET with SPR, MS, and Electrophysiology moves biosensor-based metabolite detection from qualitative observation to a quantitatively rigorous discipline. This multi-method framework deconvolutes the complex variables influencing FRET signals, providing cross-validated parameters for binding, structure, and function. For drug development, this approach strengthens the mechanistic link between target engagement (validated by SPR/MS) and functional cellular response (validated by FRET/EP), derisking the translation of biosensor discoveries into therapeutic strategies. The future lies in increasingly simultaneous, rather than sequential, application of these techniques.
Assessing Reproducability and Translational Potential for Preclinical Studies
The preclinical research pipeline, particularly in metabolite detection using Förster Resonance Energy Transfer (FRET) biosensors, faces a well-documented reproducibility crisis. This directly impedes translational potential—the likelihood that findings will successfully transition to clinical applications. Within FRET biosensor development for metabolites (e.g., glucose, ATP, cAMP, glutamate), challenges such as sensor fidelity in vivo, dynamic range variability between cell types, and inconsistent calibration methodologies contribute to irreproducible results. This whitepaper provides a technical framework for rigorously assessing and enhancing both reproducibility and translational potential in this specialized field.
Key Pillars of Reproducibility in FRET Biosensor Studies
Biosensor Design and Validation
A biosensor's core components must be fully characterized. This includes the donor/acceptor fluorophore pair (e.g., CFP/YFP, mTurquoise2/sYFP2), the metabolite-binding domain specificity, and the linker sensitivity. Reproducibility requires detailed reporting of these elements.
Experimental and Analytical Standardization
Variability in microscopy settings, calibration protocols, and data analysis algorithms is a major source of irreproducibility.
Biological Model and Reporting
The choice of cell line, culture conditions, animal model, and in vivo delivery method profoundly impacts results.
Table 1: Quantitative Summary of Common Reproducibility Pitfalls in FRET Biosensor Studies
| Pitfall Category | Specific Issue | Reported Impact on Data Variability | Recommended Mitigation |
|---|---|---|---|
| Microscopy & Acquisition | Inconsistent excitation intensity | >30% signal variance | Use power meter, report mW/mm² |
| Inconsistent camera settings (gain, binning) | Alters signal-to-noise ratio | Standardize EM gain; report bit-depth | |
| Temperature fluctuation during live-cell imaging | Alters metabolic rates & kinetics | Use environmental chamber; report ±0.5°C | |
| Calibration | Use of different calibration methods (e.g., ionomycin vs. digitonin) | K_d apparent can vary by >50% | Report full method; use internal in situ standards |
| Variable intracellular milieu (pH, [Mg2+]) | Alerts biosensor dynamic range | Co-image with rationetric pH or ion sensors | |
| Data Analysis | Inconsistent background subtraction | Can introduce >20% error in ratio | Image cell-free region; document method |
| Use of different ratio metrics (e.g., 480/535 vs. FRET/CFP) | Hinders cross-study comparison | Report raw channels and final calculated ratio | |
| Biological Model | Cell passage number & confluence | Alerts basal metabolite levels | Report passage # (<30); standardize confluence |
| Serum starvation or media composition | Drastically changes metabolic state | Report exact media formulation and time |
Detailed Experimental Protocols for Critical Assays
Protocol:In SituCalibration of a Genetically Encoded FRET Biosensor (e.g., ATP:ATeam)
Objective: To determine the dynamic range (Rmin, Rmax) and apparent K_d of the biosensor within the cellular environment.
Materials:
- Cells expressing the FRET biosensor (e.g., HEK293T expressing ATeam).
- Imaging buffer: e.g., Hanks' Balanced Salt Solution (HBSS) or culture medium without phenol red.
- Calibration reagents:
- For Rmax (zero metabolite): 10 mM 2-Deoxy-D-glucose (2-DG) in buffer, 10 mM Sodium Azide (NaN3) in buffer. CAUTION: Toxic.
- For Rmin (saturating metabolite): 10 µM Oligomycin in DMSO, 50 mM Glucose in buffer.
- Permeabilization agent: 0.1% (w/v) Digitonin in buffer (prepared fresh) OR 10 µM Ionophore (e.g., Ionomycin for Ca2+ sensors).
- FRET-capable microscope with stable temperature control (37°C) and CO2 (if using bicarbonate buffer).
Procedure:
- Image Acquisition: Plate cells on glass-bottom dishes. Acquire a 2-5 minute baseline FRET ratio (e.g., YFP/CFP emission upon CFP excitation) in imaging buffer.
- Inhibit Production & Deplete Metabolite (Rmax):
- Add 2-DG (final 1 mM) and NaN3 (final 1 mM) to inhibit glycolysis and oxidative phosphorylation, respectively.
- Incubate for 30-60 minutes until the FRET ratio stabilizes at a high value (low ATP). Record this ratio as Rmax.
- Permeabilize and Saturate (Rmin):
- Add digitonin (final 10 µg/mL) to permeabilize the plasma membrane. Incubate 2 min.
- Wash with an ATP-depletion buffer (containing hexokinase/glucose) to remove residual cytosolic ATP.
- Add a solution containing saturating Mg-ATP (e.g., 10 mM) and oligomycin (inhibits ATP synthase, preventing consumption). The ratio will drop to a low stable value (high ATP). Record this as Rmin.
- Data Calculation: The normalized FRET ratio (Rnorm) for any data point (R) is: Rnorm = (R - Rmin) / (Rmax - Rmin). The apparent Kd can be fitted from a titration curve.
Protocol: Orthogonal Validation of Biosensor Readouts using LC-MS/MS
Objective: To correlate FRET ratio changes with absolute metabolite concentrations measured by mass spectrometry.
Materials:
- Biosensor-expressing cells in multi-well plates (for parallel sampling).
- Rapid quenching solution: 40:40:20 Methanol:Acetonitrile:Water at -20°C.
- Extraction buffer: 80% methanol with internal standards (e.g., ¹³C-labeled metabolites).
- LC-MS/MS system with appropriate column (e.g., HILIC for polar metabolites).
Procedure:
- Synchronized Experiment: Run two identical plates in parallel: one for live-cell FRET imaging, one for metabolite extraction.
- Rapid Metabolite Quenching: At key time points (e.g., baseline, after stimulus), immediately aspirate media from the extraction plate and add quenching solution. Scrape cells on dry ice.
- Metabolite Extraction: Vortex, centrifuge (16,000g, 15 min, 4°C). Transfer supernatant, dry in a speedvac, and reconstitute in LC-MS compatible solvent.
- LC-MS/MS Analysis: Run samples to quantify absolute concentrations of the target metabolite (e.g., ATP, ADP/ATP ratio) and related pathway metabolites.
- Correlation Analysis: Plot FRET ratio (from imaging plate) against LC-MS/MS quantified concentration (from extraction plate) at matched time points. Perform linear or non-linear regression to validate the biosensor's quantitative accuracy in situ.
Visualizing Workflows and Pathways
Diagram Title: FRET Biosensor Metabolite Sensing Mechanism
Diagram Title: Reproducible FRET Biosensor Development Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for FRET Biosensor Metabolite Detection
| Item | Function/Description | Example Product/Catalog | Critical for Reproducibility |
|---|---|---|---|
| FRET Biosensor Plasmids | Genetically encoded sensors for specific metabolites (e.g., ATP, glucose, lactate). | ATeam (ATP), iGLIM (glucose), Laconic (lactate). | Use standard, cited constructs from Addgene; report exact variant and accession #. |
| High-Fidelity Polymerase | For error-free amplification of biosensor constructs for subcloning or viral production. | Q5 High-Fidelity DNA Polymerase, Phusion. | Reduces sequence variability in expressed sensors. |
| Transfection/Gene Delivery | For consistent biosensor expression in target cells. | Polyethylenimine (PEI), Lipofectamine 3000, Lentiviral Particles. | Standardize reagent:DNA ratio, incubation time, and expression duration (e.g., 24-48h). |
| Phenol Red-Free Medium | For live-cell imaging to avoid autofluorescence. | Gibco FluoroBrite DMEM. | Essential for consistent background signal across experiments. |
| Environmental Chamber | Maintains stable temperature (37°C), humidity, and CO2 during live imaging. | Okolab Stage Top Incubator. | Critical for cell health and metabolic stability. |
| Calibration Kit Reagents | Standardized chemicals for in situ biosensor calibration (e.g., ionophores, inhibitors). | Cayman Chemical Metabolite Calibration Kits (where available) or high-purity 2-DG, Oligomycin, Digitonin. | Use high-purity lots and report exact concentrations, sources, and batch numbers. |
| Fluorophore Standards | Reference slides or beads for daily microscope calibration and alignment. | TetraSpeck Microspheres, FocalCheck Slides. | Ensures instrument performance consistency over time and between labs. |
| Metabolite Extraction Kit | For orthogonal validation via mass spectrometry. | Biocrates AbsoluteIDQ p180 Kit or MeOH/ACN/H2O quench. | Standardizes sample preparation for cross-platform comparability. |
| Automated Analysis Software | For consistent, unbiased calculation of FRET ratios from image stacks. | ImageJ/Fiji with custom macros, CellProfiler, or commercial packages like MetaMorph. | Document all parameters, thresholds, and background subtraction methods. |
Conclusion
FRET biosensors have revolutionized our ability to interrogate metabolite dynamics with unparalleled spatiotemporal resolution in living systems. From understanding the foundational design principles to implementing robust methodological workflows, researchers can now capture metabolic flux in real-time, offering insights static assays cannot provide. Successful application requires careful optimization to overcome technical hurdles related to signal fidelity and cellular perturbation, and rigorous validation against established techniques remains paramount. As sensor design evolves with improved brightness, specificity, and multiplexing capabilities, their integration into drug discovery pipelines will accelerate. Future directions point toward high-throughput in vivo imaging, multiplexed detection of several metabolites simultaneously, and the development of clinically translatable sensors for diagnostic applications. Ultimately, FRET biosensors are not just tools for observation but powerful engines for driving discovery in metabolic disease, oncology, and neuropharmacology.